Chapter 5 Arrangement of Electrons in Atoms Light





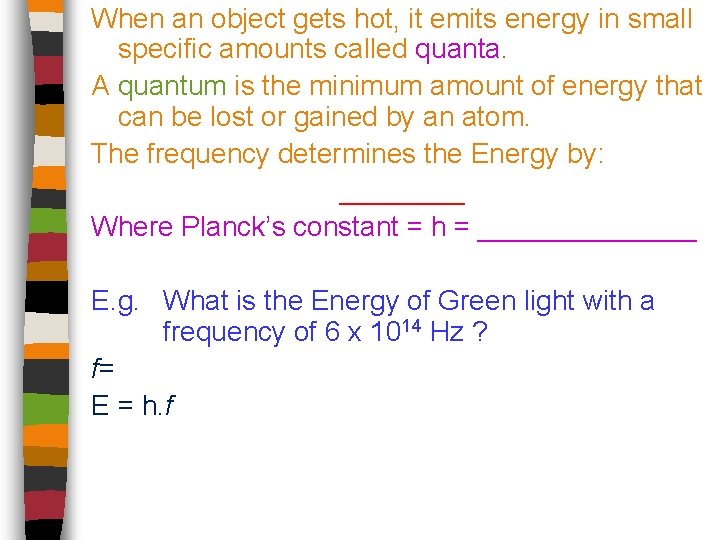

















- Slides: 32

Chapter 5 Arrangement of Electrons in Atoms

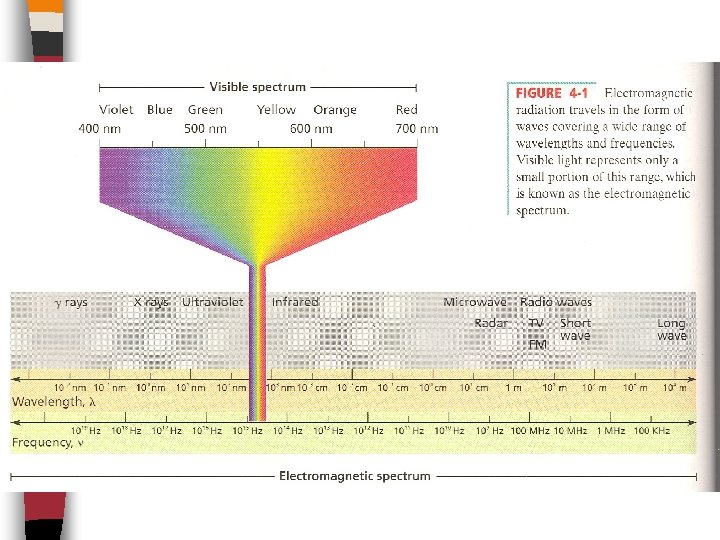
Light Dual Nature of Light: Light can act like waves, and as straight line particles. Light is one type of _______which is a form of Energy that has wavelike behavior Other types of em radiation are: x-rays, uv, infrared, microwaves & radio, and together they all form the _______________ (look at p. 92 Figure 4 -1 on next slide)


The Wave The speed of all em waves through a vacuum (space) and through air is: ________________ The length of each individual wave is known as it’s _________ ( ) which is the length between corresponding points on adjacent waves, usually measured in ______ (1 nm = 10 -9 m) The _______ (f or ) is how many waves pass a particular point in a second and is measured in waves/second = _____ (Hz)

The relationship between them is as follows: c=fx E. g. What is the wavelength & color of light that has a frequency of 6 x 1014 Hz (1/s)? f= c= = c=fx

If em radiation is directed towards a piece of metal, it may have enough energy to knock out electrons. This is known as the ____________. A German physicist: Max Planck, explained the reason why, but first let’s look at what he discovered.
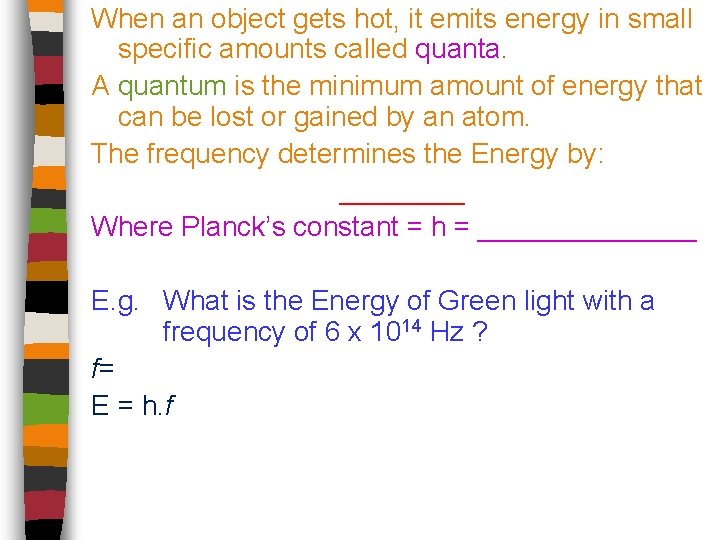
When an object gets hot, it emits energy in small specific amounts called quanta. A quantum is the minimum amount of energy that can be lost or gained by an atom. The frequency determines the Energy by: ____ Where Planck’s constant = h = _______ E. g. What is the Energy of Green light with a frequency of 6 x 1014 Hz ? f= E = h. f

