Properties of Electrons Electrons display both particle properties

Properties of Electrons • Electrons display both particle properties and wave properties. • Electrons were discovered by JJ Thompson • Thompson also measured the charge/mass ratio • Milikan was able to determine the charge on an electron • Davisson and Germer discovered the wave nature of an electron at Bell Labs in 1927

The Atom Model Different models of atoms • Thompson developed the plum pudding model of an atom 1898 • Rutherford suggested the planetary model (i. e. electrons orbit the nucleus) • Bohr applied concepts of quantization to Rutherford’s model to develop the Bohr model • Bohr model lasted 10 years, and was replaced by a wave model, called the quantum mechanical model, based on the wave nature of electrons.
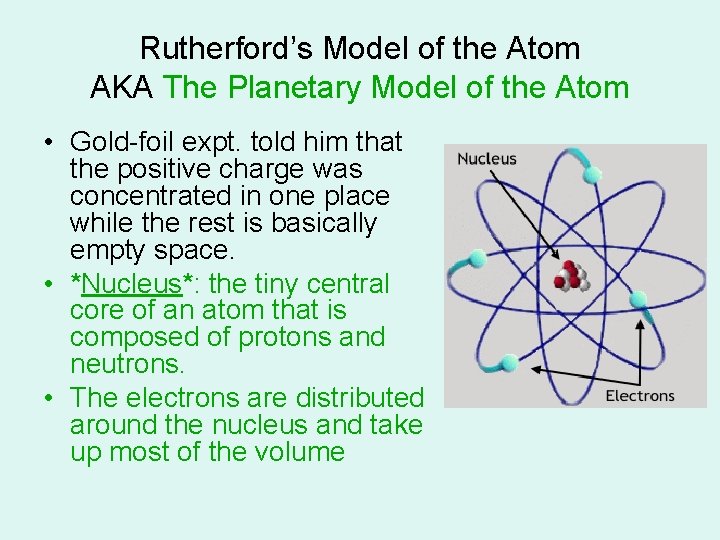
Rutherford’s Model of the Atom AKA The Planetary Model of the Atom • Gold-foil expt. told him that the positive charge was concentrated in one place while the rest is basically empty space. • *Nucleus*: the tiny central core of an atom that is composed of protons and neutrons. • The electrons are distributed around the nucleus and take up most of the volume

NCSCOS 2. 01 • Rutherford’s model could not explain the chemical properties of elements. – i. e. the characteristic color of metals given off when they are heated • Niels Bohr (1885 -1962) – A student of Rutherford took into account new data and improved upon his teacher’s model.
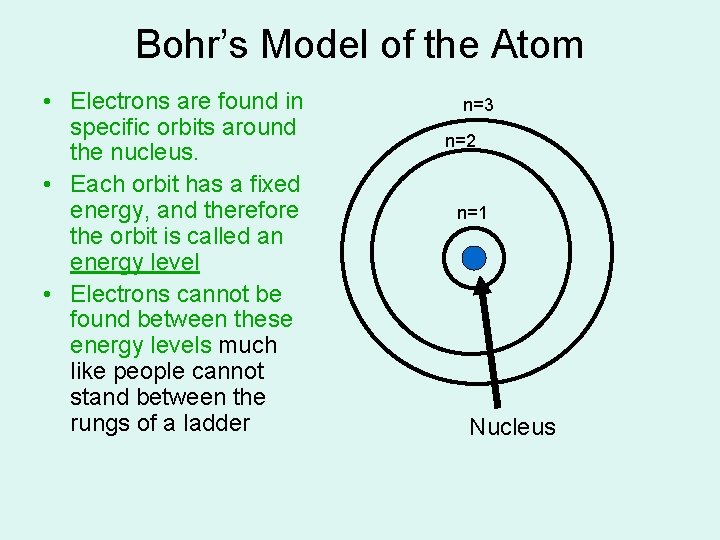
Bohr’s Model of the Atom • Electrons are found in specific orbits around the nucleus. • Each orbit has a fixed energy, and therefore the orbit is called an energy level • Electrons cannot be found between these energy levels much like people cannot stand between the rungs of a ladder n=3 n=2 n=1 Nucleus

