3 9 Electron Configurations The electron configuration describes

3. 9 Electron Configurations The electron configuration describes how the electrons are distributed in the various atomic orbitals. In a ground state hydrogen atom, the electron is found in the 1 s orbital. Ground state electron configuration of hydrogen Energy principal (n = 1) 2 s 1 s 2 p 2 p 2 p 1 1 s number of electrons in the orbital or subshell angular momentum (l = 0) The use of an up arrow indicates an electron with ms = + ½

Electron Configurations If hydrogen’s electron is found in a higher energy orbital, the atom is in an excited state. A possible excited state electron configuration of hydrogen 2 s Energy 1 2 s 1 s 2 p 2 p 2 p

Electron Configurations The helium emission spectrum is more complex than the hydrogen spectrum. There are more possible energy transitions in a helium atom because helium has two electrons.
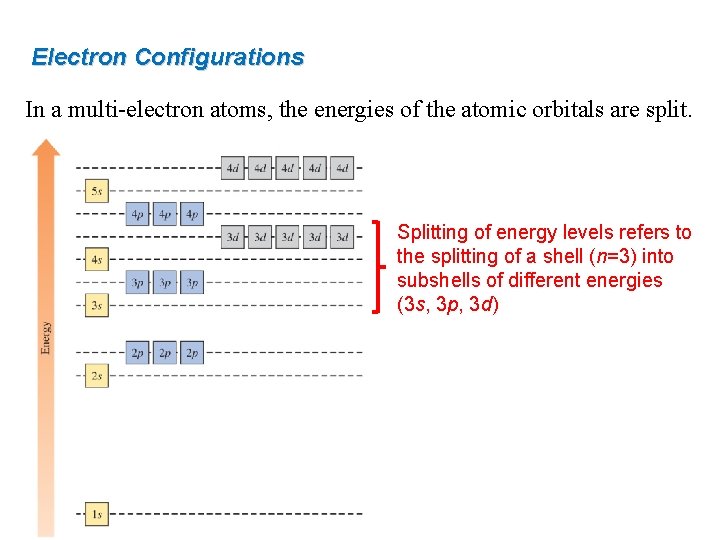
Electron Configurations In a multi-electron atoms, the energies of the atomic orbitals are split. Splitting of energy levels refers to the splitting of a shell (n=3) into subshells of different energies (3 s, 3 p, 3 d)

Electron Configurations According to the Pauli exclusion principle, no two electrons in an atom can have the same four quantum numbers. The ground state electron configuration of helium Energy 2 p 2 p 2 p 1 s 2 2 s Quantum number Principal (n) 1 s describes the 1 s orbital Angular moment (l) Magnetic (ml) describes the electrons in the 1 s orbital Electron spin (ms) 1 0 0 +½ 1 0 0 ‒½

Electron Configurations The Aufbau principle states that electrons are added to the lowest energy orbitals first before moving to higher energy orbitals. Li has a total of 3 electrons Energy The ground state electron configuration of Li 2 p 2 s 1 s 2 p 2 p 1 s 22 s 1 The third electron must go in the next available orbital with the lowest possible energy. The 1 s orbital can only accommodate 2 electrons (Pauli exclusion principle)

Electron Configurations The Aufbau principle states that electrons are added to the lowest energy orbitals first before moving to higher energy orbitals. Be has a total of 4 electrons Energy The ground state electron configuration of Be 2 p 2 s 1 s 2 p 2 p 1 s 22 s 2

Electron Configurations The Aufbau principle states that electrons are added to the lowest energy orbitals first before moving to higher energy orbitals. B has a total of 5 electrons Energy The ground state electron configuration of B 2 p 2 s 1 s 2 p 2 p 1 s 2 s 2 p 2 2 1

Electron Configurations According to Hund’s rule, the most stable arrangement of electrons is the one in which the number of electrons with the same spin is maximized. C has a total of 6 electrons The ground state electron configuration of C Energy 1 s 22 p 2 p 2 p 2 s The 2 p orbitals are of equal energy, or degenerate. 1 s Put 1 electron in each before pairing (Hund’s rule).

Electron Configurations According to Hund’s rule, the most stable arrangement of electrons is the one in which the number of electrons with the same spin is maximized. N has a total of 7 electrons The ground state electron configuration of N Energy 1 s 22 p 3 2 p 2 p 2 p 2 s The 2 p orbitals are of equal energy, or degenerate. 1 s Put 1 electron in each before pairing (Hund’s rule).