It was Einstein that proposed that light acted like a stream of particles called photons. A ______ is a particle of em radiation that has zero mass and carrying a _______ of energy There must be a minimum amount of energy to eject electrons from a metal _____, Planck realized that the em radiation providing the energy, must be of a certain frequency. Different metals need different minimum amounts of energy, and therefore frequencies

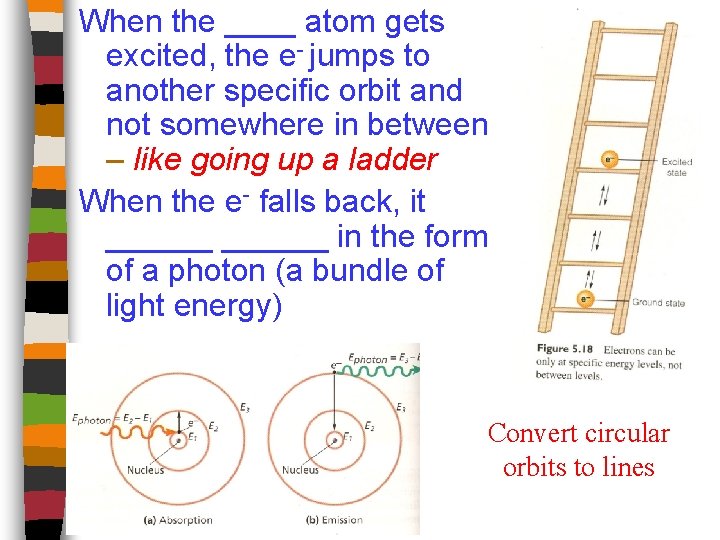
The Hydrogen Atom Consider the H atom with its one electron spinning around its one proton. When the electron is closest to the proton/nucleus which is at its lowest energy state, the atom is in its ground state. When energy, like heat, is supplied to the atom, the electron jumps to a higher orbit which is at a higher energy state. The atom is now in an excited state.

When the electron ______________________ __or a lower energy state, it emits a photon of radiation with a specific amount of energy, and not continuous amounts of energy. Therefore, specific _______ of light are emitted when excited Hydrogen “cools” down.

Bohr Model Niels Bohr (Danish) proposed a model that stated: “the e- of a H atom can circle the nucleus in allowed paths called _____” Between the orbit and the nucleus, is a large empty space where the e______ exist

When the ____ atom gets excited, the e- jumps to another specific orbit and not somewhere in between – like going up a ladder When the e- falls back, it ______ in the form of a photon (a bundle of light energy) Convert circular orbits to lines

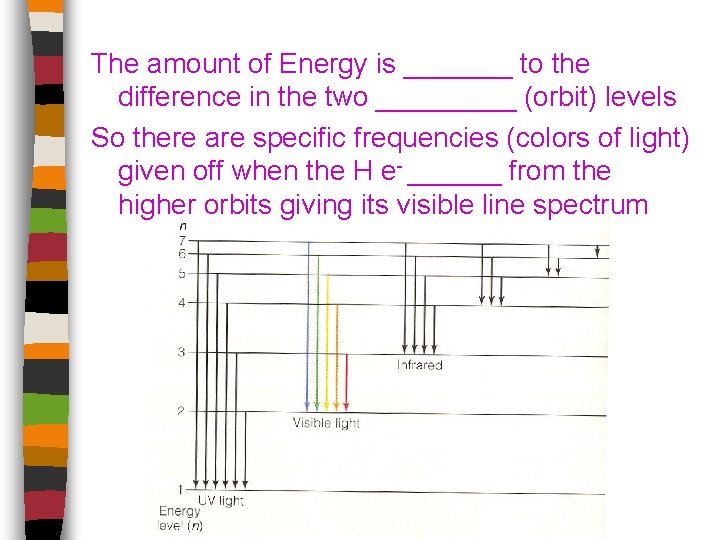
The amount of Energy is _______ to the difference in the two _____ (orbit) levels So there are specific frequencies (colors of light) given off when the H e- ______ from the higher orbits giving its visible line spectrum

