Organic Chemistry P 267 Hydrocarbon Hydro hydrogen Carbon

Organic Chemistry P 267

Hydrocarbon • Hydro: hydrogen • Carbon • Additional element: Nitrogen, Oxygen, Sulphur Atomic number of carbon = 6 Valence electrons of carbon = 4 Each carbon atom has 4 bond in compound Ex: alkanes Alkanes: hydrocarbon that consist of hydrogen and carbon, (single bond)
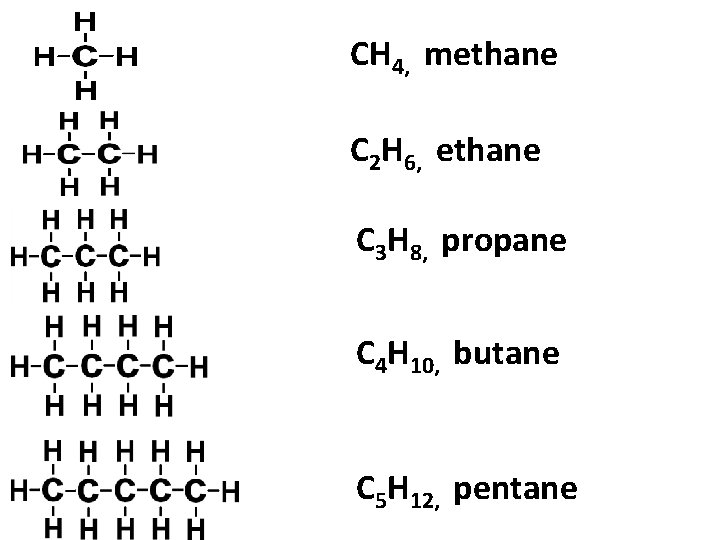
CH 4, methane C 2 H 6, ethane C 3 H 8, propane C 4 H 10, butane C 5 H 12, pentane
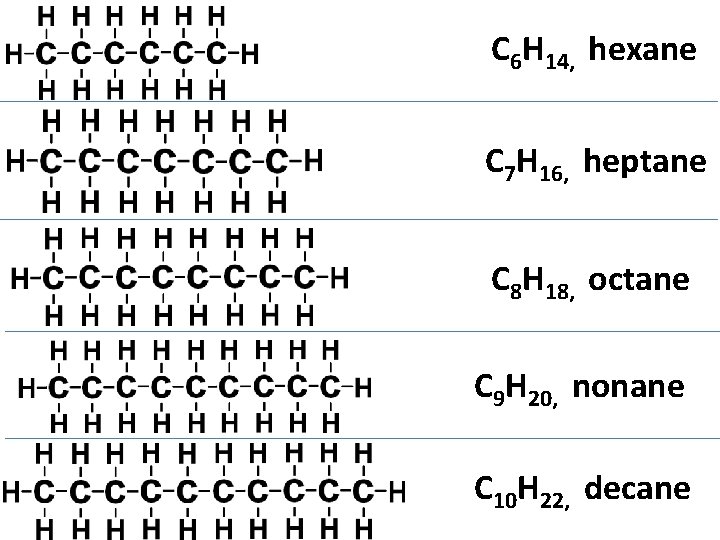
C 6 H 14, hexane C 7 H 16, heptane C 8 H 18, octane C 9 H 20, nonane C 10 H 22, decane
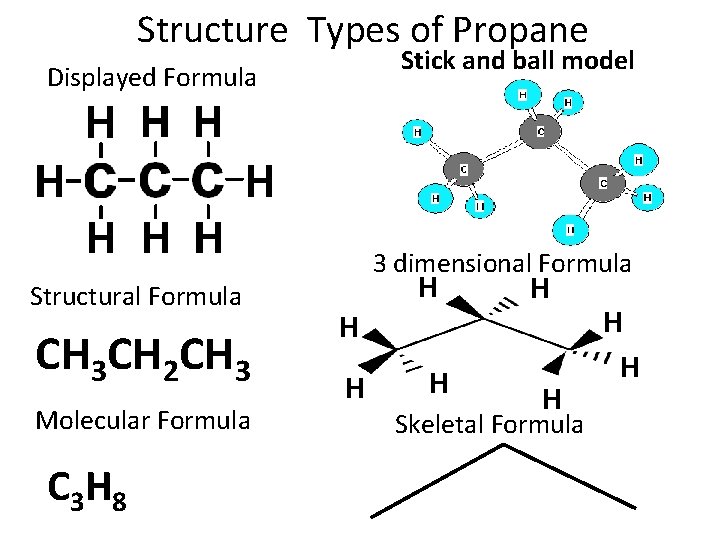
