1152022 Chem160 Dr Seemal Jelani 1 Organic Chemistry
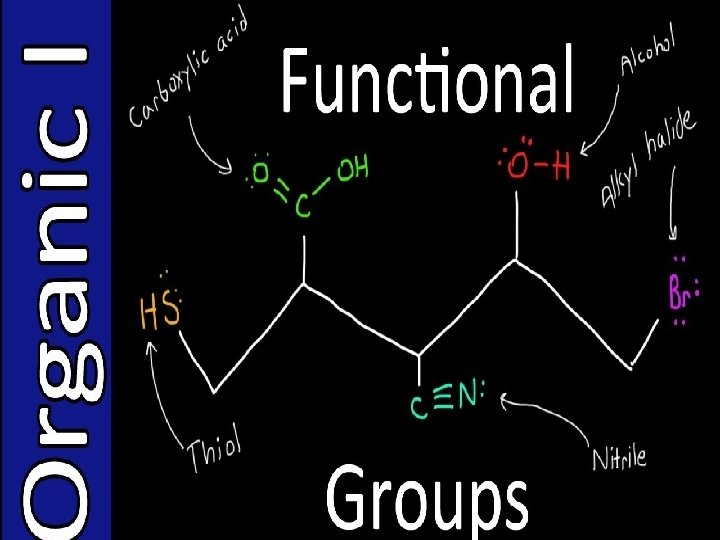
































- Slides: 62

1/15/2022 Chem-160 Dr Seemal Jelani 1

Organic Chemistry • • • Introduction Functional Groups Alkanes Alkenes Alkynes Alcohols • Acids, Esters and Amides 1/15/2022 Chem-160 Dr Seemal Jelani 2

Introduction • Most organic compounds have a “skeleton” that is composed of C-C bonds. – The C-C bonds may be single bonds, double bonds, or triple bonds. – Carbon forms a total of 4 bonds. • The “skeleton” of an organic compound has H’s attached to it. – other “heteroatoms” like O, halogens, N, S, and P may be present as well 1/15/2022 Chem-160 Dr Seemal Jelani 3

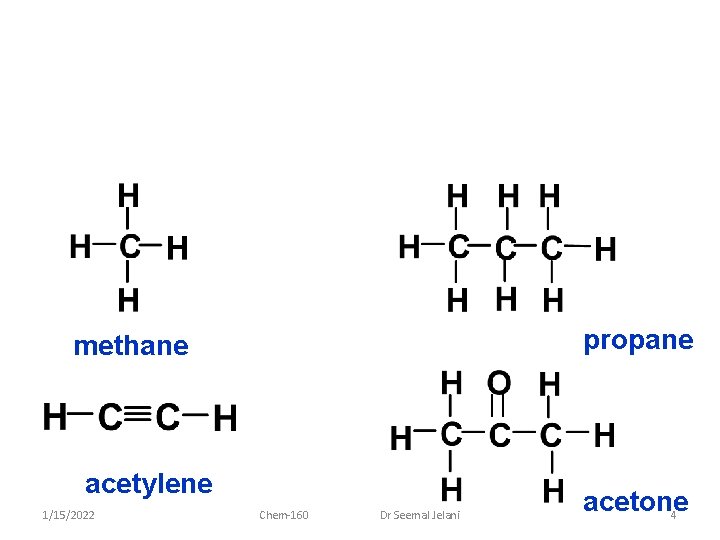
propane methane acetylene 1/15/2022 Chem-160 Dr Seemal Jelani acetone 4

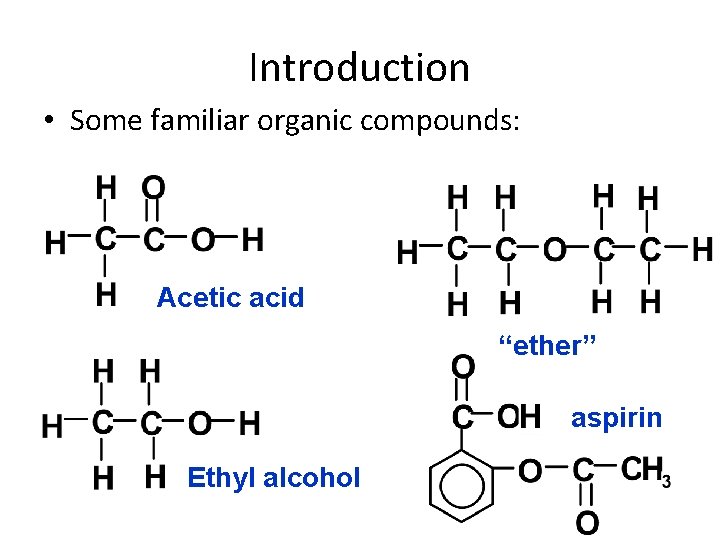
Introduction • Some familiar organic compounds: Acetic acid “ether” aspirin Ethyl alcohol 1/15/2022 Chem-160 Dr Seemal Jelani 5

Introduction • Organic compounds are commonly classified and named based on the type of functional group present. – An atom or group of atoms that influences the way the molecule functions, reacts or behaves. – The center of reactivity in an organic compound 1/15/2022 Chem-160 Dr Seemal Jelani 6

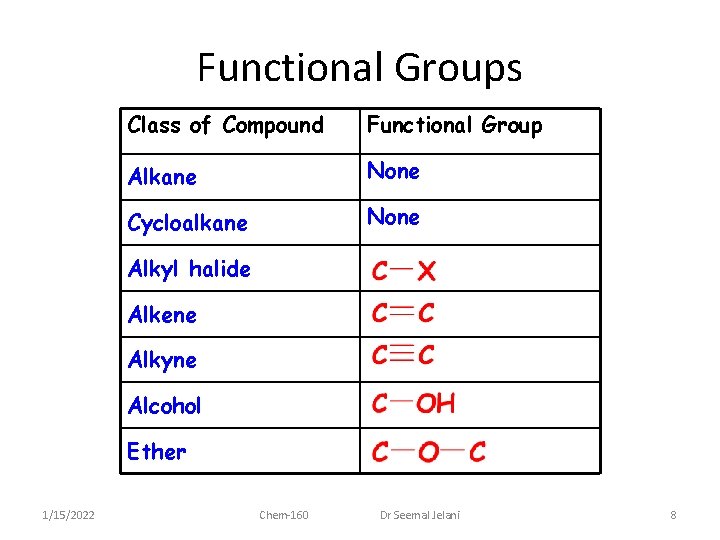
Functional Groups • On your exam, you will be responsible for recognizing and naming the various common functional groups that are found in organic compounds: – Use Table 25. 4 and the following slides to help you study 1/15/2022 Chem-160 Dr Seemal Jelani 7

