Hybridization 9302020 Dr Seemal Jelani Chem160 1 Hybridization

Hybridization 9/30/2020 Dr Seemal Jelani Chem-160 1

• Hybridization of atomic orbitals is the intermixing of atomic orbitals having an unpaired electron and approximate energy to form equal number of hybrid orbitals having the same shape, size and energy. 9/30/2020 Dr Seemal Jelani Chem-160 2

• The new orbitals which are formed are "hybrids" of the originals and have properties that are somewhere in between. • For example, a common hybridization is sp 3 where three p orbitals combine with an s orbital to form four new orbitals. Other combinations (such as sp and sp 2) are also possible. 9/30/2020 Dr Seemal Jelani Chem-160 3

9/30/2020 Dr Seemal Jelani Chem-160 4

9/30/2020 Dr Seemal Jelani Chem-160 5

9/30/2020 Dr Seemal Jelani Chem-160 6
9/30/2020 Dr Seemal Jelani Chem-160 7

The forces at work are: Electromagnetic: Repulsion of like charges: proton-proton and electron-electron Attraction of unlike charges: proton-electron. Magnetic: Electron spin introduces magnetic forces. Electrons with opposite spins produce opposite magnetic poles. Opposite magnetic poles attract. Opposite spin is required for electrons to occupy the same orbital. 9/30/2020 Dr Seemal Jelani Chem-160 8
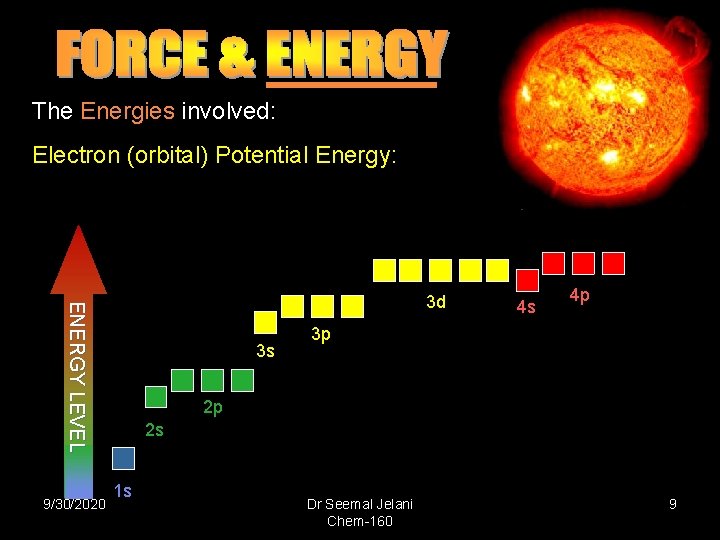
The Energies involved: Electron (orbital) Potential Energy: E NE RG Y LE V E L 3 d 9/30/2020 3 s 4 s 4 p 3 p 2 p 2 s 1 s Dr Seemal Jelani Chem-160 9
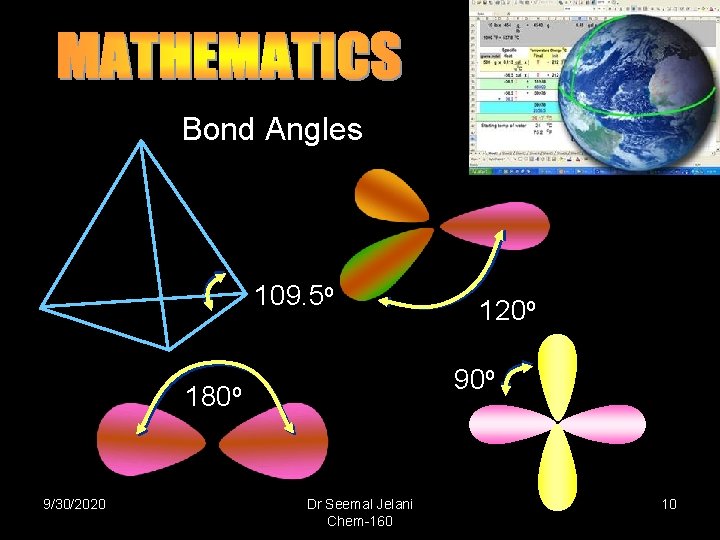
