Arrangement of Electrons in Atoms Chapter 4 Heisenberg

Arrangement of Electrons in Atoms Chapter 4

Heisenberg Uncertainty Principle It is impossible to know precisely both the velocity and the position of an –e at the same time.

Erwin Shrodinger - (1926) – derived an equation that described the location and energy of an –e in an atom. The Quantum Mechanical Model is the accepted model today.
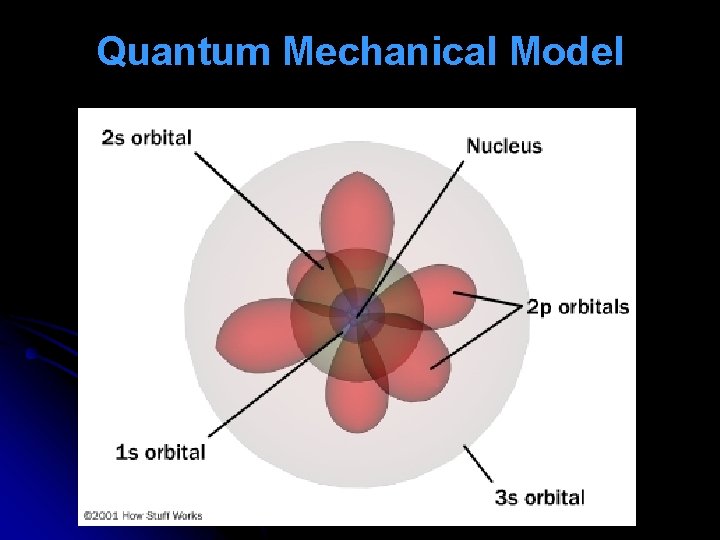
Quantum Mechanical Model

An atom has 7 Energy Levels or Quantums. Each energy level can be further arranged into sublevels.

Which can be arranged into: Orbitals – around the nucleus describing the probable location of electrons. Each orbital can hold up to 2 electrons

4 Types of Sublevels:
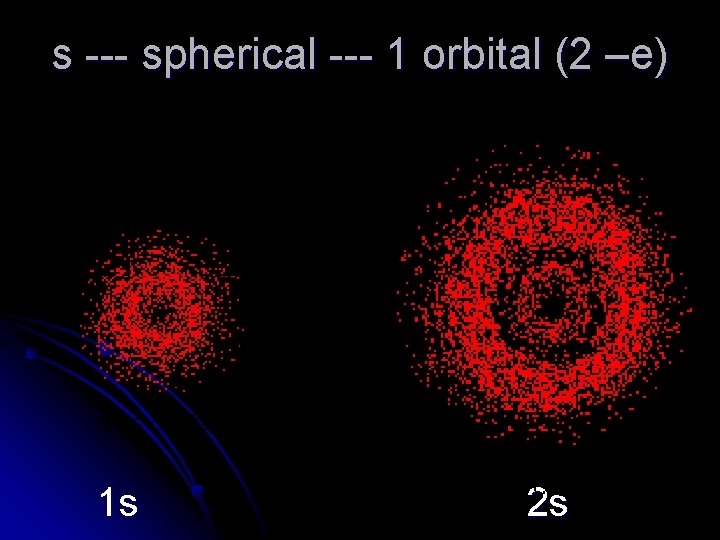
s --- spherical --- 1 orbital (2 –e) 1 s 2 s
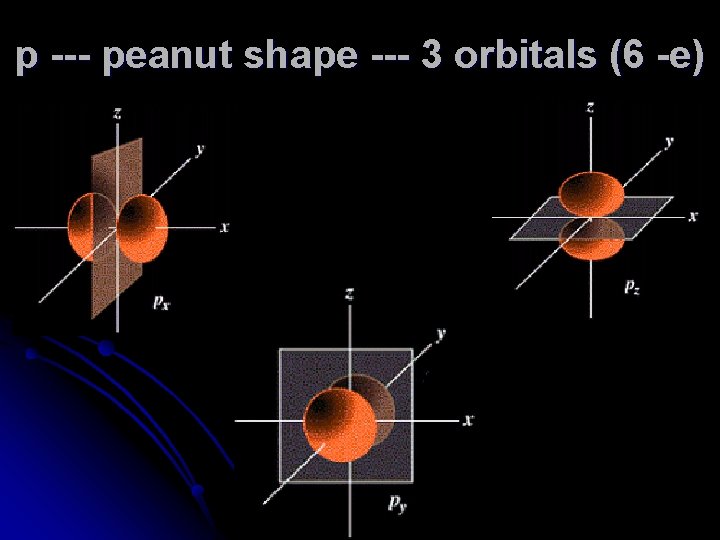
p --- peanut shape --- 3 orbitals (6 -e)
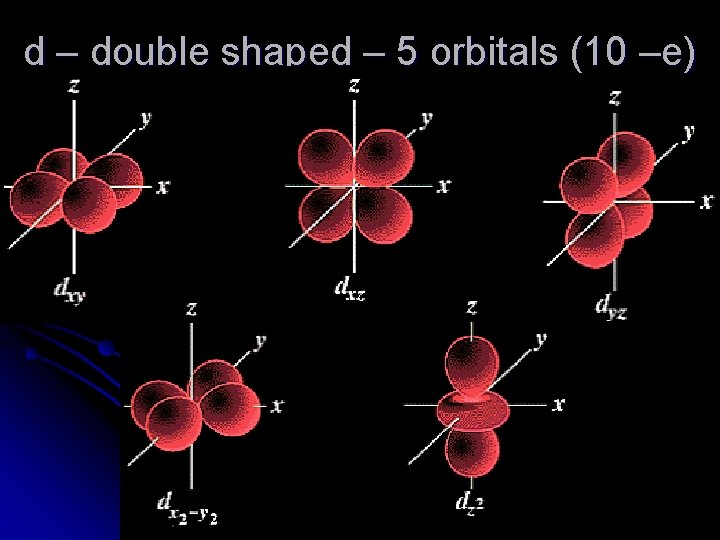
d – double shaped – 5 orbitals (10 –e)
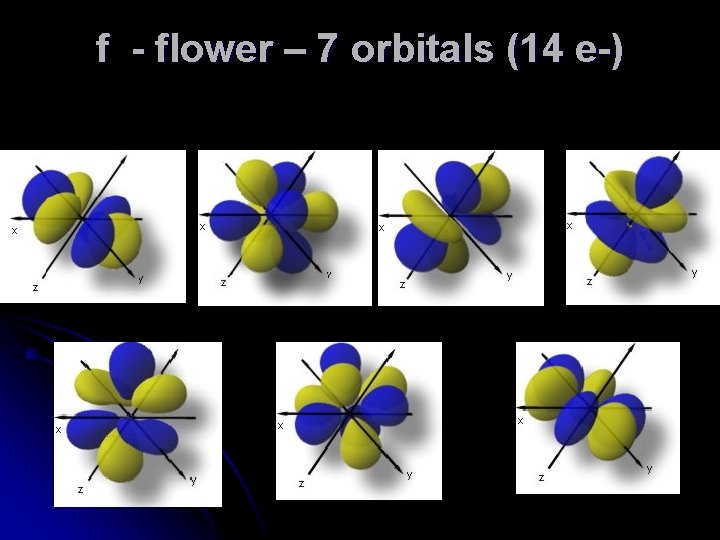
f - flower – 7 orbitals (14 e-)

