Chapter 4 Arrangement of Electrons in Atoms Properties





































![Problem • Which element does the noble-gas notation [Ne]3 s 2 represent the electron Problem • Which element does the noble-gas notation [Ne]3 s 2 represent the electron](https://slidetodoc.com/presentation_image_h2/16d25183476d16a773f8679f4ae957f4/image-48.jpg)



- Slides: 51

Chapter 4 Arrangement of Electrons in Atoms

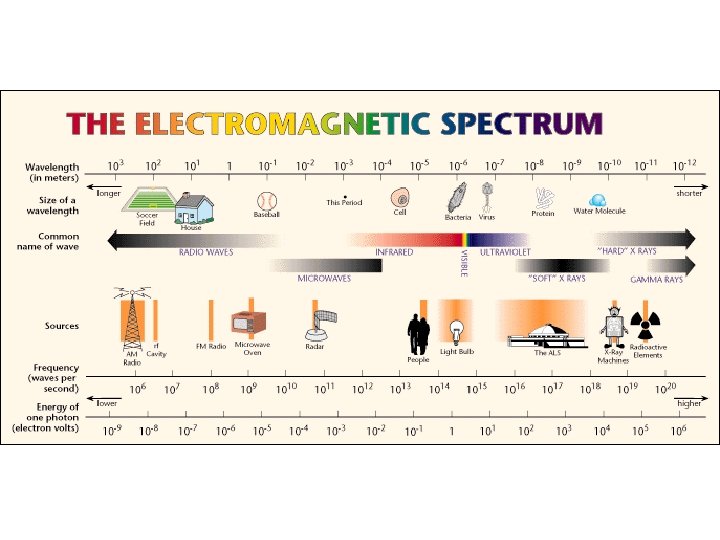
Properties of Light • Electromagnetic radiation (ER) • Form of energy the exhibits wavelike behavior • All ER forms electromagnetic spectrum • Travel at speeds of 3. 0 x 108 m/s through air • c


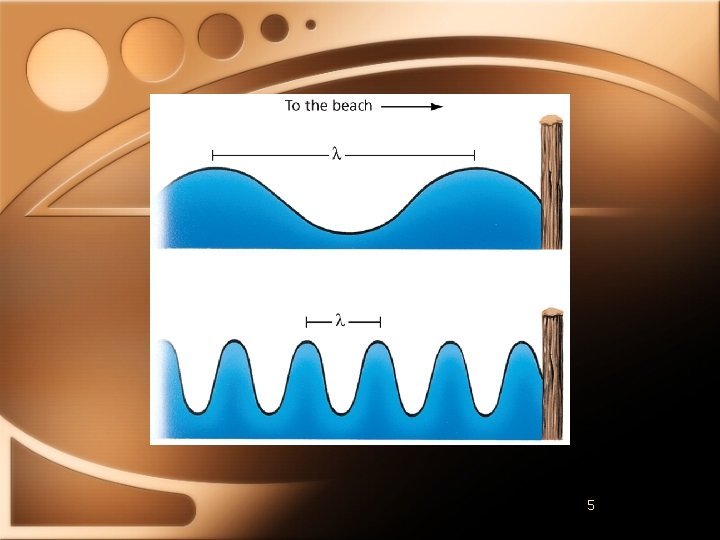
Properties of Light • Wavelength (λ ) • Distance between troughs or peaks • Frequency (ν ) • Number of waves in a specific time • Waves/second • Hertz (Hz) • c= λν • C=3. 0 x 108 m/s

5

Problems • Given the speed of light as 3. 00 x 108 m/s, calculate the wavelength of the electromagnetic radiation whose frequency is 7. 500 x 1012 Hz.