Electron Configurations According to Hund’s rule, the most stable arrangement of electrons is the one in which the number of electrons with the same spin is maximized. O has a total of 8 electrons The ground state electron configuration of O Energy 1 s 22 p 4 2 p 2 s 1 s 2 p 2 p Once all the 2 p orbitals are singly occupied, additional electrons will have to pair with those already in the orbitals.

Electron Configurations According to Hund’s rule, the most stable arrangement of electrons is the one in which the number of electrons with the same spin is maximized. F has a total of 9 electrons The ground state electron configuration of F Energy 1 s 22 p 5 2 p 2 s 1 s 2 p 2 p When there are one or more unpaired electrons, as in the case of oxygen and fluorine, the atom is called paramagnetic.

Electron Configurations According to Hund’s rule, the most stable arrangement of electrons is the one in which the number of electrons with the same spin is maximized. Ne has a total of 10 electrons The ground state electron configuration of Ne Energy 1 s 22 p 6 2 p 2 s 1 s 2 p 2 p When all of the electrons in an atom are paired, as in neon, it is called diamagnetic.

Electron Configurations General rules for writing electron configurations: 1) Electrons will reside in the available orbitals of the lowest possible energy. 2) Each orbital can accommodate a maximum of two electrons. 3) Electrons will not pair in degenerate orbitals if an empty orbital is available. 4) Orbitals will fill in the order indicated in the figure.

Worked Example 3. 10 Write the electron configuration and give the orbital diagram of a calcium (Ca) atom (Z = 20). Setup Because Z = 20, Ca has 20 electrons. They will fill in according to the diagram at right. Each s subshell can contain a maximum of two electrons, whereas each p subshell can contain a maximum of six electrons.

3. 10 Electron Configurations and the Periodic Table The electron configurations of all elements except hydrogen and helium can be represented using a noble gas core. The electron configuration of potassium (Z = 19) is 1 s 22 p 63 s 23 p 64 s 1. Because 1 s 22 p 63 s 23 p 6 is the electron configuration of argon, we can simplify potassium’s to [Ar]4 s 1. The ground state electron configuration of K: 1 s 22 p 63 s 23 p 64 s 1 [Ar]4 s 1
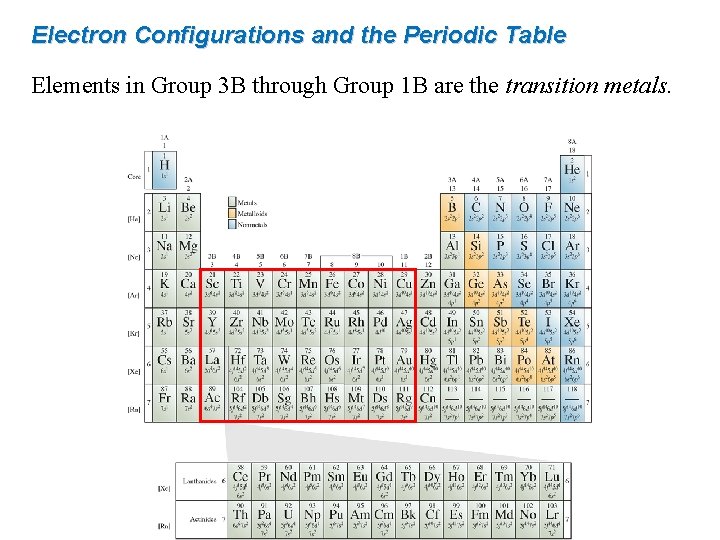
Electron Configurations and the Periodic Table Elements in Group 3 B through Group 1 B are the transition metals.

Electron Configurations and the Periodic Table Following lanthanum (La), there is a gap where the lanthanide (rare earth) series belongs.

