Chapter 4 Arrangement of Electrons in Atoms I
























- Slides: 30

Chapter 4 Arrangement of Electrons in Atoms

I. The Development of a New Atomic Model H Electromagnetic Radiation: H “radiant energy” form of nrg that has wave characteristics and can travel through a vacuum “light” H Electromagnetic Spectrum: H Distribution among various wavelengths of the radiant nrg emitted or absorbed by an object

H Wavelength ( ): corresponding points on adjacent waves---Ex: Frequency ( ): # of waves that pass a point in a specific time H c = ( ) ------inversely proportional

H c = ( ) ------inversely proportional c : m/s : m, cm, nm : waves/second--Hertz (Hz)

H Photoelectric Effect: emission of e- by certain metals when light shines on them

H Quantum: min quantity of nrg that can be lost or gained by an atom H E = (h) ( ) o J = (J s) (Hz) o Planck’s constant: 6. 626 X 10 -34 J s

Video #15 (wave function and wave particle) • Einstein o H dual wave-particle to describe light Photon: radiation with zero mass carrying a quantum of nrg o packet of nrg emitted when an e- drops nrg levels

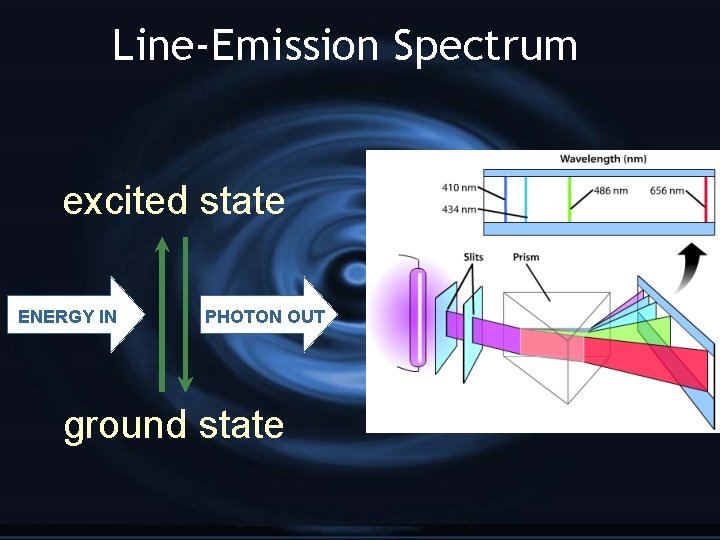
H Ground state: lowest nrg state H Excited state: higher potential nrg

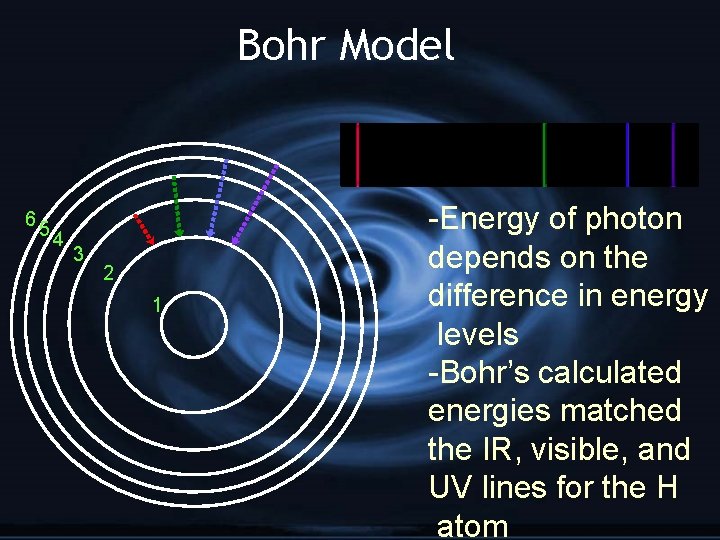
--Bohr’s Model-G e- exist only in orbits with specific amounts of energy called energy levels G Therefore… G e- can only gain or lose certain amounts of energy G only certain photons are produced