Structure Types of Propane Stick and ball model Displayed Formula Structural Formula CH 3 CH 2 CH 3 Molecular Formula C 3 H 8 3 dimensional Formula H H H Skeletal Formula H H
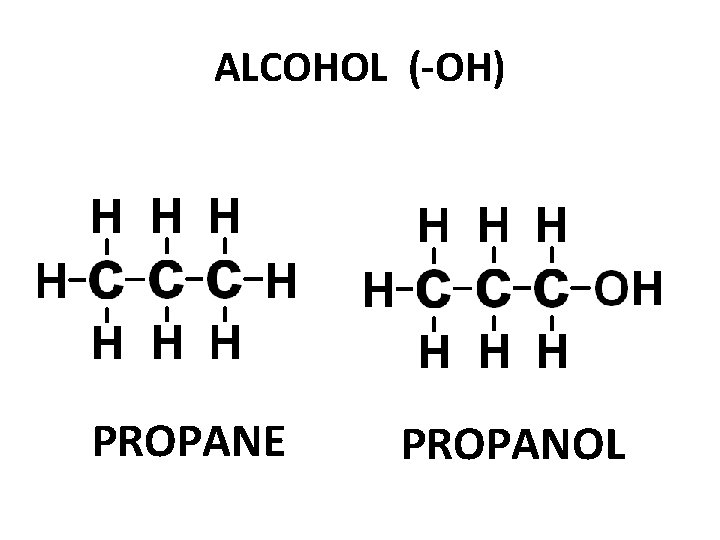
ALCOHOL (-OH) PROPANE PROPANOL
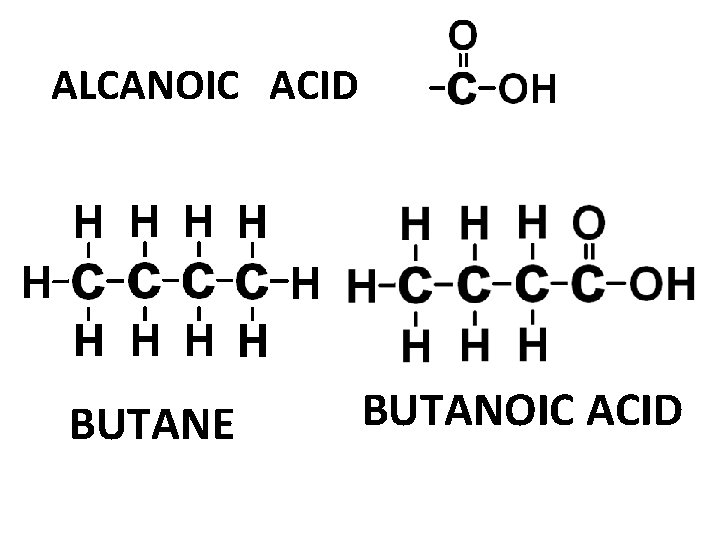
ALCANOIC ACID BUTANE BUTANOIC ACID
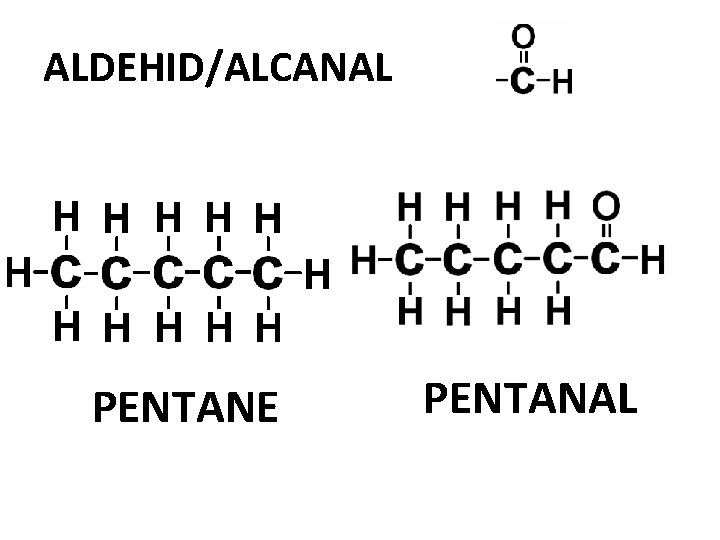
ALDEHID/ALCANAL PENTANE PENTANAL
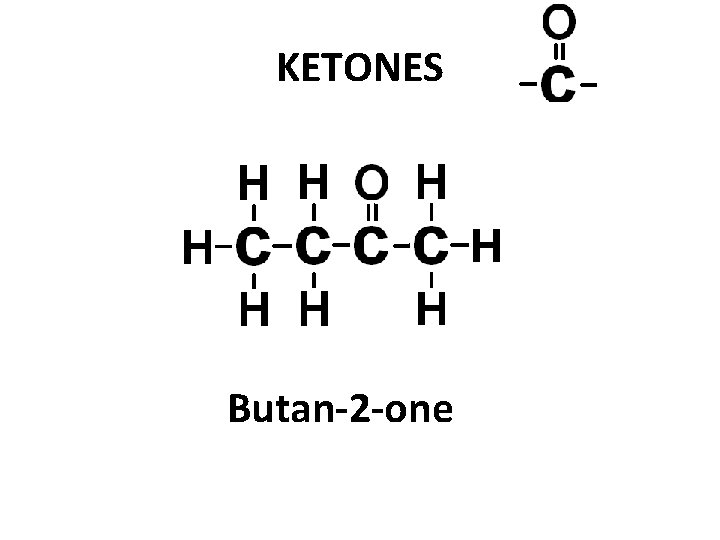
KETONES Butan-2 -one
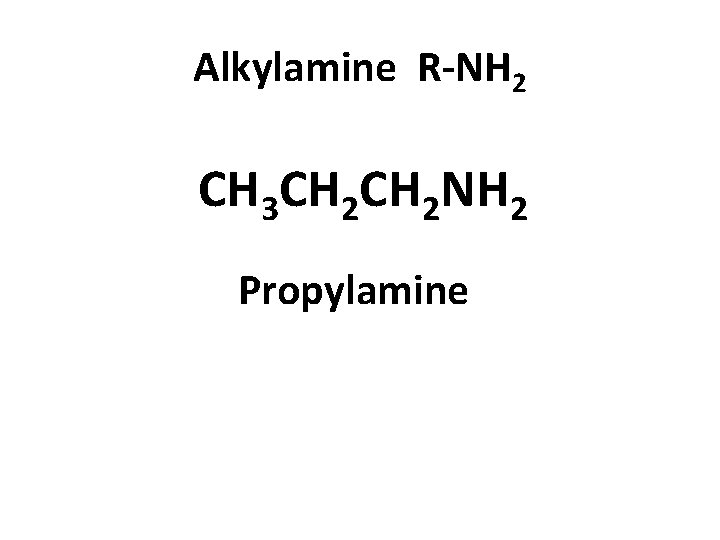
Alkylamine R-NH 2 CH 3 CH 2 NH 2 Propylamine
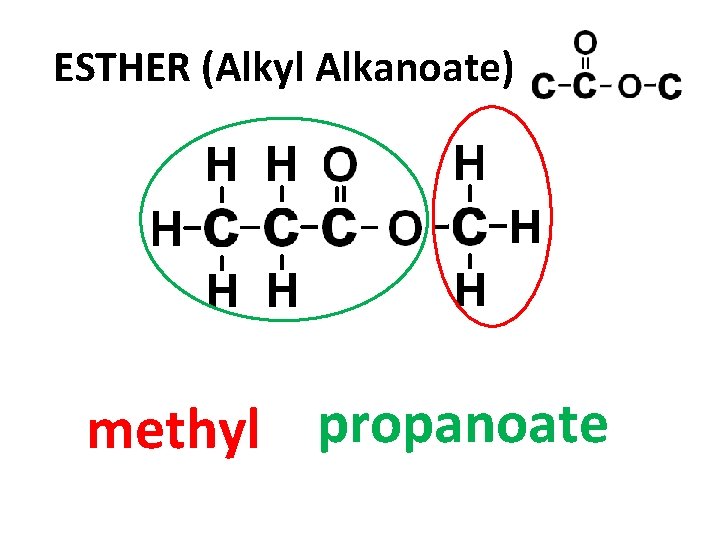
ESTHER (Alkyl Alkanoate) methyl propanoate
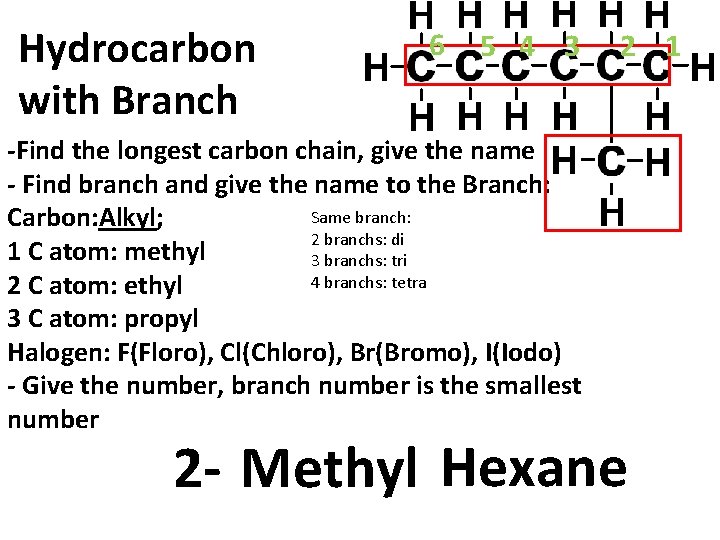