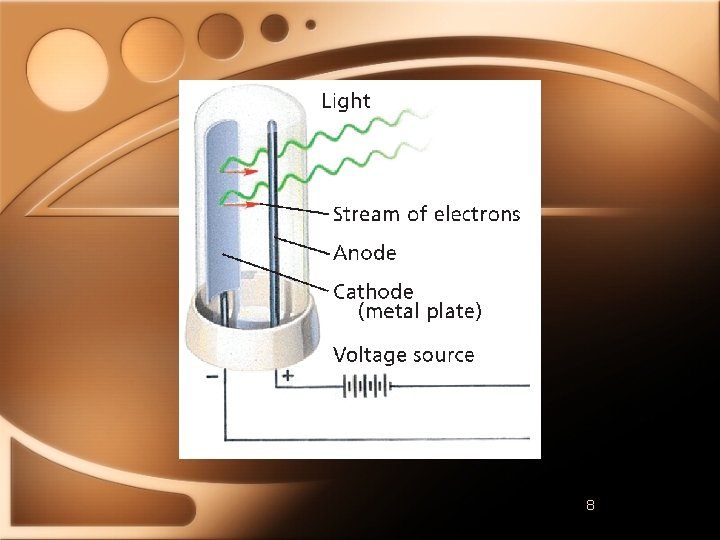
The Photoelectric Effect • Emission of electrons from metal when light shines on it • Depended on frequency of light • Max Planck • Emits energy in small, specific amounts →Quanta • Quantum is minimum quantity of energy that can be lost or gained

8

Light as Particles • E=hν • h→Planck’s constant=6. 626 x 10 -34 J⋅ s • E - energy of light • Albert Einstein • Dual wave-particle nature • Photon →particle of ER that carries a quantum of energy • Ephoton = hν

Problems • What is the frequency of a radio wave whose energy is 1. 55 x 10 -24 J per photon? • When sodium is heated, a yellow spectral line whose energy is 3. 37 x 10 -19 J per photon is produced. • What is the frequency of this light? • What is the wavelength of this light?

Light as Particles • Only whole numbers of photons are absorbed • For e- to be emitted, must be struck by photon with minimum energy for that material • Corresponds to frequency

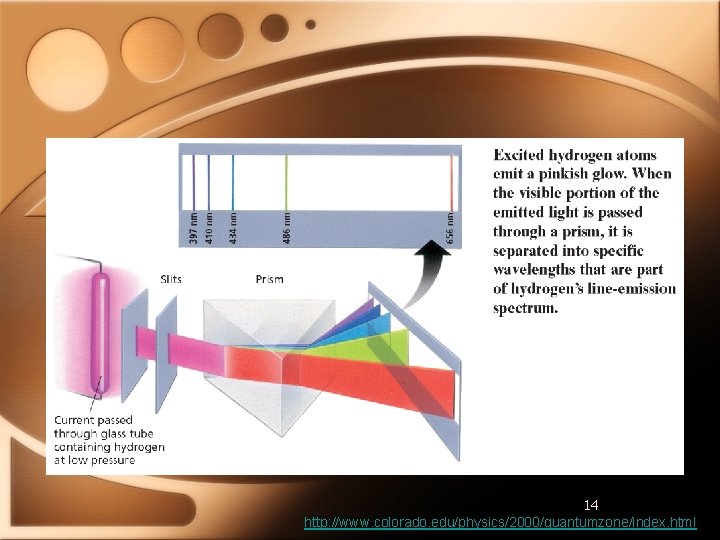
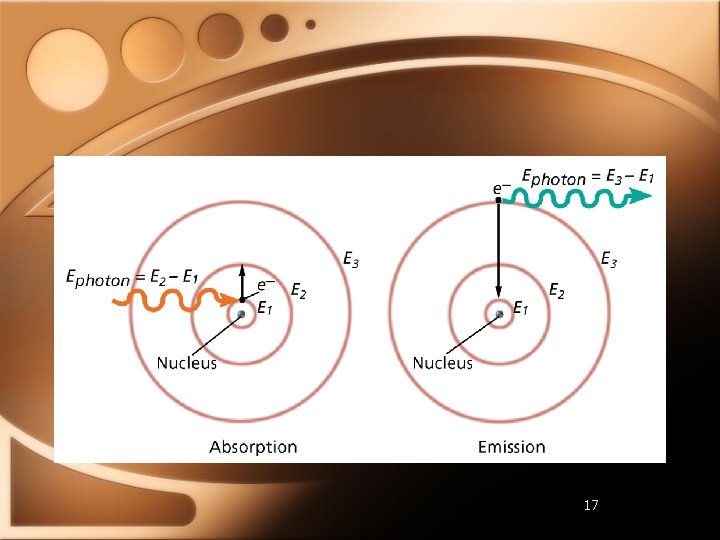
Hydrogen Line-Emission Spectrum • Ground state - lowest energy state of an atom • Excited state - atom has higher potential energy than ground state • Atom gives off energy when returning to ground state