Electronic States • The lowest energy level available to an electron in an atom is its ground state. • An excited state of an electron in an atom is any energy state above the ground state.
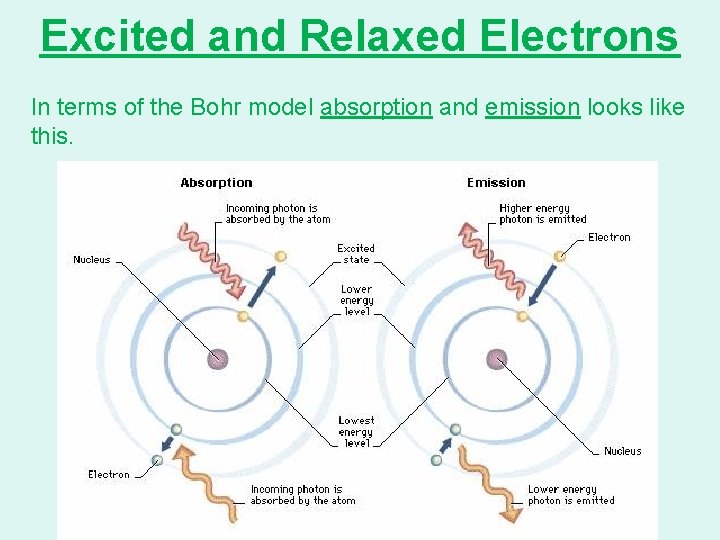
Excited and Relaxed Electrons In terms of the Bohr model absorption and emission looks like this.
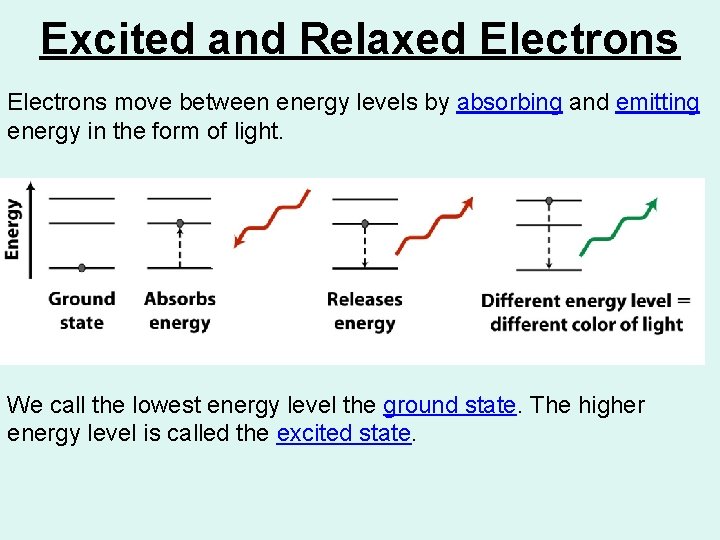
Excited and Relaxed Electrons move between energy levels by absorbing and emitting energy in the form of light. We call the lowest energy level the ground state. The higher energy level is called the excited state.

• The amount of energy that is required to do this is called a quantum of energy • A quantum of energy can be released as light, called a photon, when the electron falls from an excited state to it’s ground state. • Not all quantums are the same as the energy levels are not all evenly spaced – They are closer together at the higher levels than at the lower levels