Hydrocarbon with Branch 6 5 4 3 -Find the longest carbon chain, give the name - Find branch and give the name to the Branch: Same branch: Carbon: Alkyl; 2 branchs: di 1 C atom: methyl 3 branchs: tri 4 branchs: tetra 2 C atom: ethyl 3 C atom: propyl Halogen: F(Floro), Cl(Chloro), Br(Bromo), I(Iodo) - Give the number, branch number is the smallest number 2 1 2 - Methyl Hexane
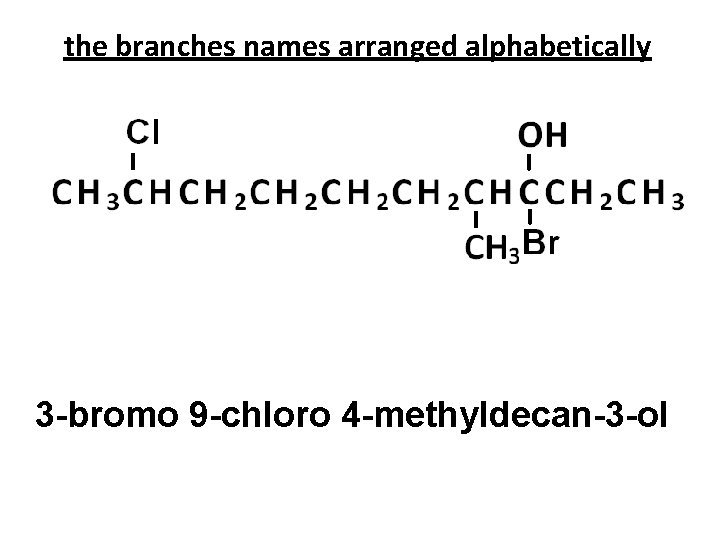
the branches names arranged alphabetically 3 -bromo 9 -chloro 4 -methyldecan-3 -ol
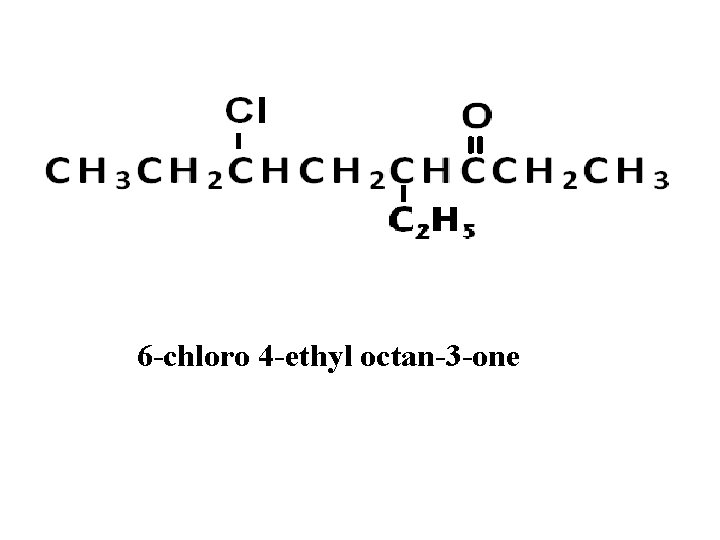
6 -chloro 4 -ethyl octan-3 -one
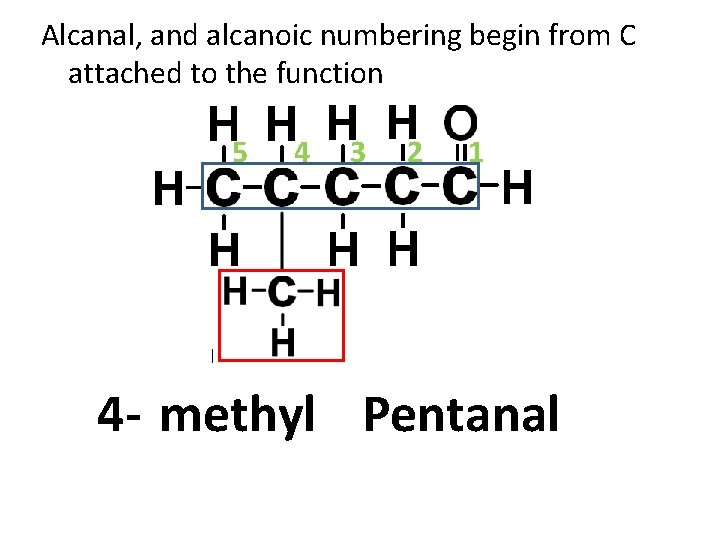
Alcanal, and alcanoic numbering begin from C attached to the function 5 4 3 2 1 4 - methyl Pentanal
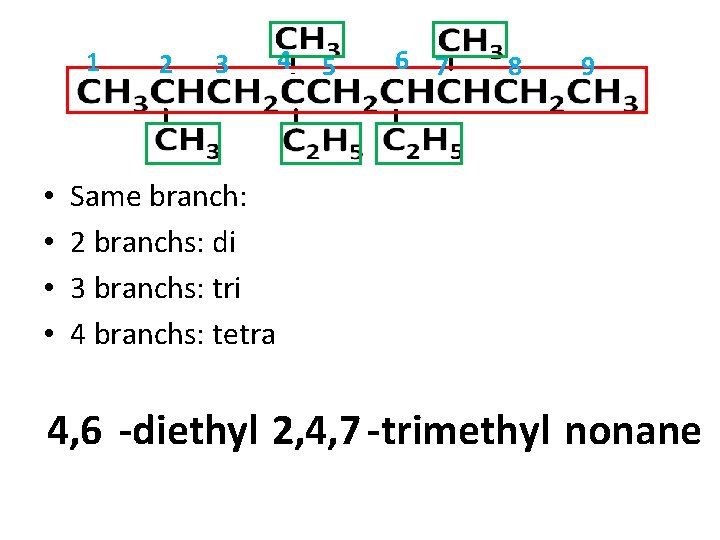