Hydrogen Line-Emission Spectrum • Hydrogen emits pink light • Specific line-emission spectrum • Quantum theory • Electrons of atoms exists only in very specific energy states • Each element emits spectra in 3 regions: • Ultraviolet (Lyman series) • Visible (Balmer series) • Infrared (Paschen series)

14 http: //www. colorado. edu/physics/2000/quantumzone/index. html

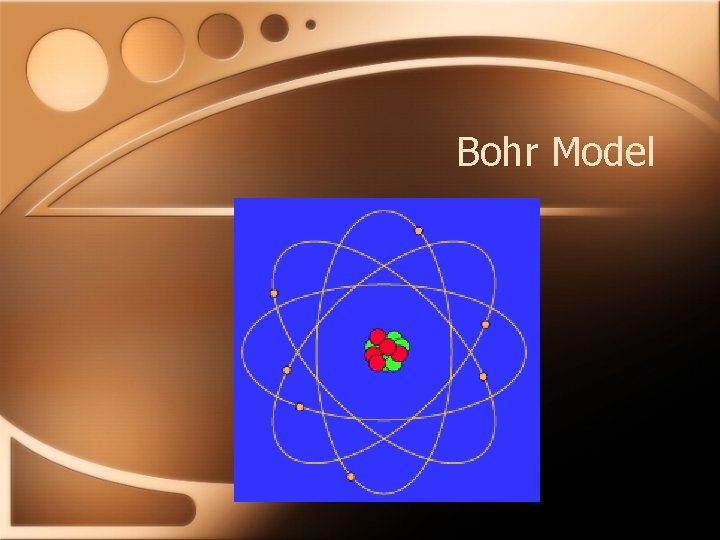
Bohr Model • Explaining line-spectrums • Electron circles in specified orbits • Lowest energy closest to nucleus • Electrons able to move from one orbit to another • Photon emitted when dropping from higher orbit to lower • Explains spectrums of atoms with only one e-

Bohr Model

17

Electrons as Waves • De Broglie • Electrons have wavelike properties • Electrons can be diffracted (bent) like light • Electrons interfere with each other like light

Heisenberg Uncertainty Principle • Impossible to determine simultaneously both the position and velocity of electron • Like airplane propeller

Schrödinger Wave Equation • Quantum theory →mathematically describes wave properties of e • Electrons do not travel in neat orbits, but in orbitals • 3 -dimentions region that indicates probable location e-

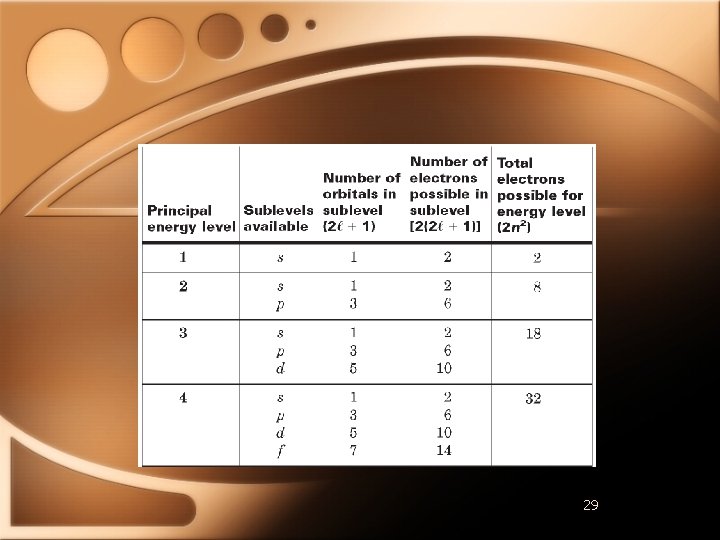
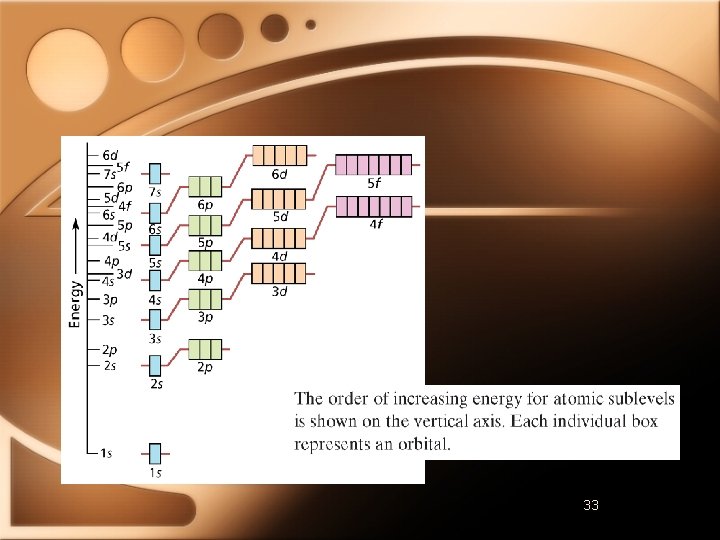
Quantum Numbers • Specify properties of atomic orbitals and properties of e- in orbitals • Principal Quantum Number • • Main energy level occupied by en As n increases so does e- energy Electron shell