Bond Angles 109. 5 o 90 o 180 o 9/30/2020 120 o Dr Seemal Jelani Chem-160 10
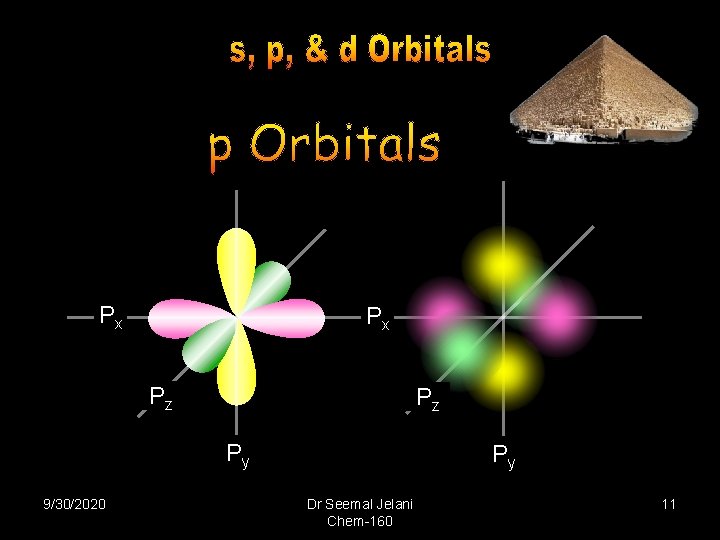
Px Px Pz Pz Py 9/30/2020 Py Dr Seemal Jelani Chem-160 11
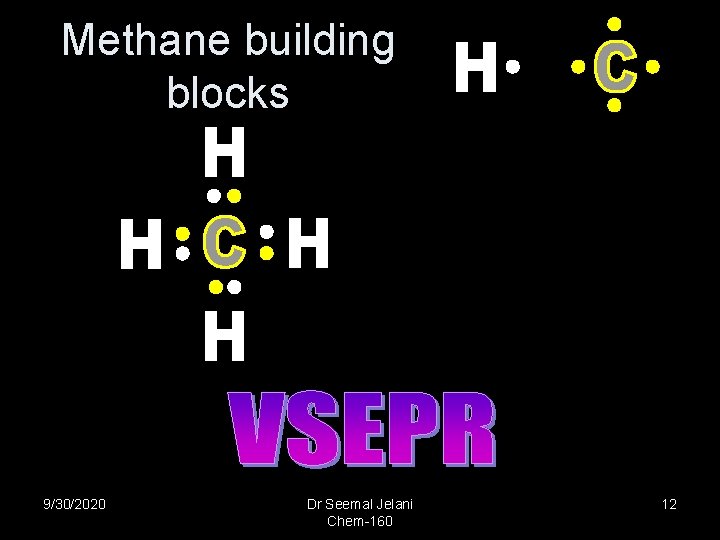
Methane building blocks 9/30/2020 Dr Seemal Jelani Chem-160 12
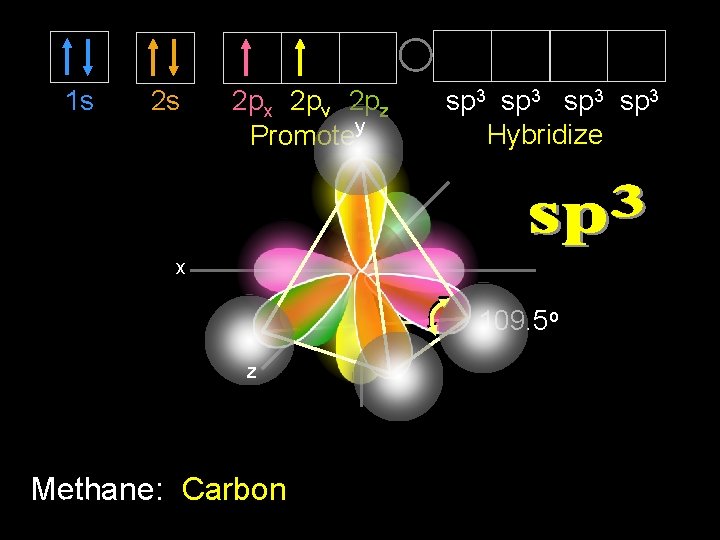
1 s 2 s 2 px 2 py 2 pz Promotey sp 3 Hybridize x 109. 5 o z Methane: Carbon 9/30/2020 Dr Seemal Jelani Chem-160 13
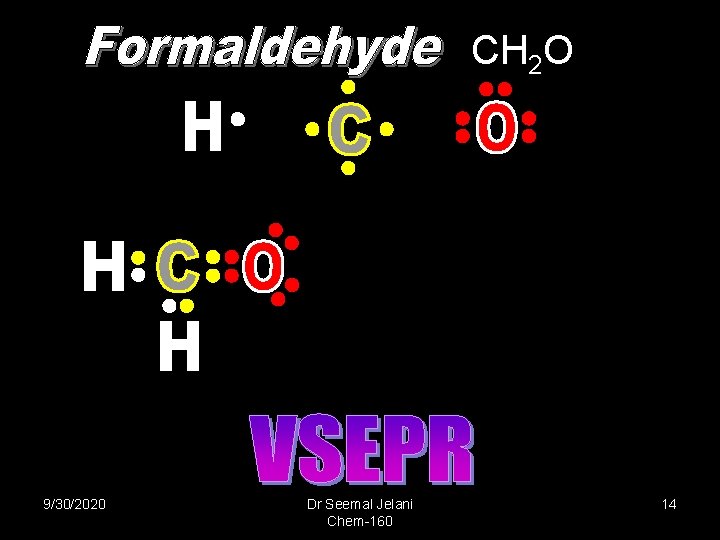
CH 2 O 120 o 9/30/2020 Dr Seemal Jelani Chem-160 14
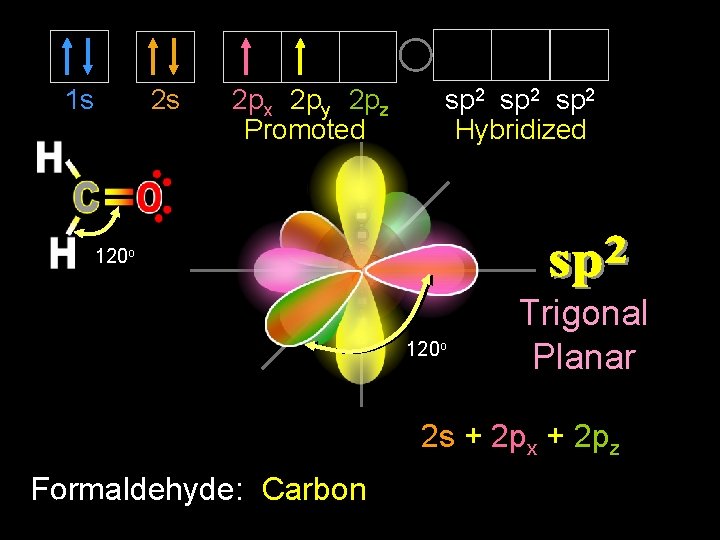
1 s 2 s sp 2 Hybridized 2 px 2 py 2 pz Promoted 120 o Trigonal Planar 2 s + 2 px + 2 pz Formaldehyde: Carbon 9/30/2020 Dr Seemal Jelani Chem-160 15
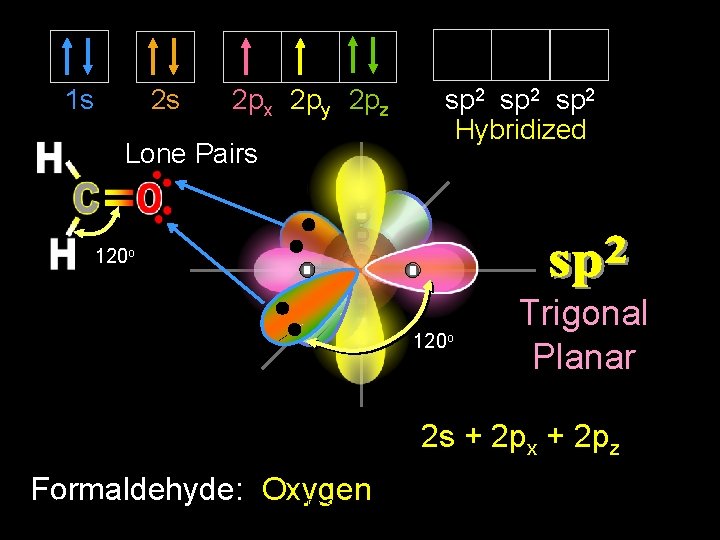