Functional Groups Class of Compound Functional Group Alkane None Cycloalkane None Alkyl halide Alkene Alkyne Alcohol Ether 1/15/2022 Chem-160 Dr Seemal Jelani 8

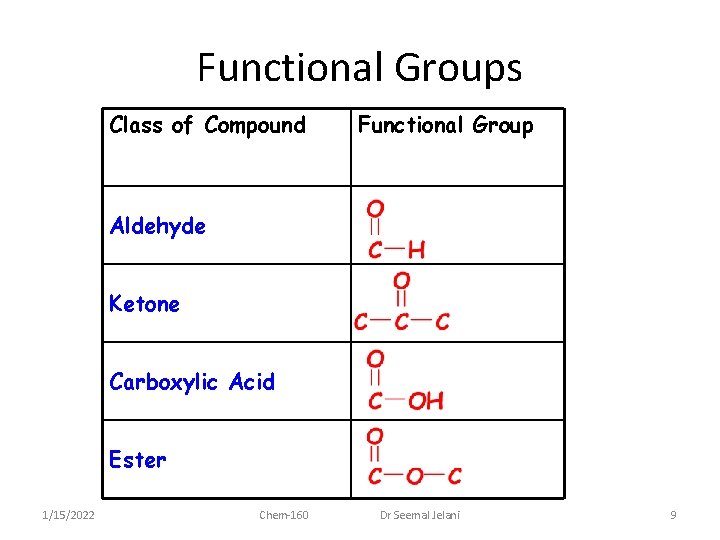
Functional Groups Class of Compound Functional Group Aldehyde Ketone Carboxylic Acid Ester 1/15/2022 Chem-160 Dr Seemal Jelani 9

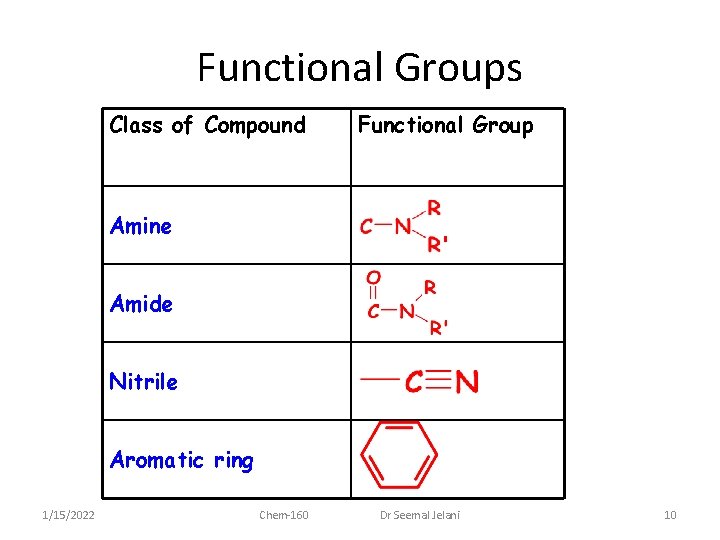
Functional Groups Class of Compound Functional Group Amine Amide Nitrile Aromatic ring 1/15/2022 Chem-160 Dr Seemal Jelani 10

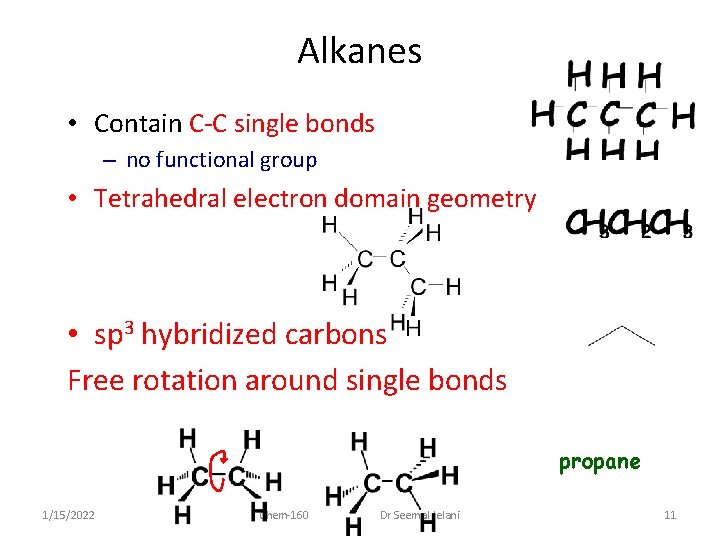
Alkanes • Contain C-C single bonds – no functional group • Tetrahedral electron domain geometry • sp 3 hybridized carbons Free rotation around single bonds propane 1/15/2022 Chem-160 Dr Seemal Jelani 11

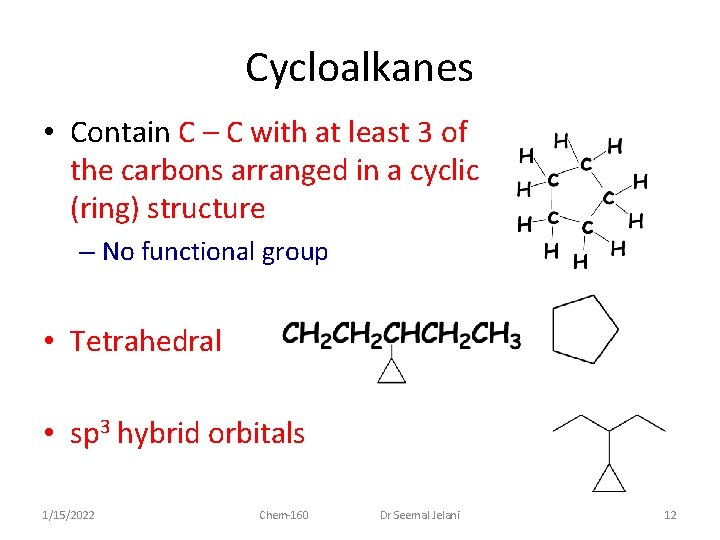
Cycloalkanes • Contain C – C with at least 3 of the carbons arranged in a cyclic (ring) structure – No functional group • Tetrahedral • sp 3 hybrid orbitals 1/15/2022 Chem-160 Dr Seemal Jelani 12