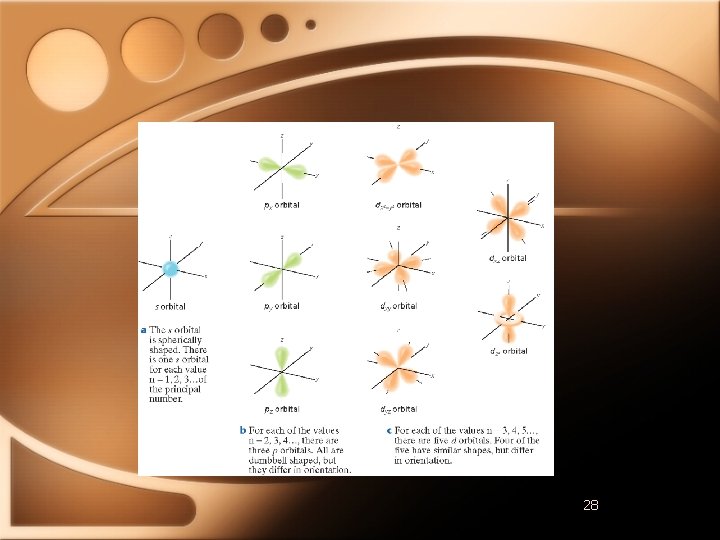
Quantum Numbers • Angular Momentum Quantum Number • • Orbitals have different shapes l Positive integers from 0 to n-1 EXAMPLE • n=2 • l=0 and l=1

Quantum Numbers l 0 1 2 3 Letter s p d f http: //winter. group. shef. ac. uk/orbitron/AOs/ 1 s/index. html

Quantum Numbers • • • For n=1, only one sublevel possible (s) For n=2, two sublevels possible (s & p) For n=3, three sublevels possible (s, p, & d) For n=4, four sublevels possible (s, p, d, & f) Atomic orbitals named by principal quantum number followed by letter of sublevel • 1 s 2 p 4 d

Quantum Numbers • Magnetic Quantum Number (m) • Describes the orientation of orbital around nucleus • S sublevel • Spherical - one possible orientation • P sublevel • Lobes extend along x, y, or z axis around nucleus • 3 p orbital in each p sublevel

Quantum Numbers • D Sublevel • 5 different d orbitals in d sublevel

Quantum Numbers • Electron is spinning on an axis • Spin Quantum Number • 2 possible values (+1/2 or -1/2) • A single orbital can only hold 2 electrons with opposite spins

28

29

30

Problems • When n=2, how many sublevels are possible? How many orbitals?

Electron Configurations • 1 s 22 p 2 - electron configuration of carbon • Arrangement of electrons in an atom • Ground-state configuration - lowest energy