1 • • 2 3 4 5 6 7 8 9 Same branch: 2 branchs: di 3 branchs: tri 4 branchs: tetra 4, 6 -diethyl 2, 4, 7 -trimethyl nonane
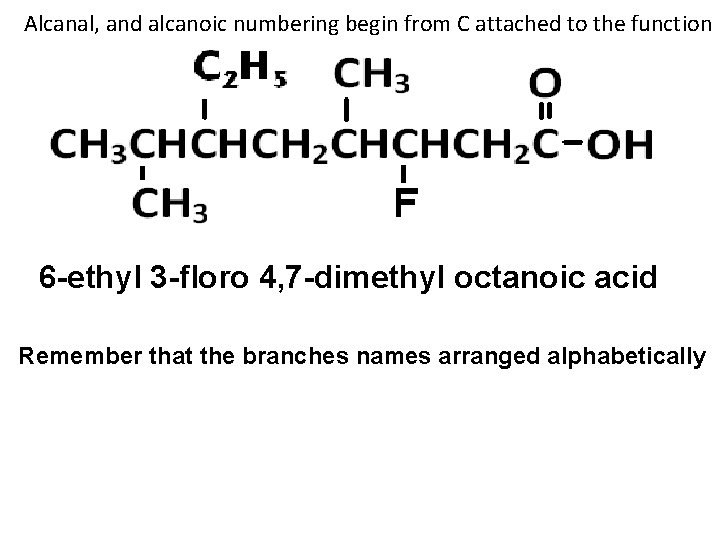
Alcanal, and alcanoic numbering begin from C attached to the function 6 -ethyl 3 -floro 4, 7 -dimethyl octanoic acid Remember that the branches names arranged alphabetically

Task • Copy the table 15. 2 page 270 in to your book
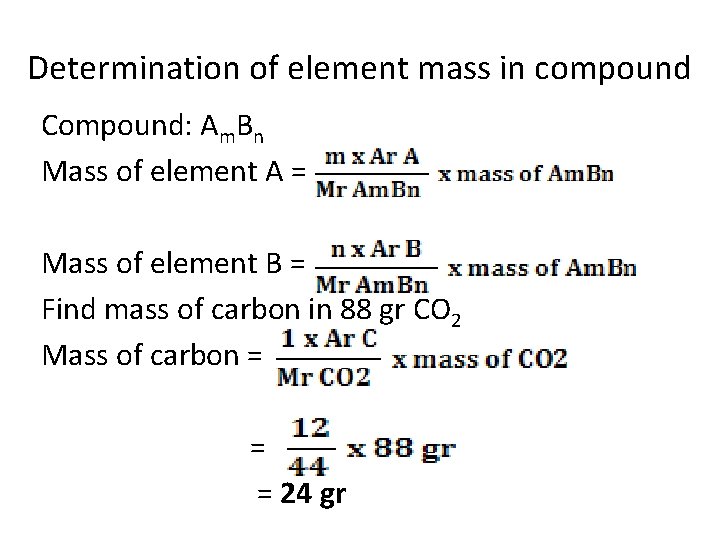
Determination of element mass in compound Compound: Am. Bn Mass of element A = Mass of element B = Find mass of carbon in 88 gr CO 2 Mass of carbon = = = 24 gr

Determination of empirical formulae of organic compound 100, 00 g amino acid(Cp. Hq. Or. Ns) react with oxygen and produced 117, 10 g of carbon dioxide and 59, 92 g of water. In