Practice Problems 1. What wavelength of energy is given off when an electron moves from the n=3 level to n=1? Answer: 103 nm 2. n=6 to n=2 Answer: 410 nm 3. n=2 to n=1 Answer: 122 nm 4. n=5 to n=3 Answer: 1282 nm 5. n=4 to n=2 Answer: 486 nm
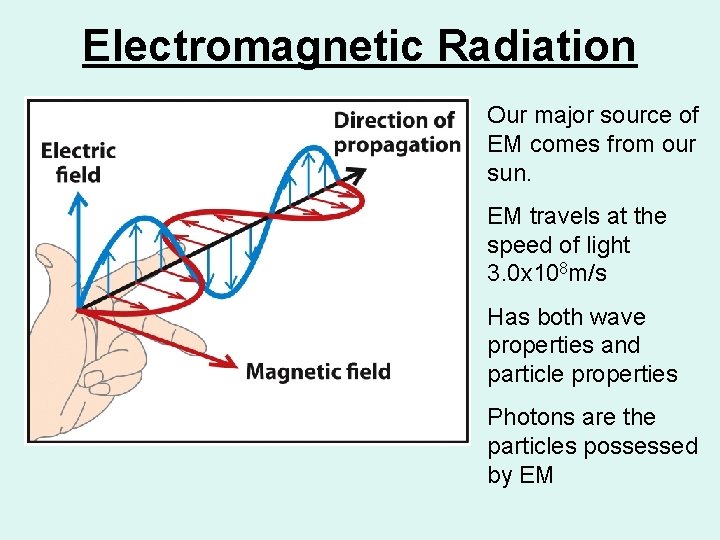
Electromagnetic Radiation Our major source of EM comes from our sun. EM travels at the speed of light 3. 0 x 108 m/s Has both wave properties and particle properties Photons are the particles possessed by EM
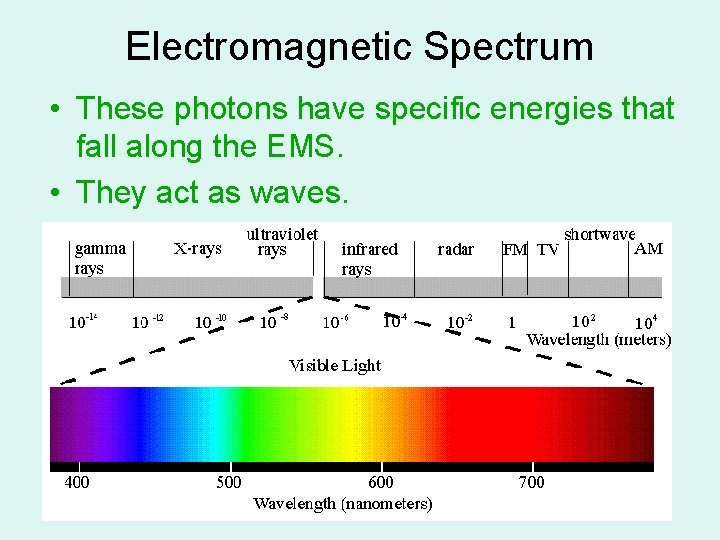
Electromagnetic Spectrum • These photons have specific energies that fall along the EMS. • They act as waves.

Parts of Waves • Wavelength (λ): Distance b/w two equivalent points on a wave. – Unit: m, nm • Frequency (ν): Number of waves that pass a given point per second. – Unit: Hz, s-1

Relationship b/w Frequency and Wavelength • Inversely Proportional – i. e. long wavelength = low frequency and vice versa • c=λν – c: speed of light (3. 00 x 108 m/s) Example: What is the frequency of a wave with a wavelength of 1. 2 x 10 -10 m? c=λν 3. 00 x 108 m/s = (1. 2 x 10 -10 m) (ν) ν = 2. 5 x 1018 s-1

Calculating Photon Energies (Honors) • E=hν – E= energy of the photon – h= Planck’s Constant (6. 626 x 10 -34 J·s) – ν=Frequency Example: What is the energy of a photon whose frequency is 3. 0 X 1012 Hz? E=hν E=(6. 626 x 10 -34 J·s)(3. 0 x 1012 Hz) E= 2. 0 x 10 -21 J

Practice Problems 1. A red light has a wavelength of 728 nm. What is the frequency of the light? (hint 1 nm = 1 x 10 -9 m) Answer: 4. 12 x 1014 s-1 2. What is the wavelength of the radio station 96. 9 the Kat? (Hint: ν = 96. 9 MHz, 1 MHz = 1, 000 Hz) Answer: 3. 10 m 3. Honors: What is the energy of a quantum of light with a frequency of 7. 39 x 1014 Hz? Answer: 4. 90 x 10 -19 J 4. Honors: What is the wavelength of a photon with an energy of 8. 54 x 10 -19 J? Answer: 2. 33 x 10 -7 m