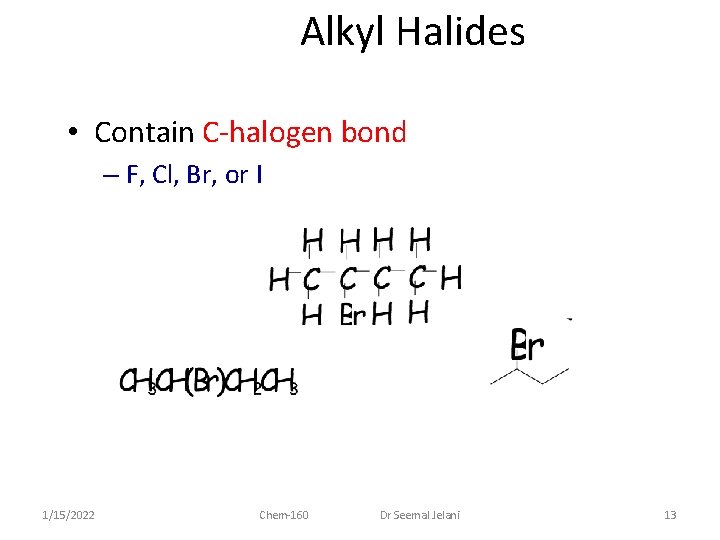
Alkyl Halides • Contain C-halogen bond – F, Cl, Br, or I 1/15/2022 Chem-160 Dr Seemal Jelani 13

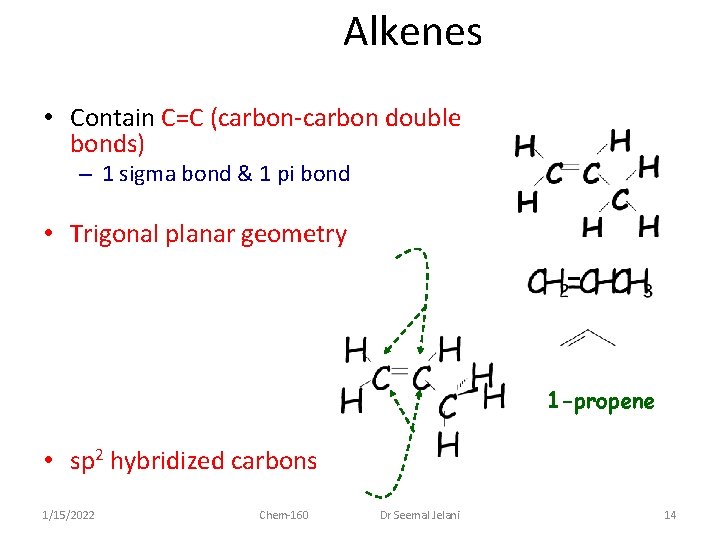
Alkenes • Contain C=C (carbon-carbon double bonds) – 1 sigma bond & 1 pi bond • Trigonal planar geometry 1 -propene • sp 2 hybridized carbons 1/15/2022 Chem-160 Dr Seemal Jelani 14

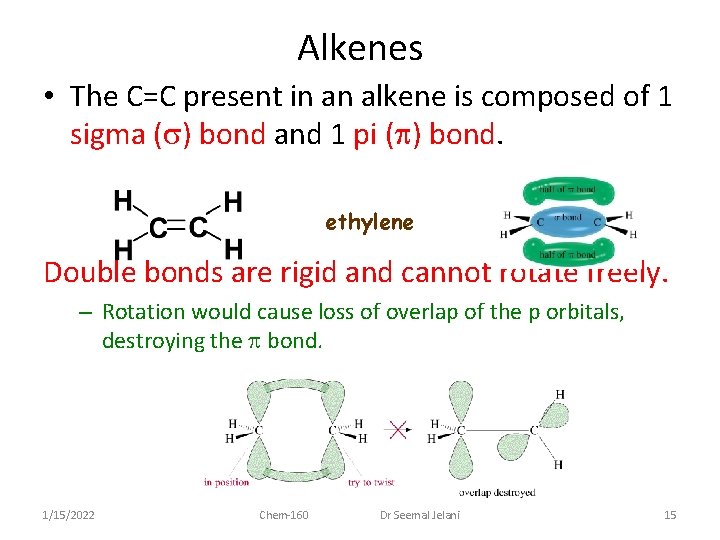
Alkenes • The C=C present in an alkene is composed of 1 sigma (s) bond and 1 pi (p) bond. ethylene Double bonds are rigid and cannot rotate freely. – Rotation would cause loss of overlap of the p orbitals, destroying the p bond. 1/15/2022 Chem-160 Dr Seemal Jelani 15

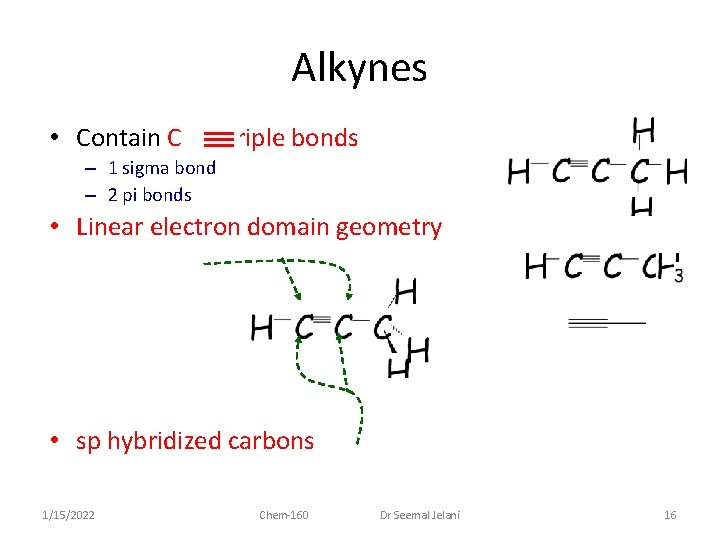
Alkynes • Contain C C triple bonds – 1 sigma bond – 2 pi bonds • Linear electron domain geometry • sp hybridized carbons 1/15/2022 Chem-160 Dr Seemal Jelani 16

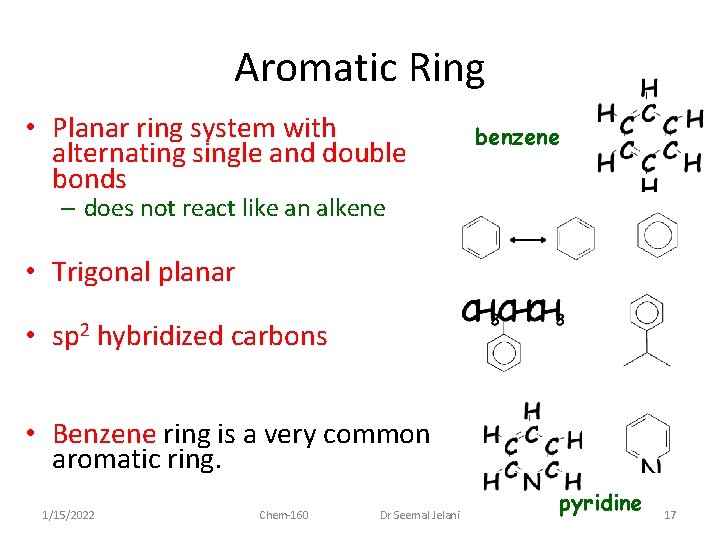
Aromatic Ring • Planar ring system with alternating single and double bonds benzene – does not react like an alkene • Trigonal planar • sp 2 hybridized carbons • Benzene ring is a very common aromatic ring. 1/15/2022 Chem-160 Dr Seemal Jelani pyridine 17

