Chapter 4 Arrangement of Electrons in Atoms Light






























- Slides: 33

Chapter 4 Arrangement of Electrons in Atoms

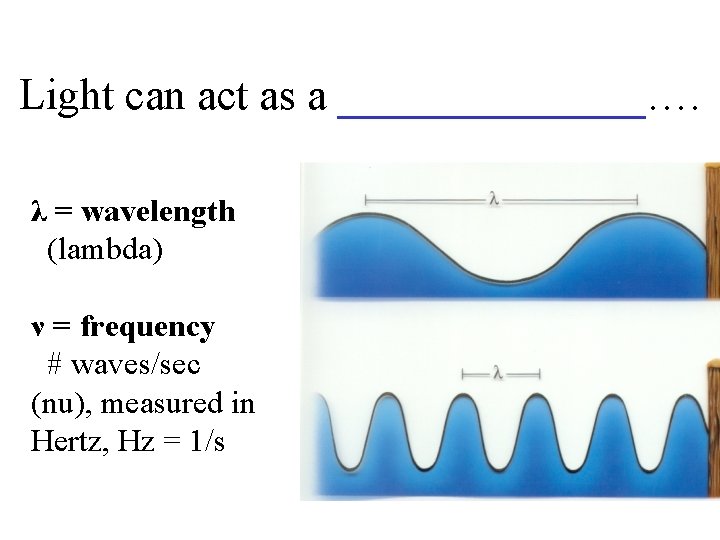
Light can act as a _______…. λ = wavelength (lambda) ν = frequency # waves/sec (nu), measured in Hertz, Hz = 1/s

c=λν • c = speed of light (3. 0 x 108 m/s) • What is the frequency of a beam of red light whose wavelength = 659 nm?

What is the frequency of a beam of red light whose wavelength = 659 nm? • • c=λν ______ m/s =_______ = _______ ν = ______1/s =______

_______ is the only electromagnetic (EM) radiation we can see • electromagnetic radiation (EM): form of energy that travels through space as a _____

Light can also act as a ______ • Supported by 2 experiments: – Photoelectric effect – Hydrogen atom spectrum

Particle description of light • Max Planck, 1900 – studied ______ of light by hot objects • Proposed that energy is not ______ continuously but in small little ______ called _______ – This is a particle property • “quantum”: min am’t of energy than can be lost or gained by an atom • Energy ________

Planck proposed the following relationship between a quantum of energy and its frequency of radiation: • Energy frequency • E= ( frequency ) • where h = Planck’s constant 6. 626 x 10 -34 J • s (or J/Hz)

Duality of light explained photoelectric effect! Metal must be struck by a photon possessing a min. am’t of E – below this am’t, the electron won’t leave the metal! More light of the same E (same ν) just released more electrons…not more energetic

• Ground state: atoms whose electrons are in their ______ energy level • Excited state: an atom, having _____ E, jumps its electron(s) to a higher E level. The electron must jump completely from one level to another.

• De-excitation: after a short time the electron ______________ and _____ a photon (packet of E) equivalent to the energy difference between the 2 steps. The photon will produce a spectral line with a discrete wavelength (color) associated with it. • Continuous spectrum: all wavelengths are present (i. e. sunlight) • Emission (bright line) spectrum: limited # of specific bright lines that are produced by pass the light emitted by an excited atom through a prism

• Absorption (dark line) spectrum: light emitted by an excited atom passes through a substance that filters out certain wavelengths and thus produces a spectrum with missing (dark) lines. • Bright lines of an emission spectrum are ___________ as the dark lines of an absorption spectrum for a given element.

Bohr Model of the Atom, 1913 • Studied the absorption of light by ______ • Absorption of light at definite wavelengths corresponds to the definite changes in the E of the ________ • Electrons can circle the nucleus at _____________ distances…only in allowed paths, or orbits (Satellite model) • Energy of an electron is ______ when it is in orbits farther away from the nucleus • His calculated energy values agreed with the observed spectral lines for hydrogen • Model did NOT work when applied to multi-electron elements

Duality of light • Proposed by Einstein after studying the works of Planck and Bohr • Light has been traditionally thought of as a _____ (it has freq. & wavelength) • But now. . light can also be thought of as a ______ of particles called photons • photons: particle of EM radiation having _______ mass and carrying a _______ of energy • Explained photoelectric effect: EM radiation is absorbed only in whole numbers of photons

4 -2 The Quantum Model of the Atom

De Broglie, 1924 • If light could behave as both a wave and a particle, then could an electron (a particle) also behave as both a particle and a wave ? ? ? ? • He said “_____” because…. – Since electrons could only exist at specific energies, and E can be equated to frequency (E = hν), they have wave properties – And electrons can be “focused” like light (electron microscopes) – particle nature of electrons had already been confirmed (cathode rays, oil drop exper, etc. )

• Electrons have a dual wave-particle nature

Heisenberg Uncertainty Principle • It is impossible to determine simultaneously both the _____ and _____ of an electron or any other particle

Schrodinger Wave Equation, 1926 • If electrons have wave properties, then wave equations can be applied to electrons (and other small particles) • Laid foundation for ___________: describes ___________ the wave properties of electrons and other very small particles • Gives the ___________ of finding an electron at a given place in time around the nucleus