Electron Configurations and the Periodic Table After actinum (Ac) comes the actinide series.
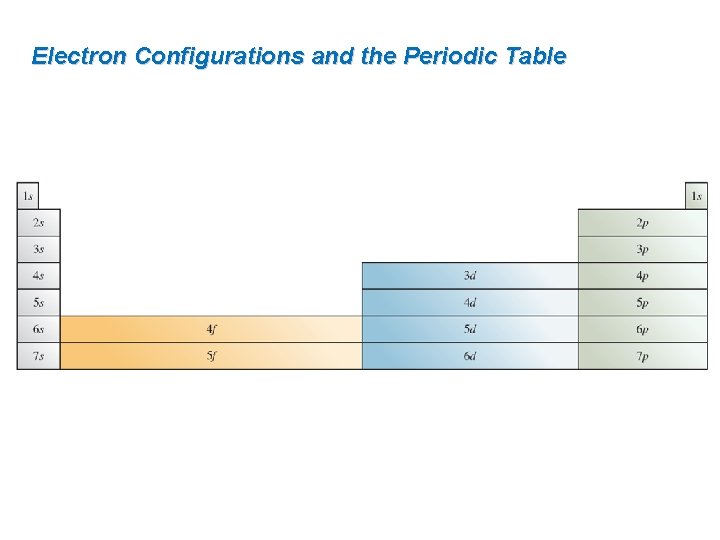
Electron Configurations and the Periodic Table

Electron Configurations and the Periodic Table There are several notable exceptions to the order of electron filling for some of the transition metals. Ø Chromium (Z = 24) is [Ar]4 s 13 d 5 and not [Ar]4 s 23 d 4 as expected. Ø Copper (Z = 29) is [Ar]4 s 13 d 10 and not [Ar]4 s 23 d 9 as expected. The reason for these anomalies is the slightly greater stability of d subshells that are either half-filled (d 5) or completely filled (d 10). Cr [Ar] 4 s 3 d 3 d 3 d Greater stability with half-filled 3 d subshell

Electron Configurations and the Periodic Table There are several notable exceptions to the order of electron filling for some of the transition metals. Ø Chromium (Z = 24) is [Ar]4 s 13 d 5 and not [Ar]4 s 23 d 4 as expected. Ø Copper (Z = 29) is [Ar]4 s 13 d 10 and not [Ar]4 s 23 d 9 as expected. The reason for these anomalies is the slightly greater stability of d subshells that are either half-filled (d 5) or completely filled (d 10). Cu [Ar] 4 s 3 d 3 d 3 d Greater stability with filled 3 d subshell

Worked Example 3. 11 Write the electron configuration for an arsenic atom (Z = 33) in the ground state. Setup The noble gas core for As is [Ar], where Z = 18 for Ar. The order of filling beyond the noble gas core is 4 s, 3 d, and 4 p. Fifteen electrons go into these subshells because there are 33 – 18 = 15 electrons in As beyond its noble gas core. Text Practice 3. 93 3. 98 3. 104 a, b, c, d 3. 106 3. 113

Study Guide for sections 3. 9 -3. 10 DAY 7, Terms to know: Sections 3. 9 -3. 10 electron configuration, Aufbau principle, Hund’s rule, orbital diagram DAY 7, Specific outcomes and skills that may be tested on exam 1: Sections 3. 9 -3. 10 • Be able to use the Pauli exclusion principle and Aufbau principle to give a complete or abbreviated electron configuration for an atom in either its ground state or one possible excited state • Given an electron configuration, be able to give a complete elemental symbol for an atom • Be able to use the Pauli exclusion principle, Aufbau principle, and Hund’s rule to give a complete or abbreviated orbital diagram for an atom either its ground state or one possible excited state • Given an orbital diagram, be able to give a complete elemental symbol for an atom or ion • Be able to recognize and explain how Cr and Cu are exceptions to the Aufbau principle

Extra Practice Problems for sections 3. 9 -3. 10 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 3. 95 3. 97 3. 99 3. 101 3. 103 3. 105 3. 117 3. 119 3. 137 3. 139

On day 8, we will have exam 1 Prep for day 9 Must watch videos: http: //www. learnerstv. com/video/Free-video-Lecture-3354 -Chemistry. htm (ionization energy) http: //www. learnerstv. com/video/Free-video-Lecture-3355 -Chemistry. htm (periodic trends) Other helpful videos: http: //ocw. mit. edu/courses/chemistry/5 -111 -principles-of-chemical-science-fall-2008/videolectures/lecture-9/ (MIT) http: //ps. uci. edu/content/chem-1 a-general-chemistry (UC-Irvine lectures 7) Read sections 4. 1 -4. 6
- Slides: 26