1 s 2 s 2 px 2 py 2 pz Lone Pairs sp 2 Hybridized 120 o Trigonal Planar 2 s + 2 px + 2 pz Formaldehyde: Oxygen 9/30/2020 Dr Seemal Jelani Chem-160 16
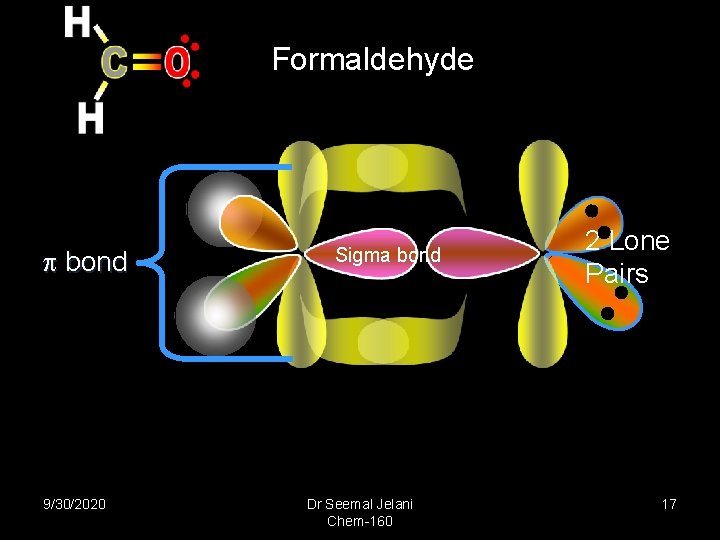
Formaldehyde bond 9/30/2020 Sigma bond Dr Seemal Jelani Chem-160 2 Lone Pairs 17
9/30/2020 Dr Seemal Jelani Chem-160 18
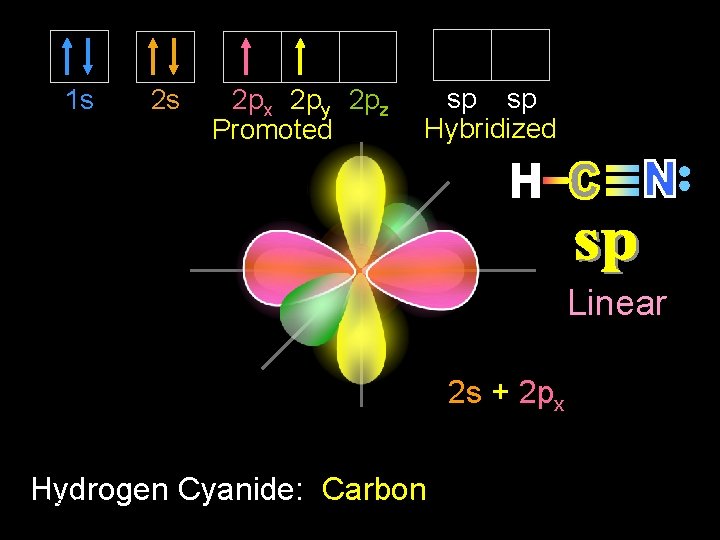
1 s 2 s 2 px 2 py 2 pz Promoted sp sp Hybridized Linear 2 s + 2 px Hydrogen Cyanide: Dr. Carbon 9/30/2020 Seemal Jelani Chem-160 19
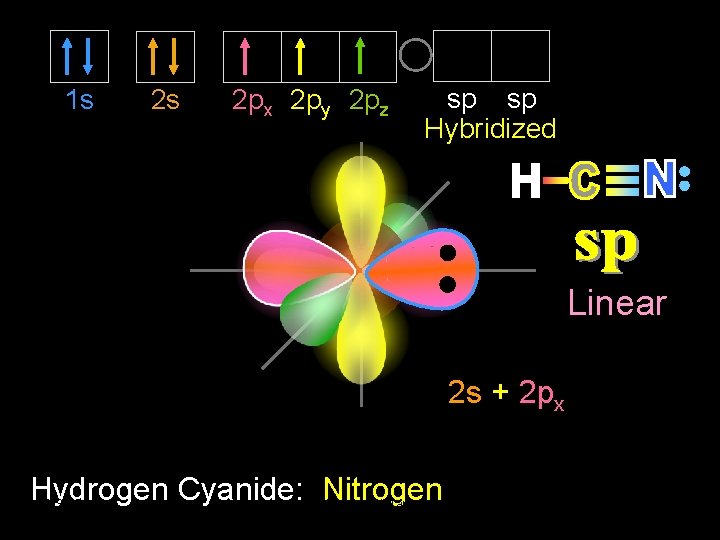
1 s 2 s 2 px 2 py 2 pz sp sp Hybridized Linear 2 s + 2 px Hydrogen Cyanide: Dr. Nitrogen 9/30/2020 Seemal Jelani Chem-160 20
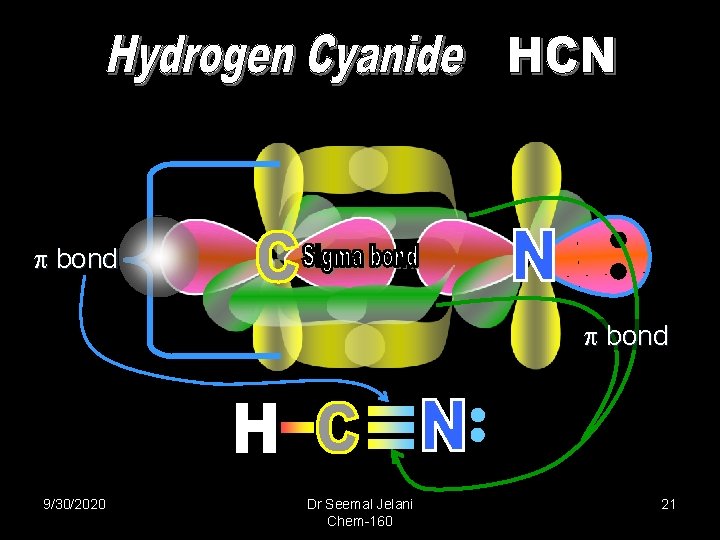
bond 9/30/2020 Dr Seemal Jelani Chem-160 21