Functional Groups • Alkanes are often called saturated hydrocarbons – Organic compounds composed of carbon and hydrogen that contain the largest possible number of hydrogen atoms per carbon atom. • Alkenes, alkynes, and aromatic hydrocarbons are called unsaturated hydrocarbons – Organic compounds composed of carbon and hydrogen that contain less hydrogen than an alkane having the same number of carbon atoms 1/15/2022 Chem-160 Dr Seemal Jelani 18

Alcohols • Contain C-O-H bond – hydroxyl group • Alcohols form hydrogen bonds. • Amphoteric (acids as well as base) 1/15/2022 Chem-160 Dr Seemal Jelani 19

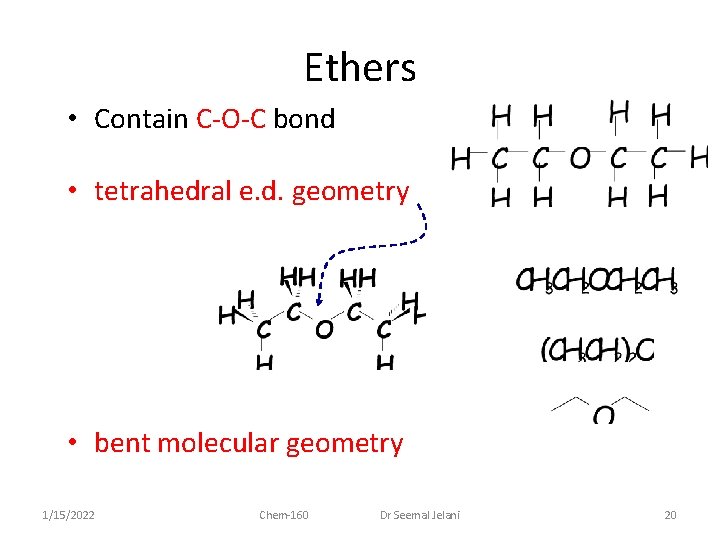
Ethers • Contain C-O-C bond • tetrahedral e. d. geometry • bent molecular geometry 1/15/2022 Chem-160 Dr Seemal Jelani 20

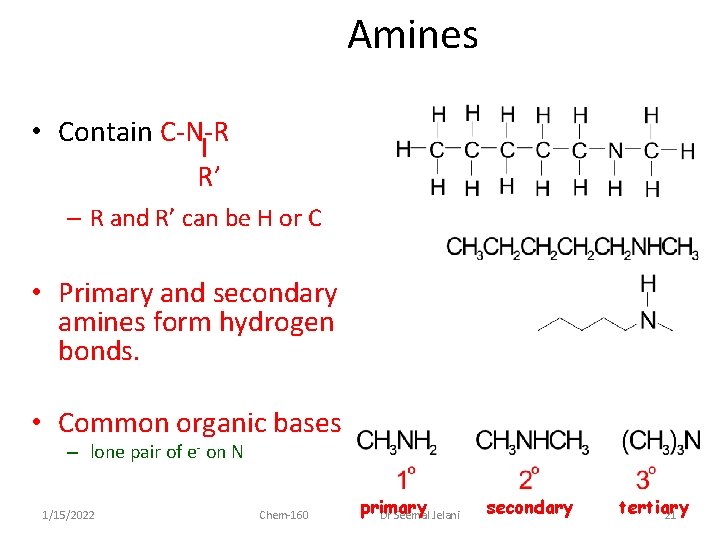
Amines • Contain C-N-R R’ – R and R’ can be H or C • Primary and secondary amines form hydrogen bonds. • Common organic bases – lone pair of e- on N 1/15/2022 Chem-160 primary Dr Seemal Jelani secondary tertiary 21

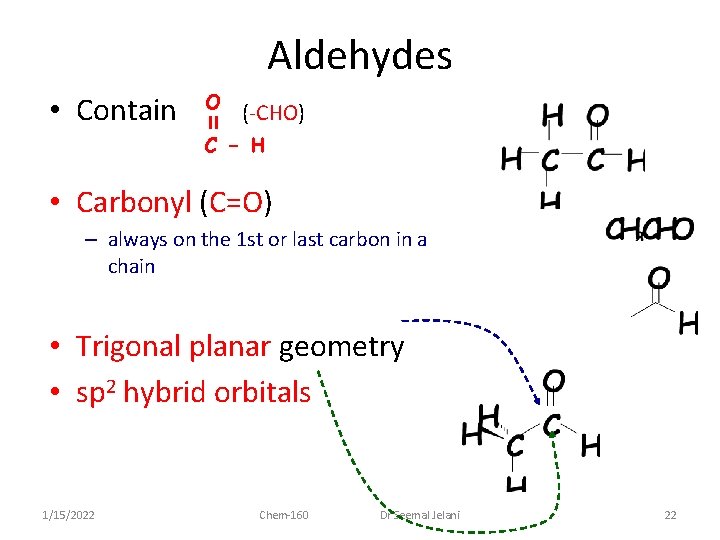
Aldehydes • Contain O C (-CHO) -H • Carbonyl (C=O) – always on the 1 st or last carbon in a chain • Trigonal planar geometry • sp 2 hybrid orbitals 1/15/2022 Chem-160 Dr Seemal Jelani 22

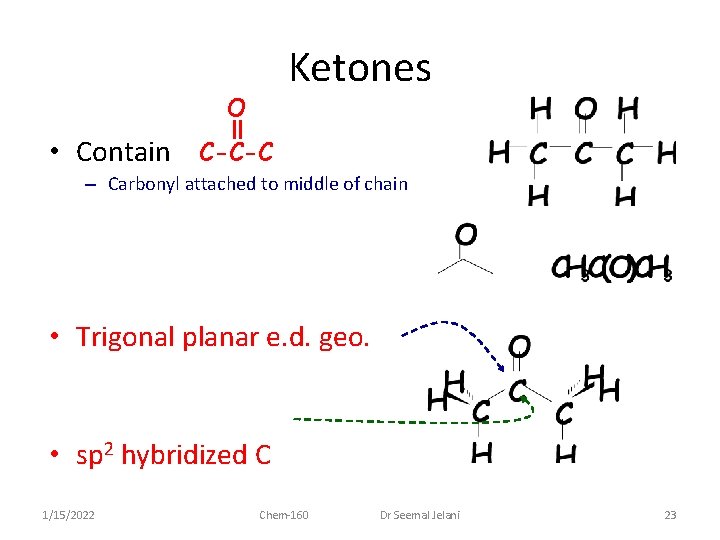
Ketones O • Contain C-C-C – Carbonyl attached to middle of chain • Trigonal planar e. d. geo. • sp 2 hybridized C 1/15/2022 Chem-160 Dr Seemal Jelani 23