33

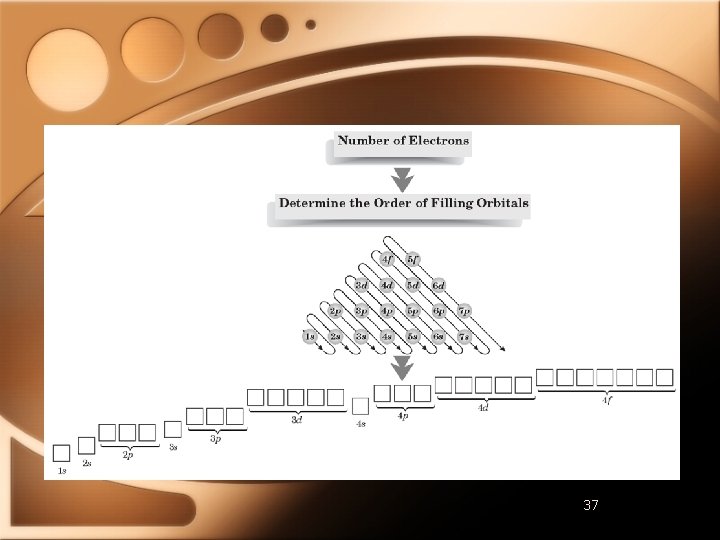
Electron Configuration Rules 1. Electron occupies lowest-energy orbital possible • Aufbau principle 2. No two electrons in atom can have same set of four quantum numbers • Pauli exclusion principle 3. Orbitals of equal energy are each occupied by one electron before another is added • Hund’s rule

Representing Electron Configurations • Orbital Notation • Unoccupied orbital is represented by a line, __, with orbital name underneath • Orbital containing 1 e- _↑ _ • Orbital containing 2 e- ↓↑ • H _↑ He ↓↑ • 1 s 1 s

Representing Electron Configurations • Electron-Configuration Notation • • Eliminates lines and arrows Number of electrons shown as a superscript Hydrogen - 1 s 1 Helium - 1 s 2

37

Problem • The electron configuration of boron is 1 s 22 p 1. • How many electrons are present in an atom of boron? • What is the atomic number for boron? • Write the orbital notation for boron.

Problem • The electron configuration of carbon is 1 s 22 p 2. • How many electrons are present in a carbon atom? • What is the atomic number of carbon? • Write the orbital notation for carbon.

Problem • Write the electron configuration of the element sulfur, which has an atomic number of 16. • Write the orbital notation for sulfur. • How many unpaired electrons does a sulfur atom contain?

Elements of the Second Period • Electrons start filling the s sublevel in the 2 nd main energy level • Lithium - 1 s 22 s 1 • 2 s is the highest occupied level • 1 s is an inner-shell • Beryllium - 1 s 22 s 2 • Boron - 1 s 22 p 1

Elements of the Third Period • Start filling s sublevel of 3 rd energy level • Sodium - 1 s 22 p 63 s 1 • Noble-Gas Notation • Noble gas in brackets represents complete noble gas configuration • Add remaining electrons • [Ne]3 s 1

Elements of the Fourth Period • Begin filling 4 s orbital • 3 d sublevel is next to be filled • 4 p sublevel is then filled

Elements of the Fifth Period • Sublevels filled in this order: • 5 s • 4 d • 5 p

Problem • Write both the complete electron-configuration notation and the noble-gas notation for iron, Fe. • How many electron-containing orbitals are in an atom of iron? How many of these orbitals are completely filled? How many unpaired electrons are there in an atom of iron? In which sublevel are the unpaired electrons located?

Problem • Write both the complete electronconfiguration notation and the noble-gas notation for zirconium, Zr. • How many electron-containing orbitals are in an atom of zirconium? How many of these orbitals are filled? How many unpaired electrons are there in an atom of zirconium?

Problem • Identify the element whose atoms have two electrons in the p sublevel of their second main energy level. • What is the total number of electrons in the second main energy level of an atom of this element? • Name the element in the third period that has the same number of electrons in its outermost main energy level as this element.
![Problem Which element does the noblegas notation Ne3 s 2 represent the electron Problem • Which element does the noble-gas notation [Ne]3 s 2 represent the electron](https://slidetodoc.com/presentation_image_h2/16d25183476d16a773f8679f4ae957f4/image-48.jpg)
Problem • Which element does the noble-gas notation [Ne]3 s 2 represent the electron configuration of? • How many inner-shell electrons do its atoms contain?

Elements of the Sixth Period • Electrons fill orbitals in this order: • • • 6 s 5 d 4 f 5 d 6 p

Problem • Write the noble-gas notation for the mercury atom. • Write the noble-gas notation for the lead atom.

Chapter Review • Pg. 124 • 1, 4, 10, 11, 12, 13, 18, 22, 23, 24, 25, 30 cde, 31 bd, 32 d, 33, 35 c, 38 beg