kjeldahl a second of amino acid produced 22, 64 g ammonia, determine the empirical formula of amino acid Answer - write the information mass of amino acid = 100 gr mass of carbon dioxide (CO 2) = 117, 10 gr mass of water(H 2 O)= 59, 92 g mass of ammonia (NH 3) = 22, 64
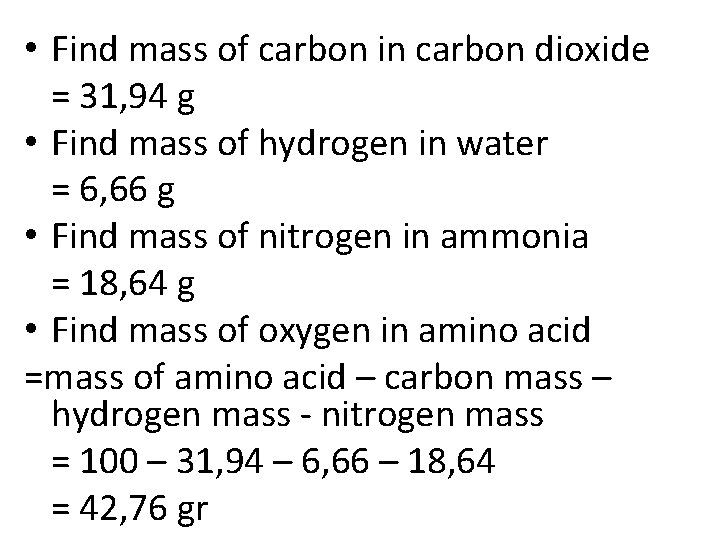
• Find mass of carbon in carbon dioxide = 31, 94 g • Find mass of hydrogen in water = 6, 66 g • Find mass of nitrogen in ammonia = 18, 64 g • Find mass of oxygen in amino acid =mass of amino acid – carbon mass – hydrogen mass - nitrogen mass = 100 – 31, 94 – 6, 66 – 18, 64 = 42, 76 gr

• Compare mass of the elements C : H : O : N 31, 94 : 6, 66 : 42, 7 : 18, 64 • Compare mol of the elements 31, 