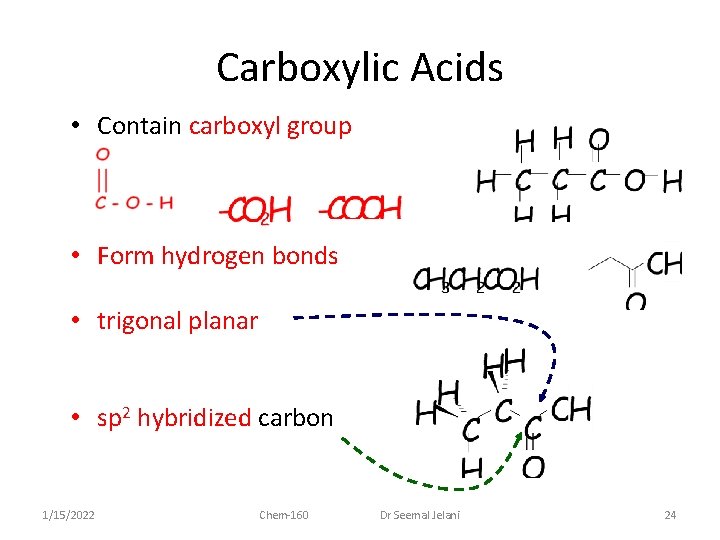
Carboxylic Acids • Contain carboxyl group • Form hydrogen bonds • trigonal planar • sp 2 hybridized carbon 1/15/2022 Chem-160 Dr Seemal Jelani 24

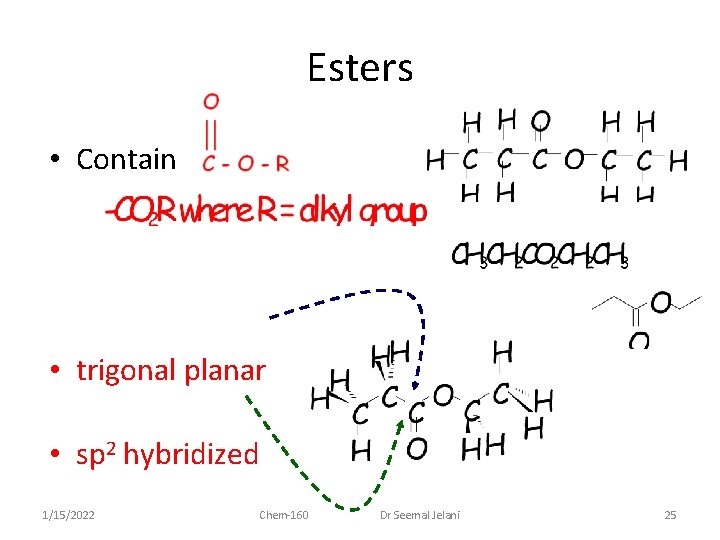
Esters • Contain • trigonal planar • sp 2 hybridized 1/15/2022 Chem-160 Dr Seemal Jelani 25

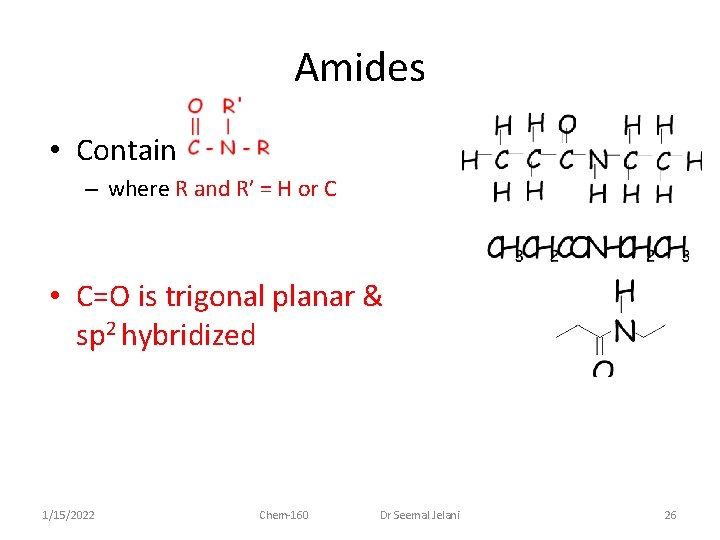
Amides • Contain – where R and R’ = H or C • C=O is trigonal planar & sp 2 hybridized 1/15/2022 Chem-160 Dr Seemal Jelani 26

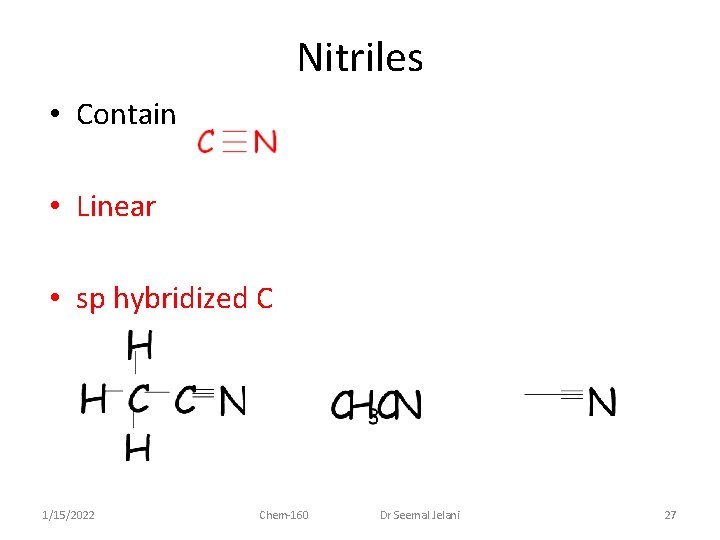
Nitriles • Contain • Linear • sp hybridized C 1/15/2022 Chem-160 Dr Seemal Jelani 27