Line-Emission Spectrum excited state ENERGY IN PHOTON OUT ground state

Bohr Model 65 4 3 2 1 -Energy of photon depends on the difference in energy levels -Bohr’s calculated energies matched the IR, visible, and UV lines for the H atom

Other Elements G Each element has a unique bright-line emission spectrum. G “Atomic Fingerprint” Helium z. Bohr’s calculations only worked for hydrogen! ----pg 97

II. The Quantum Model of the Atom G A. Electrons as Waves o Diffraction: bending of a wave as it passes by the edge of an object o Interference: results when waves overlap

EVIDENCE: DIFFRACTION PATTERNS VISIBLE LIGHT ELECTRONS

Video # 13(What is the Heisenberg Uncert) z. Heisenberg Uncertainty Principle y. Impossible to know both the velocity and position of an electron at the same time

Video #14(Quantum Mechanics: Schrod) G Schrödinger Wave Equation (1926) G finite # of solutions quantized energy levels G defines probability of finding an e-

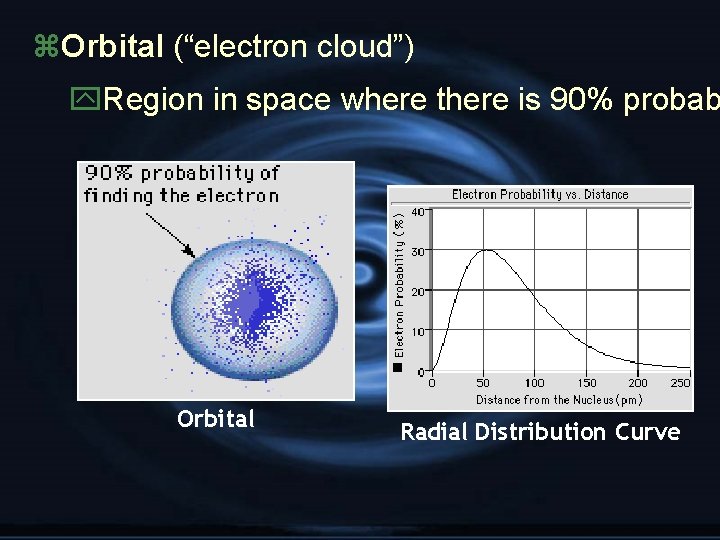
A. Atomic Orbitals and Quantum Numbers G Orbital: probable location of an e. G Quantum #: properties of atomic orbitals and properties of e-’s in orbitals G Principal quantum #: (n), indicates main nrg level occupied by the eo n = 1 -----occupies 1 st nrg level

G Angular momentum quantum #: (l), indicates shape of orbital G Magnetic quantum #: (m), orientation of an orbital G Spin quantum #: which spin state (+)(-) G ***See table 4 -2 pg 104

z. Orbital (“electron cloud”) y. Region in space where there is 90% probab Orbital Radial Distribution Curve

z. Four Quantum Numbers: y. Specify the “address” of each electron in an UPPER LEVEL