94/12 : 6, 66/1 : 42, 76/16 : 18, 64/14 2, 67 : 6, 66 : 2, 67 : 1, 33 • Divide by the smallest mole 2 : 5 : 2 : 1 • Determine the empirical formula C 2 H 5 O 2 N
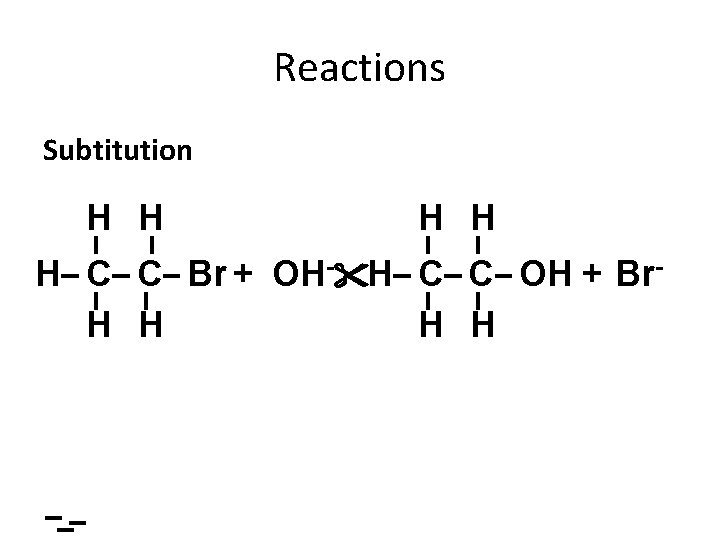
Reactions Subtitution H H H C C Br + OH- H C C OH + Br. H H
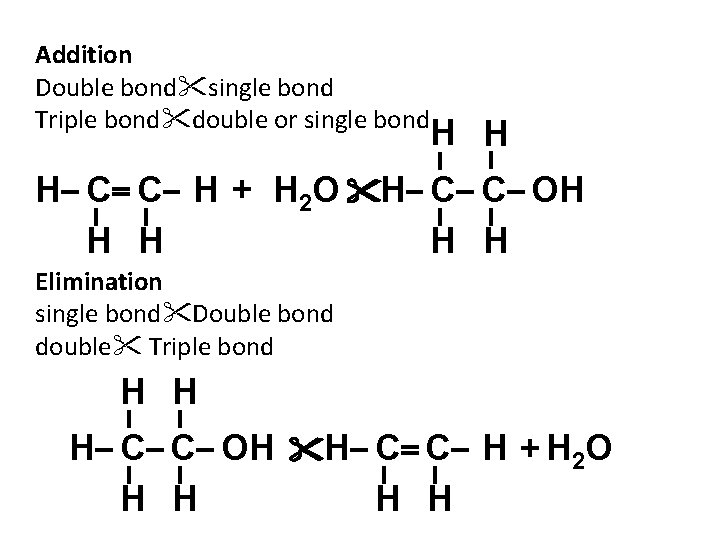
Addition Double bond single bond Triple bond double or single bond H H H C C H + H 2 O H C C OH H H Elimination single bond Double bond double Triple bond H H H C C OH H C C H + H 2 O H H
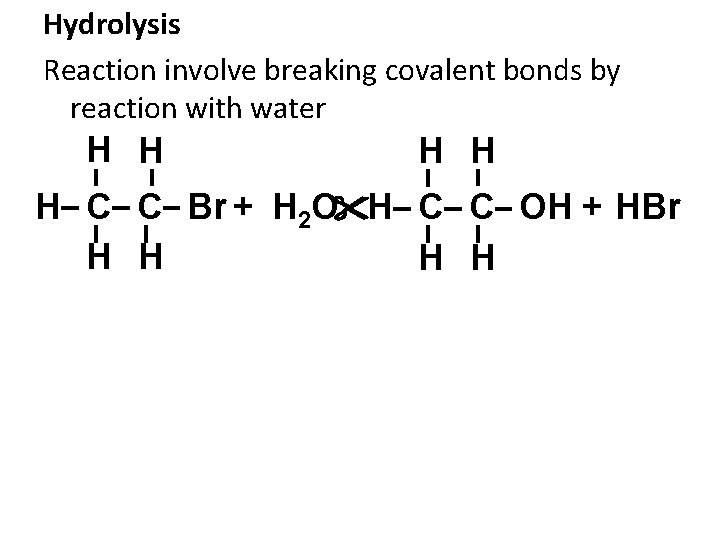
Hydrolysis Reaction involve breaking covalent bonds by reaction with water H H H C C Br + H 2 O H C C OH + HBr H H