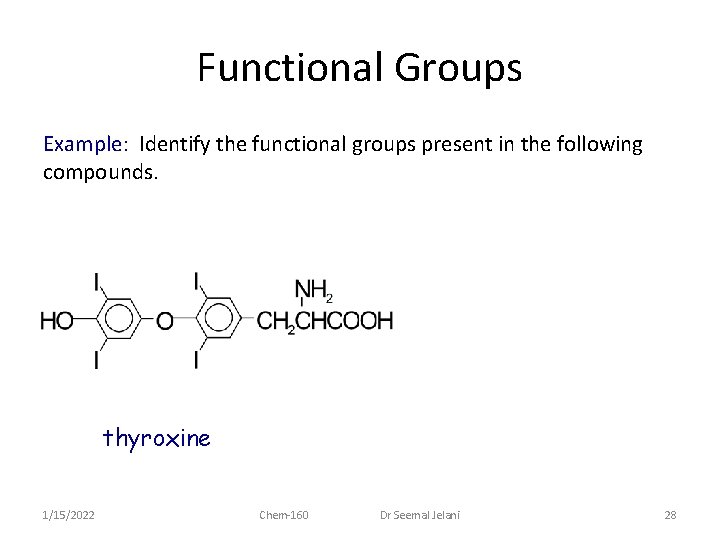
Functional Groups Example: Identify the functional groups present in the following compounds. thyroxine 1/15/2022 Chem-160 Dr Seemal Jelani 28

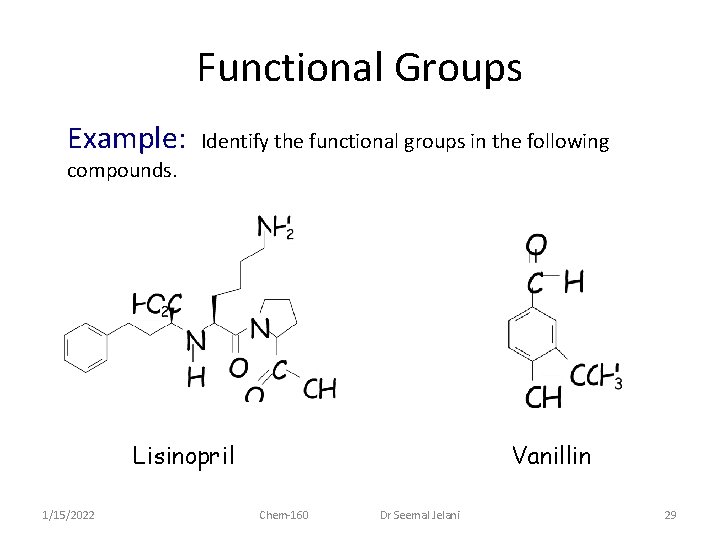
Functional Groups Example: Identify the functional groups in the following compounds. Lisinopril 1/15/2022 Vanillin Chem-160 Dr Seemal Jelani 29

Depicting Structures of Organic Compounds • Organic compounds can be depicted using a variety of formulas: – – – – Empirical formula Molecular formula Lewis structure Full structural formula Three dimensional drawings Condensed structural formula Line angle drawings 1/15/2022 Chem-160 Dr Seemal Jelani 30

Depicting Structures of Organic Compounds • Ethyl acetate is an organic molecule with: – empirical formula = C 2 H 4 O – molecular formula = C 4 H 8 O 2 1/15/2022 Chem-160 Dr Seemal Jelani 31

Depicting Structures of Organic Compounds • Ethyl acetate is an organic molecule with: – Lewis structure: • depicts all covalent bonds using a straight line and shows all nonbonding pairs of electrons – Full structural formula: • a Lewis structure without the nonbonding electrons 1/15/2022 Chem-160 Dr Seemal Jelani 32

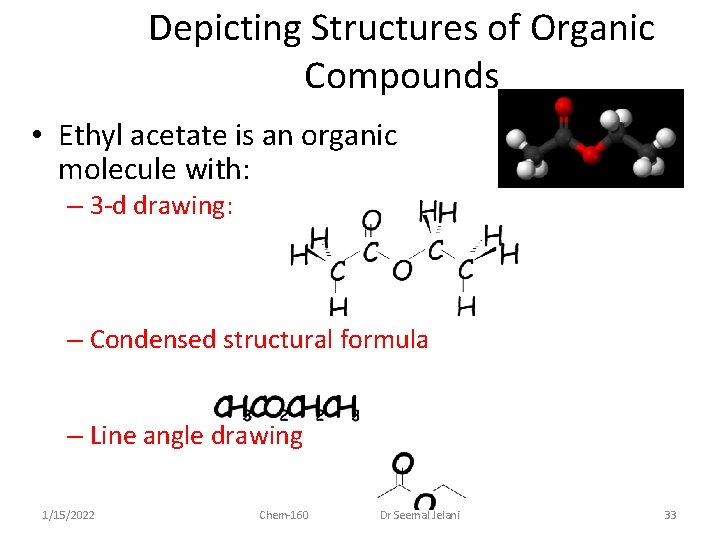
Depicting Structures of Organic Compounds • Ethyl acetate is an organic molecule with: – 3 -d drawing: – Condensed structural formula – Line angle drawing 1/15/2022 Chem-160 Dr Seemal Jelani 33

Alkanes Some of the simplest alkanes: 1/15/2022 You must know. Drthese!!! Chem-160 Seemal Jelani 34

Alkanes Some of the simplest alkanes: 1/15/2022 You must know these!!! Chem-160 Dr Seemal Jelani 35

Alkanes • The previous alkanes are also called straightchain hydrocarbons: – all of the carbon atoms are joined in a continuous chain • Alkanes containing 4 or more carbons can also form branched-chain hydrocarbons (branched hydrocarbons) – some of the carbon atoms form a “branch” or sidechain off of the main chain 1/15/2022 Chem-160 Dr Seemal Jelani 36

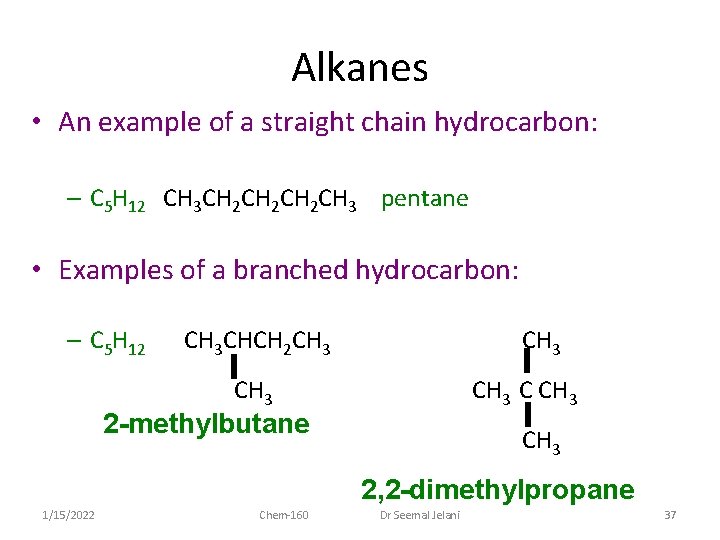
Alkanes • An example of a straight chain hydrocarbon: – C 5 H 12 CH 3 CH 2 CH 2 CH 3 pentane • Examples of a branched hydrocarbon: – C 5 H 12 CH 3 CHCH 2 CH 3 C CH 3 2 -methylbutane CH 3 2, 2 -dimethylpropane 1/15/2022 Chem-160 Dr Seemal Jelani 37