Quantum Model of Atom Question is: Why can’t e-’s be in an orbit between the ____ Energy levels? French scientist Louis De. Broglie pointed out that the electron orbits acted like the behavior of waves. i. e. you can only have a certain amount of waves in a given _________ – not ½ of a wave If you can only have specific # of waves, then based on the formula _______ you can only have specific f’s, which in turn translates into specific Energy levels.

Heisenberg Uncertainty Principle States that: “it is impossible to determine _______ the position and velocity of an electron or any other particle” This is because whatever you use to observe subatomic particles, will affect the particles themselves. i. e you can’t observe/measure without disturbing it.

Quantum Numbers __________(n) indicates the main energy level (or shell) occupied by the electron. Principle ___________ is also referred to as the Principle Energy Level (n). n = 1, 2, 3, 4… where n = 1 is the closest to the nucleus. (Diagram: H atom Circular orbits to linear 1, 2, 3, …)

The electron now has an _____ number of Energy levels (shells) to jump to depending on the Energy input.

Each Energy level (n) divides up further into sublevels/subshells. How many divisions depends on the original Energy level. The 1 st E level has 1 sublevel (s only) 2 nd 2 sublevels (s and p) 3 rd 3 sublevels (s, p, and d) 4 th 4 sublevels (s, p, d and f) : : i. e nth n sublevels.

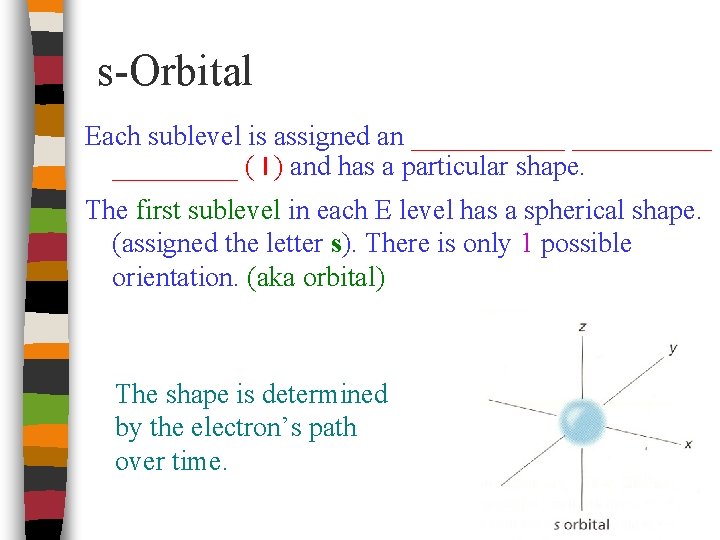
s-Orbital Each sublevel is assigned an _________ ( l ) and has a particular shape. The first sublevel in each E level has a spherical shape. (assigned the letter s). There is only 1 possible orientation. (aka orbital) The shape is determined by the electron’s path over time.

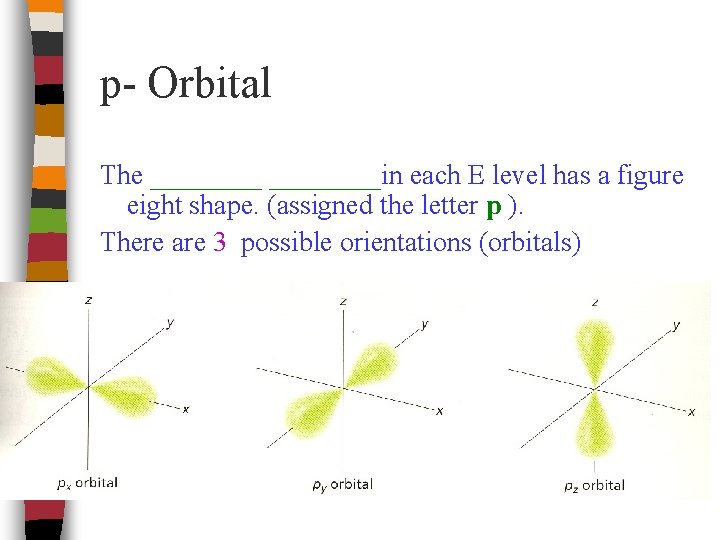
p- Orbital The ________in each E level has a figure eight shape. (assigned the letter p ). There are 3 possible orientations (orbitals)