Behavior of Waves • Waves refract or bend when they pass from one medium to another with different densities. • Diffraction is the bending of electromagnetic radiation as it passes around the edge of an object or through narrow openings. • Interference is the interaction of waves that results in either reinforcing their amplitudes or canceling them out.
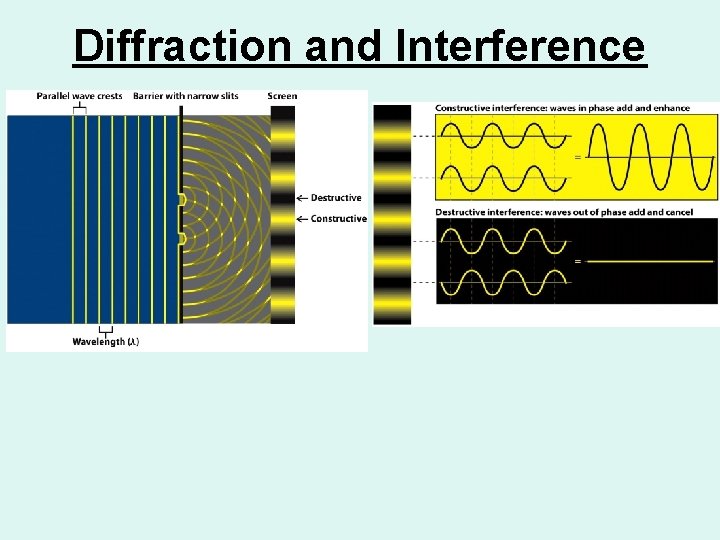
Diffraction and Interference
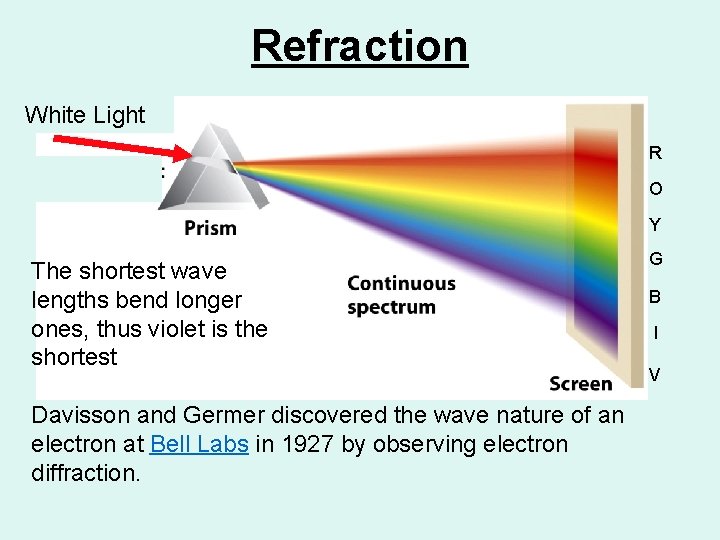
Refraction White Light R O Y The shortest wave lengths bend longer ones, thus violet is the shortest Davisson and Germer discovered the wave nature of an electron at Bell Labs in 1927 by observing electron diffraction. G B I V
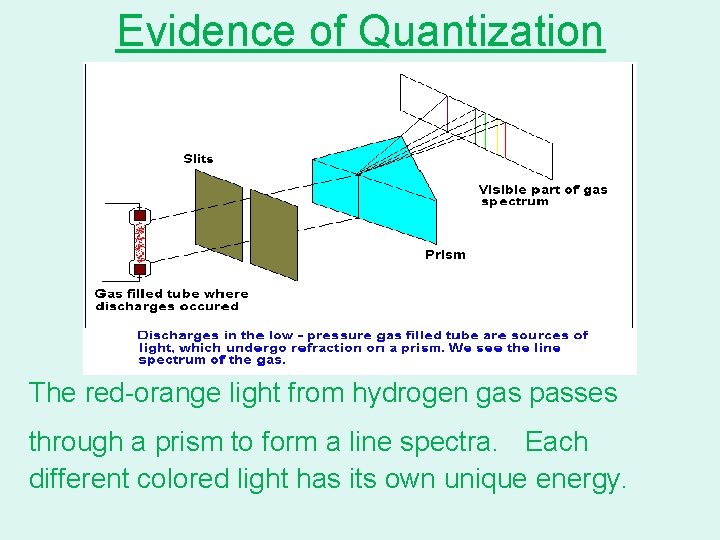
Evidence of Quantization The red-orange light from hydrogen gas passes through a prism to form a line spectra. Each different colored light has its own unique energy.
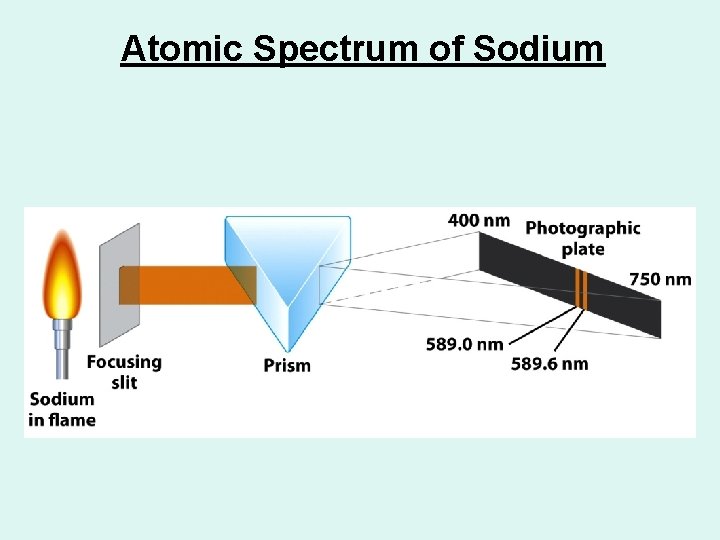
Atomic Spectrum of Sodium