Electron Configuration --- the arrangement of electrons in atoms.

Rules Governing Electron Configurations

Aufbau Principle An electron occupies the lowest energy orbital that it can enter. 1 s 2 s 3 s 4 s 5 s 6 s 7 s 2 p 3 p 4 p 5 p 6 p 7 p 3 d 4 d 5 d 6 d 7 d 4 f 5 f 6 f

Hund’s Rule Orbitals of the same energy (ex: 3 p) must each hold one electron before a second electron is assigned. Ex: 3 p

Pauli Exclusion Principle No two electrons in the same orbital can have the same spin. 3 p
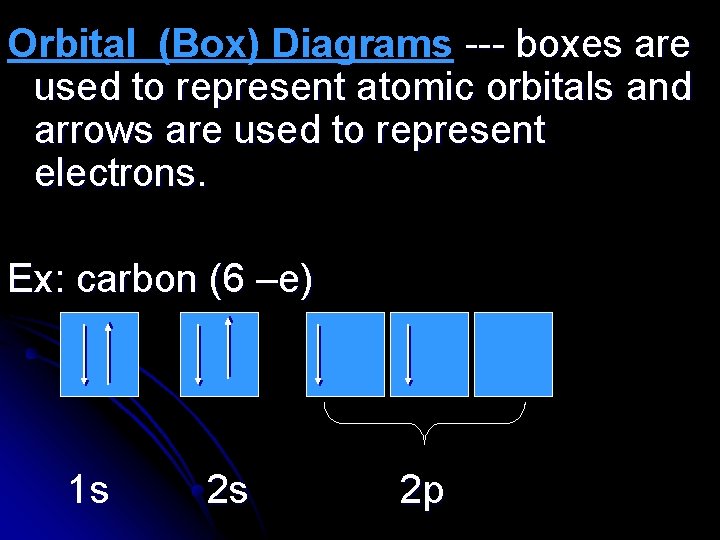
Orbital (Box) Diagrams --- boxes are used to represent atomic orbitals and arrows are used to represent electrons. Ex: carbon (6 –e) 1 s 2 s 2 p

Electron Configuration --- uses superscripts to specify how many electrons are found in each sublevel. Ex: chlorine (17 –e) 2 2 6 2 5 1 s 2 s 2 p 3 s 3 p

Noble Gas Configuration --- uses the noble gas for the previous period (in brackets) followed by the electron configuration for the energy level being filled. Ex: bromine [Ar] 4 s 2 3 d 10 4 p 5
- Slides: 19