Alkanes • The three structures shown previously for C 5 H 12 are structural isomers: – compounds with the same molecular formula but different bonding arrangements • Structural isomers generally have different properties: – different melting points – different boiling points – often different chemical reactivity 1/15/2022 Chem-160 Dr Seemal Jelani 38

Alkanes • Organic compounds can be named either using common names or IUPAC names. • You must be able to name alkanes, alkenes, alkynes, and alcohols with 10 or fewer carbons in the main chain using the IUPAC naming system. 1/15/2022 Chem-160 Dr Seemal Jelani 39

Alkanes • Alkane Nomenclature: – Find the longest continuous chain of carbon atoms and use the name of the chain for the base name of the compound: • longest chain may not always be written in a straight line 1 CH 3 -2 CH - CH 3 3 CH 2 1/15/2022 - CH 3 4 5 Chem-160 Base name: hexane 6 Dr Seemal Jelani 40

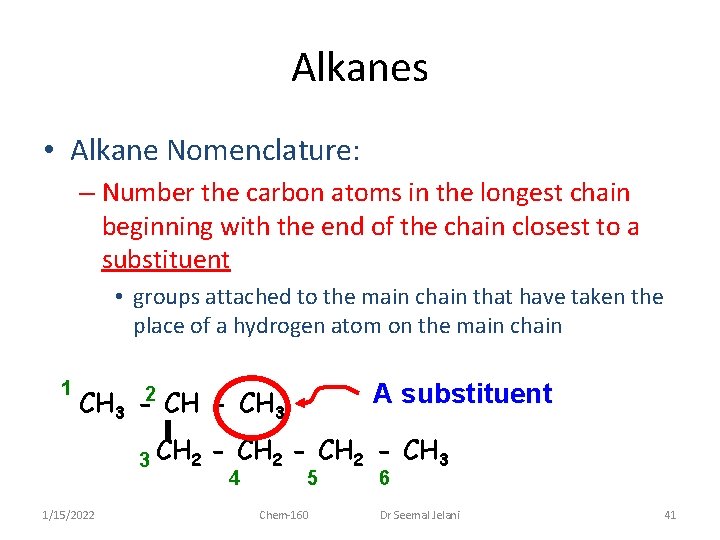
Alkanes • Alkane Nomenclature: – Number the carbon atoms in the longest chain beginning with the end of the chain closest to a substituent • groups attached to the main chain that have taken the place of a hydrogen atom on the main chain 1 A substituent CH 3 -2 CH - CH 3 3 CH 2 1/15/2022 - CH 3 4 5 Chem-160 6 Dr Seemal Jelani 41

Alkanes • Alkane Nomenclature: – Name and give the location of each substituent group • A substituent group that is formed by removing an H atom from an alkane is called an alkyl group: – Name alkyl groups by dropping the “ane” ending of the parent alkane and adding “yl” 1/15/2022 Chem-160 Dr Seemal Jelani 42

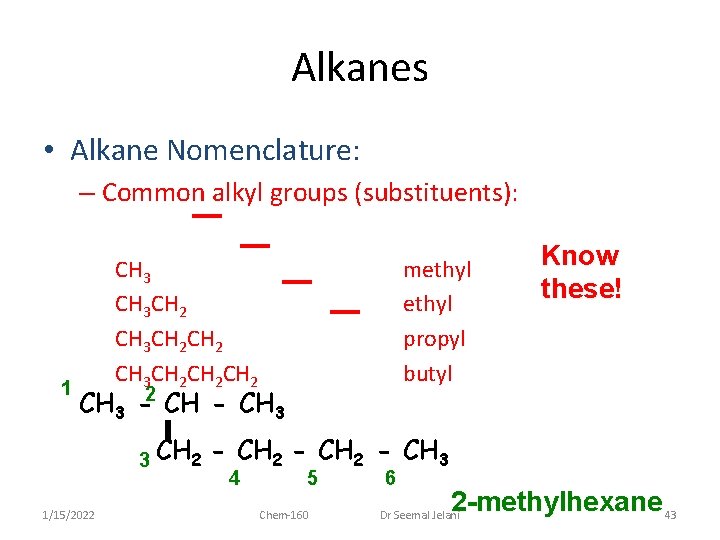
Alkanes • Alkane Nomenclature: – Common alkyl groups (substituents): 1 CH 3 CH 2 CH 2 methyl propyl butyl CH 3 -2 CH - CH 3 3 CH 2 1/15/2022 Know these! - CH 2 - CH 3 4 5 Chem-160 6 2 -methylhexane 43 Dr Seemal Jelani

Alkanes • Alkane Nomenclature: – Halogen atoms are another common class of substituents. • Name halogen substituents as “halo”: – – 1/15/2022 Cl chloro Brbromo I iodo F fluoro Chem-160 Dr Seemal Jelani 44

Alkanes • Alkane Nomenclature: – When two or more substituents are present, list them in alphabetical order: • Butyl vs. ethyl vs. methyl vs. propyl – When more than one of the same substituent is present (i. e. two methyl groups), use prefixes to indicate the number: • • 1/15/2022 Di = two Tri = three Tetra = four Penta = five Know these. Chem-160 Dr Seemal Jelani 45

Alkanes Example: Name the following compounds: CH 3 CH 2 CH 3 CH 3 CHCHCH 3 1/15/2022 Chem-160 Dr Seemal Jelani 46

Alkanes Example: Name the following compounds: CH 3 CH 2 CHCH 3 CH 2 Br CH 2 CH 3 CHCHCH 3 Cl 1/15/2022 Chem-160 Dr Seemal Jelani 47

Alkanes • You must also be able to write the structure of an alkane when given the IUPAC name. • To do so: – Identify the main chain and draw the carbons in it – Identify the substituents (type and #) and attach them to the appropriate carbon atoms on the main chain. – Add hydrogen atoms to the carbons to make a total of 4 bonds to each carbon 1/15/2022 Chem-160 Dr Seemal Jelani 48

Alkanes Example: Write the condensed structure for the following compounds: 3, 3 -dimethylpentane 3 -ethyl-2 -methylhexane 2 -methyl-4 -propyloctane 1, 2 -dichloro-3 -methylheptane 1/15/2022 Chem-160 Dr Seemal Jelani 49

Alkenes • Alkenes: – unsaturated hydrocarbons that contain a C=C double bond • Alkene Nomenclature: – Names of alkenes are based on the longest continuous chain of carbon atoms that contains the double bond. 1/15/2022 Chem-160 Dr Seemal Jelani 50