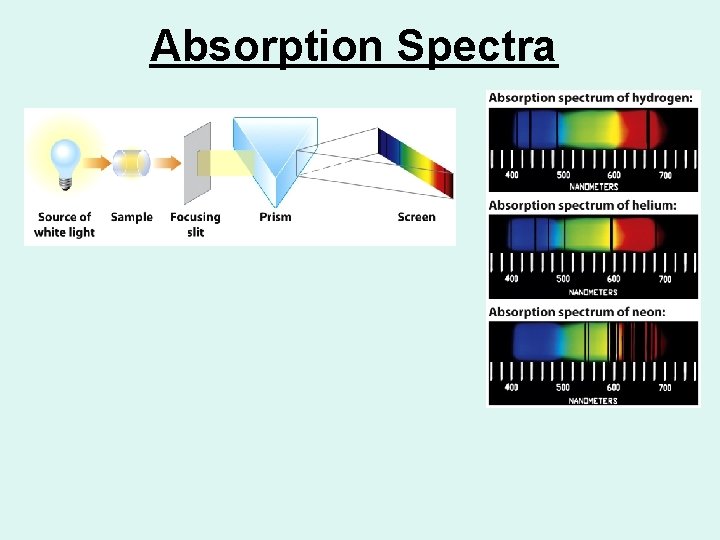
Absorption Spectra

Types of Spectra • Atomic emission spectra consist of bright lines on a dark background. • Atomic absorption spectra consist of characteristic series of dark lines produced when free gaseous atoms are illuminated by external sources of radiation.
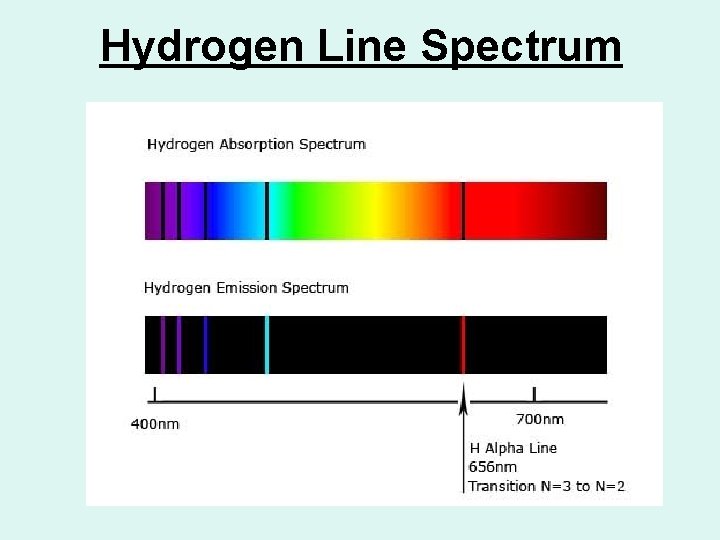
Hydrogen Line Spectrum

Problems with the Bohr Model • The Bohr model applies only to one electron atoms. • The Bohr model doesn’t account for the observed spectra of multielectron elements or ions. • The movement of electrons in atoms is much less clearly defined than Bohr allowed.
- Slides: 26