Breaking Bonds in Different Ways Homolytic fission Each atom break to free radical (atom with unpaired electron + Heterolytic fission Each atom break to ion (cation and anion) ++ -
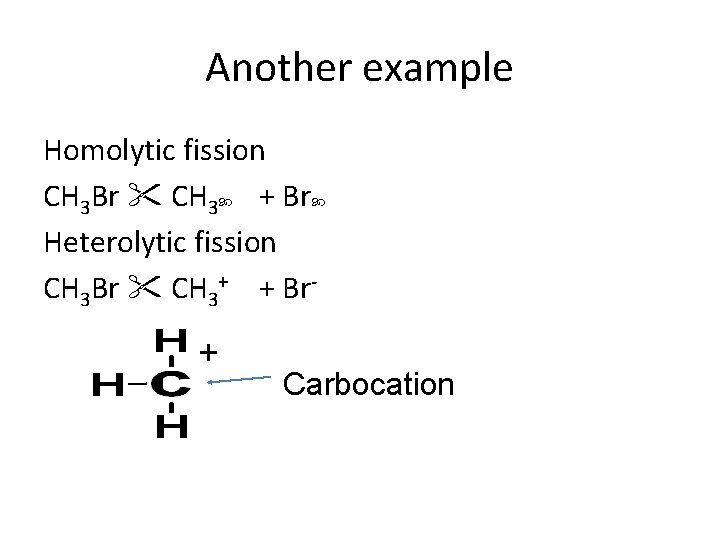
Another example Homolytic fission CH 3 Br CH 3 + Br Heterolytic fission CH 3 Br CH 3+ + Br- + Carbocation
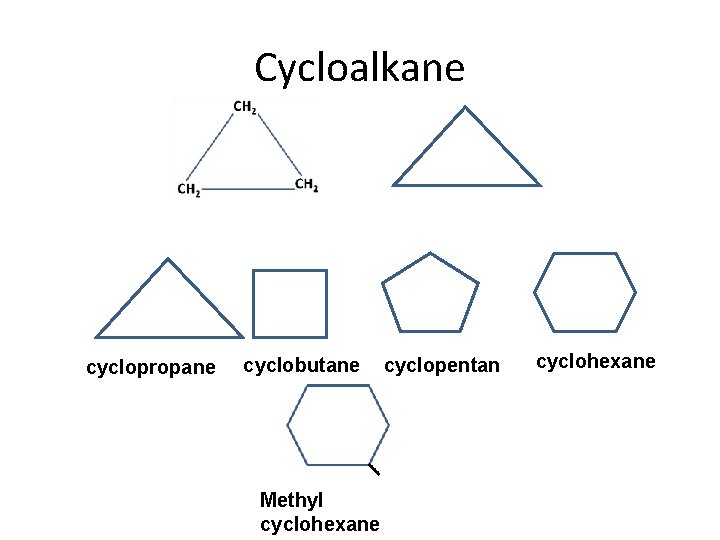
Cycloalkane cyclopropane cyclobutane Methyl cyclohexane cyclopentan e cyclohexane
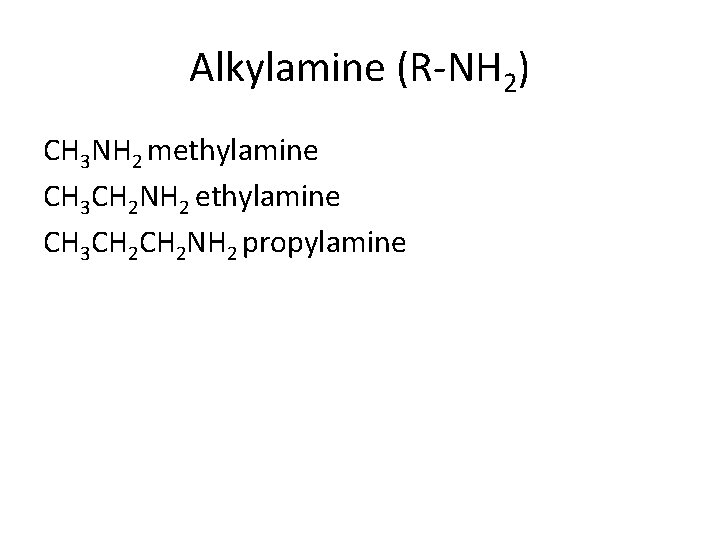
Alkylamine (R-NH 2) CH 3 NH 2 methylamine CH 3 CH 2 NH 2 propylamine
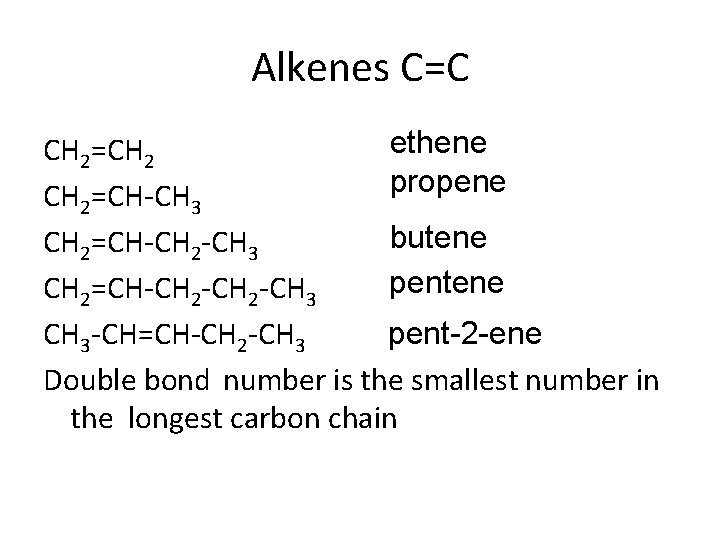
Alkenes C=C ethene CH 2=CH 2 propene CH 2=CH-CH 3 butene CH 2=CH-CH 2 -CH 3 pentene CH 2=CH-CH 2 -CH 3 -CH=CH-CH 2 -CH 3 pent-2 -ene Double bond number is the smallest number in the longest carbon chain