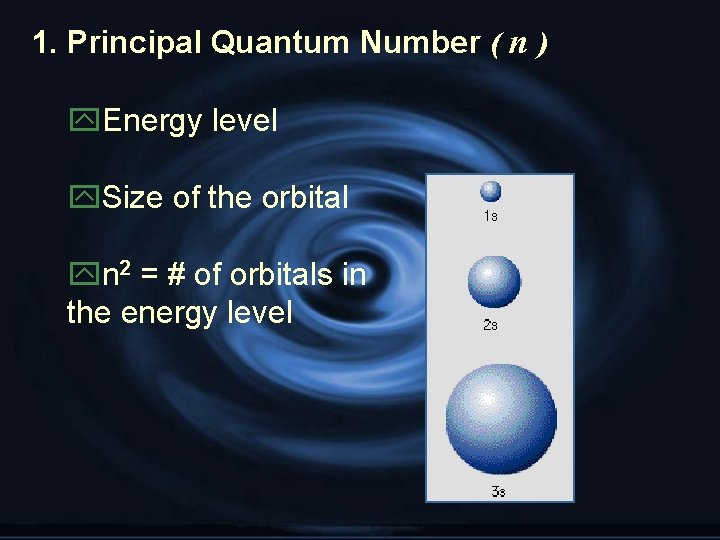
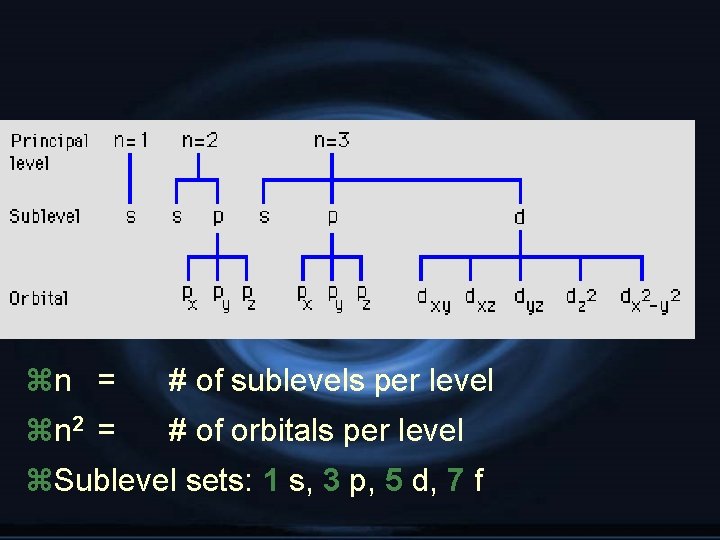
1. Principal Quantum Number ( n ) y. Energy level y. Size of the orbital yn 2 = # of orbitals in the energy level

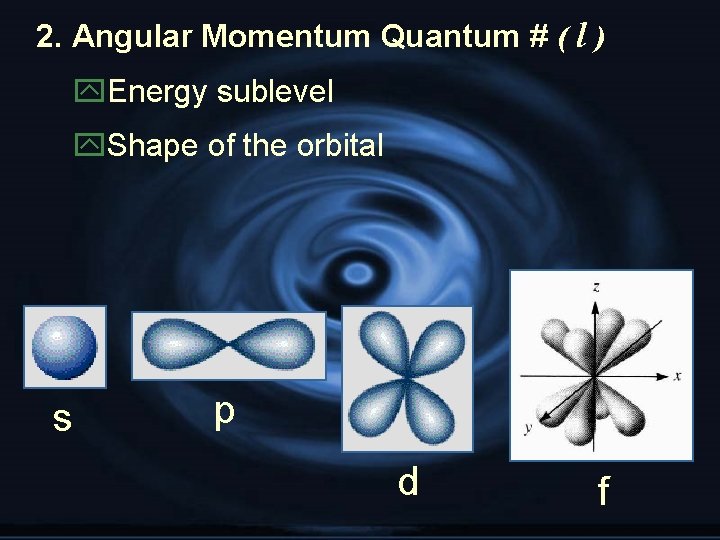
2. Angular Momentum Quantum # ( l ) y. Energy sublevel y. Shape of the orbital s p d f

zn = # of sublevels per level zn 2 = # of orbitals per level z. Sublevel sets: 1 s, 3 p, 5 d, 7 f

3. Magnetic Quantum Number ( ml ) y. Orientation of orbital y. Specifies the exact orbital within each sublevel

4. Spin Quantum Number ( ms ) y. Electron spin +½ or -½ y. An orbital can hold 2 electrons that spin in opposite directions.

III. Electron Configuration G Aufbau principle: lowest nrg orbits fill first G Pauli exclusion principle: no 2 e-’s can have the same 4 quantum #’s. This is where spin allows 2 e-’s to be in the same orbit o Ex:

G Hund’s rule: orbital of equal nrg are occupied by 1 e-, before any is occupied by 2 e-’s o Ex: G Orbital Notation: ex: pg 107 G Electron Config Notation: pg 107 G Electron Dot diagram: ex

G Noble gases: G are inert G complete octet G --show ex----

G Table 4 -3 pg 110 1. 2. 3. 4. Principal # energy level Ang. Mom. # sublevel (s, p, d, f) Magnetic # orbital Spin # electron

Feeling overwhelmed?