PCl 5 9/30/2020 Dr Seemal Jelani Chem-160 22
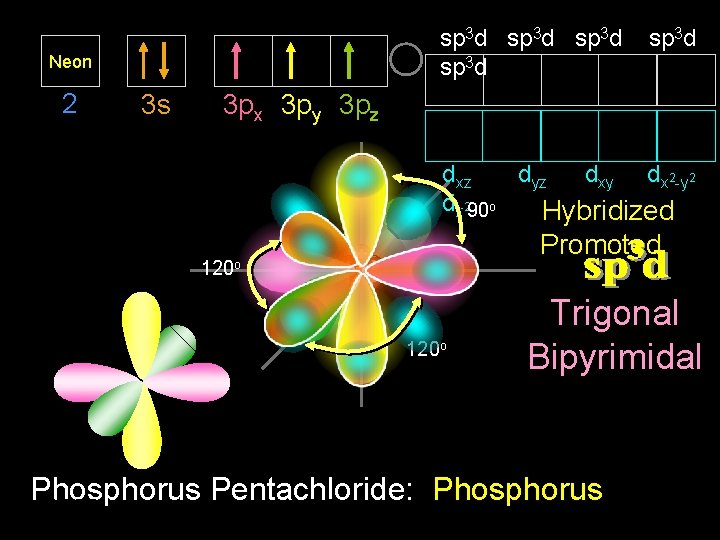
sp 3 d Neon 2 3 s sp 3 d 3 px 3 py 3 pz dxz dyz dxy dx 2 -y 2 dz 290 o Hybridized Promoted 120 o Trigonal Bipyrimidal Phosphorus Pentachloride: Phosphorus 9/30/2020 Dr Seemal Jelani Chem-160 23
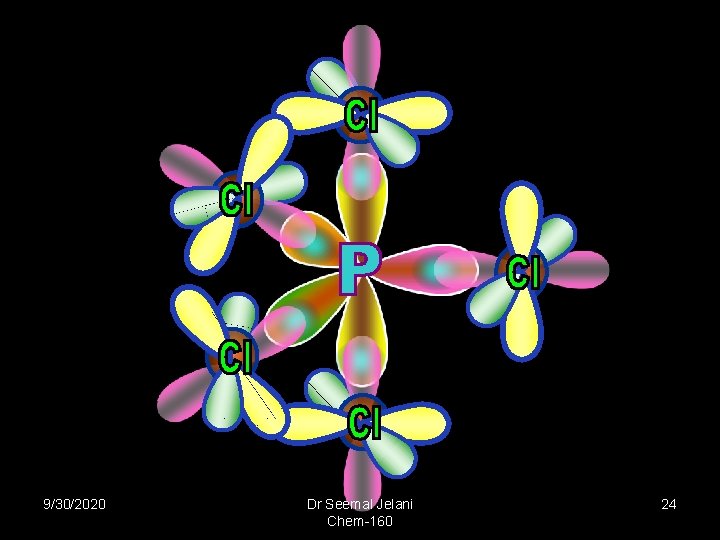
9/30/2020 Dr Seemal Jelani Chem-160 24

9/30/2020 Dr Seemal Jelani Chem-160 25
- Slides: 25