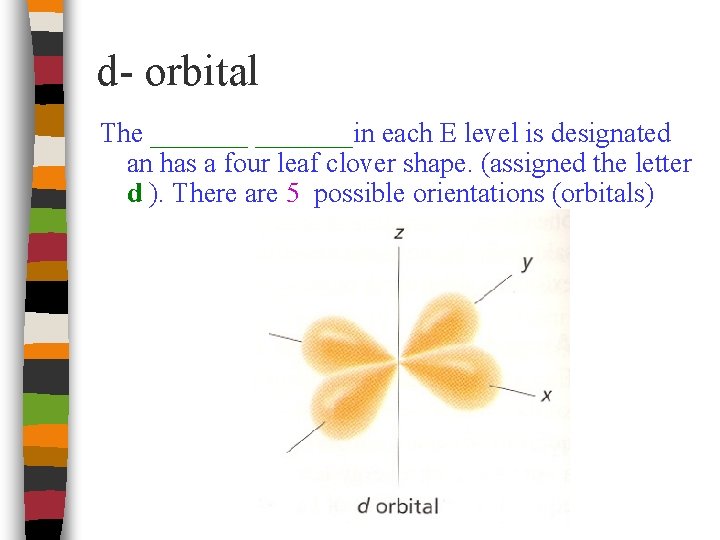
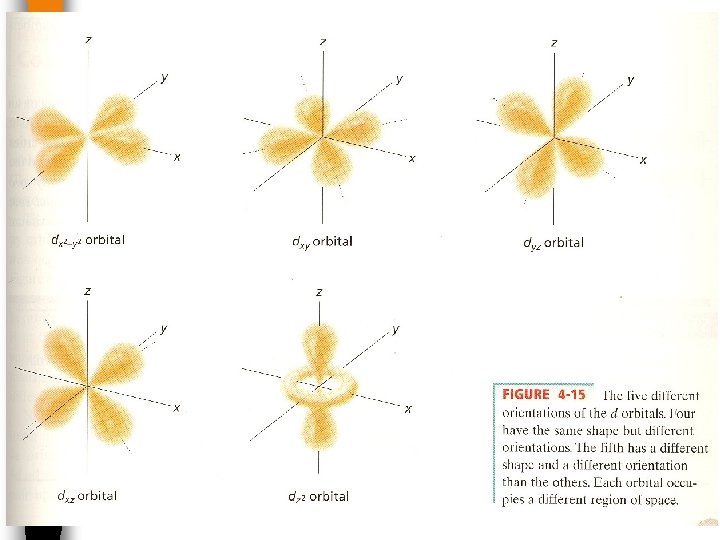
d- orbital The _______in each E level is designated an has a four leaf clover shape. (assigned the letter d ). There are 5 possible orientations (orbitals)


f, g and h are indescribable! The ________in each E level has an indescribable shape. (assigned the letter ___ ). There are 7 possible orientations (orbitals) The _________in each E level has an indescribable shape. (assigned the letter ___ ). There are 9 possible orientations (orbitals). The ________in each E level has an indescribable shape. (assigned the letter ____ ). There are 11 possible orientations (orbitals). Summary on board

To sum up: . . Principle E level # sublevels (n). 1 1 1 s 2 2 2 s 2 p 3 3 3 s 3 p 3 d 4 4 4 s 4 p 4 d 4 f 5 5 5 s 5 p 5 d 5 f 5 g 6 6 6 s 6 p 6 d 6 f 6 g 6 h We expect the order of the orbitals to increase as follows: 1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 5 g 6 s 6 p …