Quantum Theory (con’t) • Electrons do NOT travel around the nucleus like planets around the sun • Electrons exist in certain regions called _______: a 3 -D region around the nucleus that indicates the _____ location of an electron

Quantum Numbers • Specify the properties of atomic orbitals and the properties of electrons in orbitals • There are _____ quantum numbers…. the first three of which come from solutions to Schrodinger’s wave equation

Principal Quantum Number, n • Indicates the __________ (or shell) occupied by the electron • n = 1, 2, 3 etc. • The ↑n value, the ______ the electron is from nucleus∴ principal quant # also gives ____________ • Total # electrons on a level = _____ • Total # orbitals on a level = n 2 since an orbital can hold two electrons (2 n 2/2= n 2)

Angular Momentum Quantum Number, l • Also known as “second quantum number” • Also known as “azimuthal quantum number” • Most energy levels (all but n=1) have orbitals of different _____, called ______ • Describes the _______ of the orbital • # orbital shapes possible is equal to the _____________ – If n=3, then there can be 3 orbital shapes • The values of l = 0, 1, 2, ……. (n-1) – If n=3, then l can be ___, or ___.

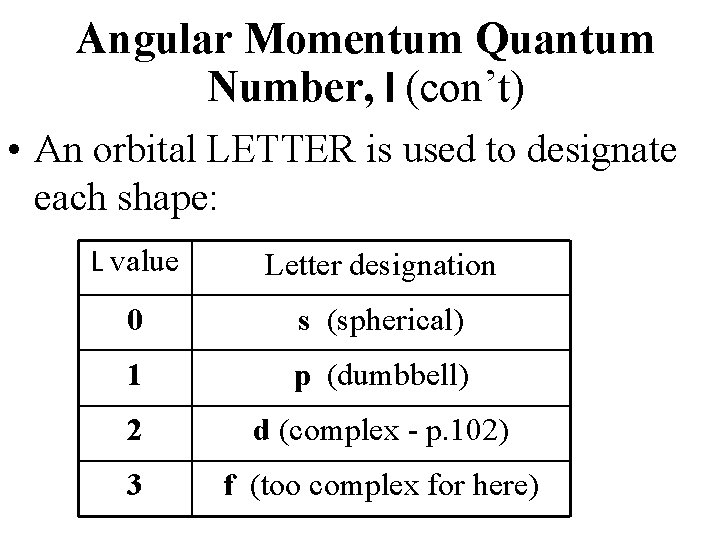
Angular Momentum Quantum Number, l (con’t) • An orbital LETTER is used to designate each shape: L value Letter designation 0 s (spherical) 1 p (dumbbell) 2 d (complex - p. 102) 3 f (too complex for here)

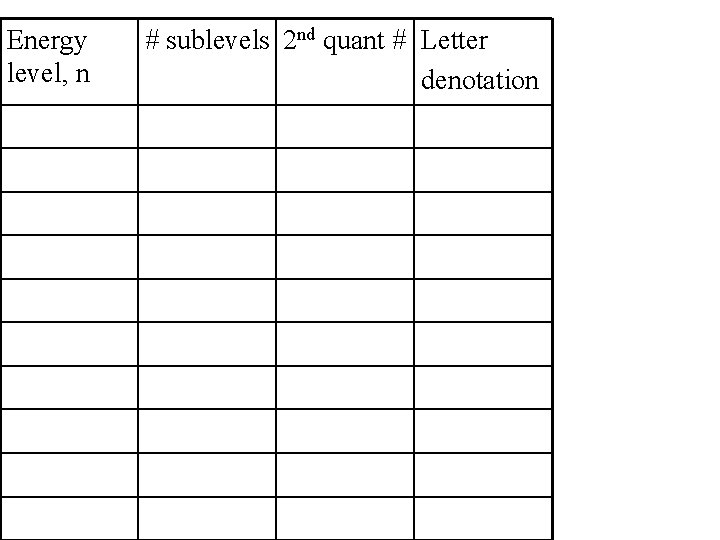
Energy level, n # sublevels 2 nd quant # Letter denotation

Magnetic Quantum Number, ml • also known as the 3 rd quantum number • indicates the orientation of an orbital around the nucleus • = (2 l + 1)

Magnetic Quantum Number, m (con’t) • Sum of all orbitals in a sublevel (or E level) is a spherical cloud! Orbital shape # orientations s 1 p 3 d 5 f 7

Spin quantum number, s • Describes the spin of the electron • Could be either clockwise ( + ½) or counterclockwise (- ½ ) • Each orbital can hold a maximum of two electrons, a pair, spinning in opposite directions

4 -3 ELECTRON CONFIGURATIONS

Hund’s Rule • Orbitals of equal energy are ____ occupied by one electron before any orbital is occupied by a second electron, and… • All electrons in singly occupied orbitals must have the same _____

Aufbau Principle • An electron occupies the ________ orbital that can receive it

Noble Gas Notation • What is the noble gas electron notation for calcium? • 1 s 22 p 63 s 23 p 64 s 2 • What is the noble gas config for S? • 1 s 22 p 63 s 23 p 4

PREDICTED