Alkenes • Alkene Nomenclature – Find the longest continuous carbon chain containing the double bond. – Change the “ane” ending from the corresponding alkane to “ene” • butane • propane • octane 1/15/2022 butene propene octene Chem-160 Dr Seemal Jelani 51

Alkenes • Alkene Nomenclature – Indicate the location of the double bond using a prefix number • designate the carbon atom that is part of the double bond AND nearest to the end of the chain – Name all other substituents in a manner similar to the alkanes. – Use a prefix to indicate the geometric isomer present, if necessary. 1/15/2022 Chem-160 Dr Seemal Jelani 52

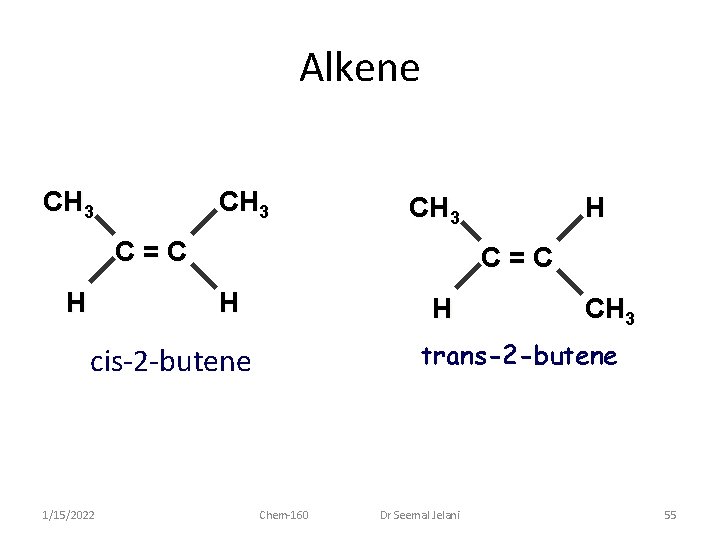
Alkenes • Alkene Nomenclature • Different geometric isomers are possible for many alkenes. – Compounds that have the same molecular formula and the same groups bonded to each other, but different spatial arrangements of the groups • cis isomer • trans isomer 1/15/2022 Chem-160 Dr Seemal Jelani 53

Alkenes • Alkene Nomenclature – Cis isomer: • two identical groups (on adjacent carbons) on the same side of the C = C double bond – Trans isomer: • two identical groups (on adjacent carbons) on opposite sides of the C = C double bond 1/15/2022 Chem-160 Dr Seemal Jelani 54

Alkene CH 3 C=C H H CH 3 trans-2 -butene cis-2 -butene 1/15/2022 H Chem-160 Dr Seemal Jelani 55

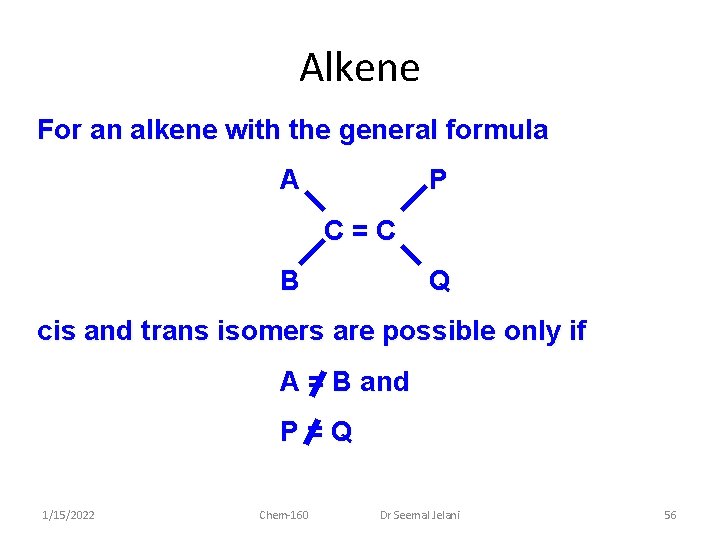
Alkene For an alkene with the general formula A P C=C B Q cis and trans isomers are possible only if A = B and P=Q 1/15/2022 Chem-160 Dr Seemal Jelani 56

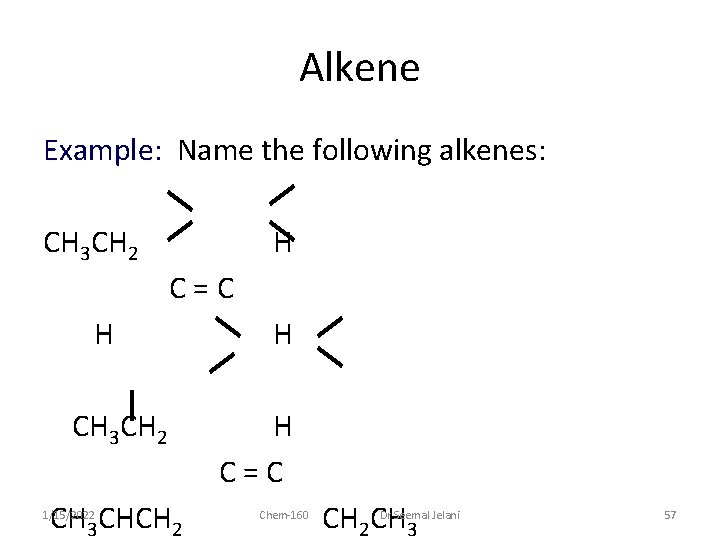
Alkene Example: Name the following alkenes: CH 3 CH 2 H C=C H CH 3 CH 2 CH 3 CHCH 2 1/15/2022 H H C=C Chem-160 CH 2 CH 3 Dr Seemal Jelani 57

Alkenes Example: Draw the structures for the following compounds: 2 -chloro-3 -methyl-2 -butene trans-3, 4 -dimethyl-2 -pentene cis-6 -methyl-3 -heptene 1/15/2022 Chem-160 Dr Seemal Jelani 58

Alkynes • Alkynes: – unsaturated hydrocarbons that contain a C C triple bond • Alkyne Nomenclature: – Identify the longest continuous chain containing the triple bond – To find the base name, change the ending of the alkane from “ane” to “yne” 1/15/2022 corresponding Chem-160 Dr Seemal Jelani 59

Alkynes • Alkyne Nomenclature: – Use a number to designate the position of the triple bond • number from the end of the chain closest to the triple bond – just like with alkenes – Name substituents like you do with alkanes and alkenes 1/15/2022 Chem-160 Dr Seemal Jelani 60

Alkynes Example: Name the following compounds: CH 3 CH 2 C CCHCH 3 CH 2 CH 3 Cl. CH 2 C CH 1/15/2022 Chem-160 Dr Seemal Jelani 61

Alkynes Example: Draw the following alkynes. 4 -chloro-2 -pentyne 3 -propyl-1 -hexyne 1/15/2022 Chem-160 Dr Seemal Jelani 62