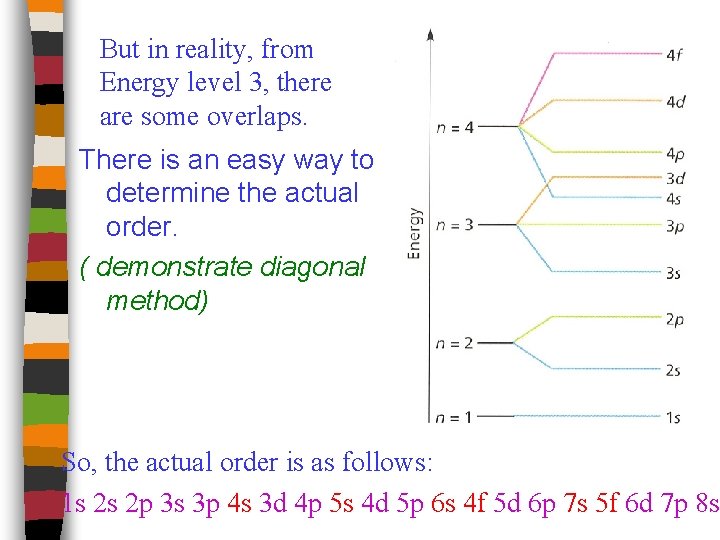
But in reality, from Energy level 3, there are some overlaps. There is an easy way to determine the actual order. ( demonstrate diagonal method) So, the actual order is as follows: 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s 4 d 5 p 6 s 4 f 5 d 6 p 7 s 5 f 6 d 7 p 8 s

Now each _______ (orientation) can hold 2 ____. The s sublevel which has 1 orbital can hold 2 e- ’s p 3 6 d 5 10 f 7 14 g 9 18 h 11 22.

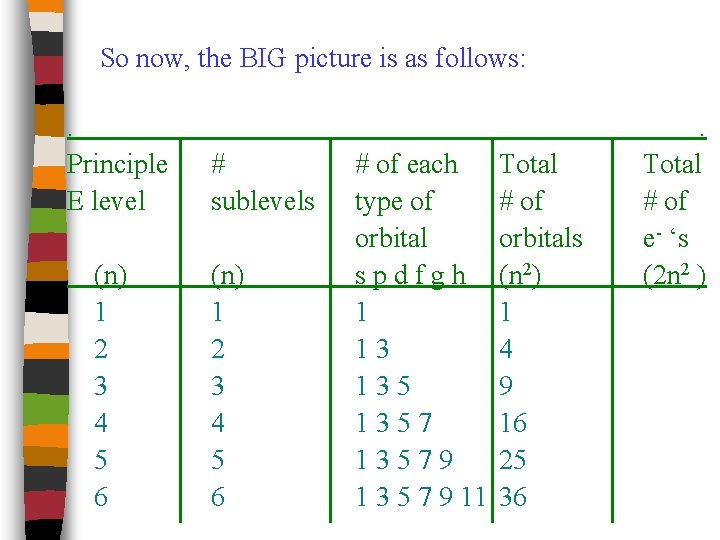
So now, the BIG picture is as follows: . Principle E level (n) 1 2 3 4 5 6 # sublevels (n) 1 2 3 4 5 6 # of each type of orbital spdfgh 1 13 13579 1 3 5 7 9 11 Total # of orbitals (n 2) 1 4 9 16 25 36 . Total # of e- ‘s (2 n 2 )

Electron Configuration Each orbital (orientation) can hold _____and can be represented by a box, circle or underline. (diagram) (complete orbital chart) For each electron, they orbit with a spin around in 2 different ways – ____________. One type of spin produces a magnetic field with a North up (_____), and the other produces a North down (_____) (diagram) The electrons are represented by _______ pointing up or down, depending on their spin. (diagram)

According to the: Pauli Exclusion principle, in order for 2 electrons to occupy the same orbital, they must have opposite spins. e. g. Hydrogen has 1 e- that occupies the lowest Energy level – 1 s. 1 s Helium has 2 e-’s that also occupies the lowest Energy level – 1 s. Opposing arrows that represent opposite spins. 1 s

Rules of Procedure for Electron Configuration: 1. ______: an electron occupies the lowest 2. 3. energy orbital that can receive it. (i. e. start from the bottom) __________: no 2 electrons in the same atom can have the same set of 4 quantum numbers. (i. e. no more than 2 electrons per orbital) ________: orbitals of equal energy are each occupied by one electron before any orbital is occupied by a 2 nd electron, and all electrons in singly occupied orbitals must have the same spin. (i. e. don’t double up until each orbital has an up arrow)

Now do Oxygen & explain:

____________: the way electrons are arranged amongst the orbitals. A special notation is used which consists of: the principle E level, the sublevel & the # of electrons in that sublevel. e. g. the 1 electron in Hydrogen occupies the 1 st orbital, in the 1 st sublevel, in the 1 st E level. (diagram) e. g. the 2 electrons in Helium also occupy the 1 st orbital, in the 1 st sublevel, in the 1 st E level. (diagram) e. g. for Lithium, which has 3 electrons, it’s as follows: (diagram)