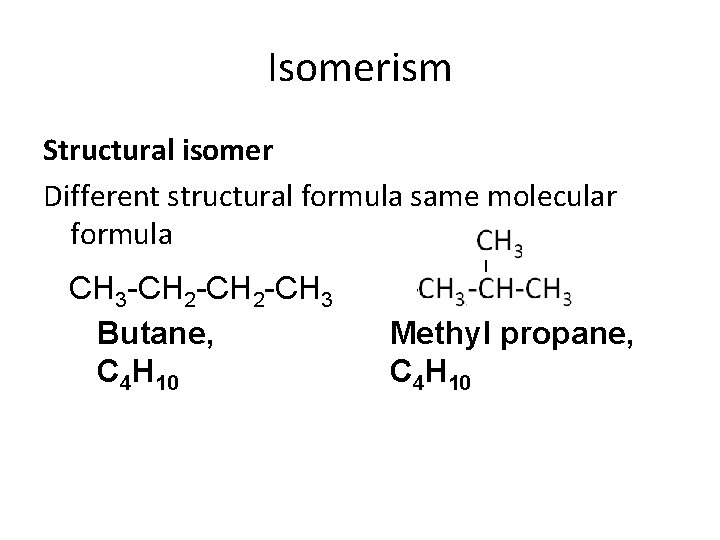
Isomerism Structural isomer Different structural formula same molecular formula CH 3 -CH 2 -CH 3 Butane, C 4 H 10 Methyl propane, C 4 H 10
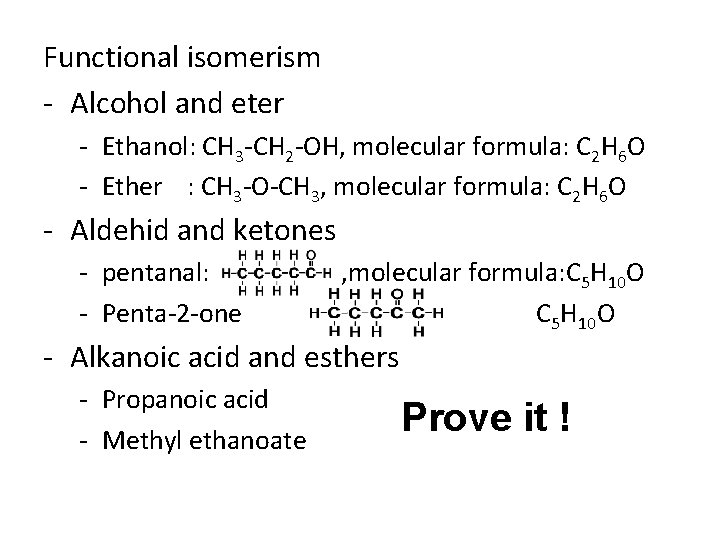
Functional isomerism - Alcohol and eter - Ethanol: CH 3 -CH 2 -OH, molecular formula: C 2 H 6 O - Ether : CH 3 -O-CH 3, molecular formula: C 2 H 6 O - Aldehid and ketones - pentanal: - Penta-2 -one , molecular formula: C 5 H 10 O - Alkanoic acid and esthers - Propanoic acid - Methyl ethanoate Prove it !
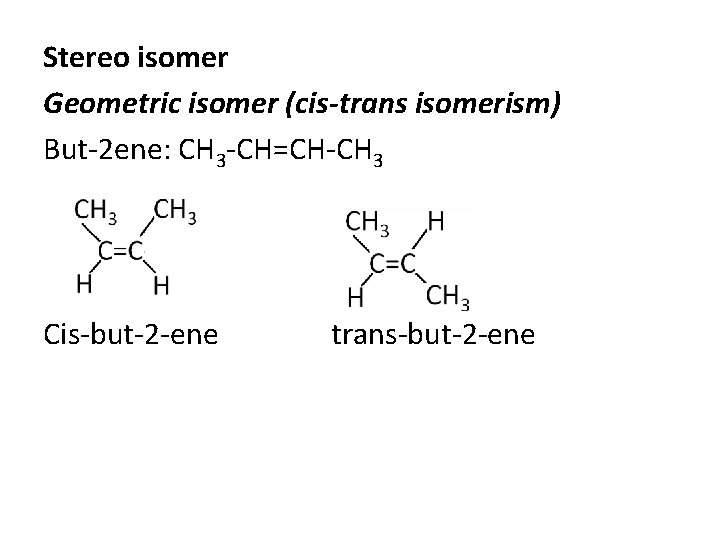
Stereo isomer Geometric isomer (cis-trans isomerism) But-2 ene: CH 3 -CH=CH-CH 3 Cis-but-2 -ene trans-but-2 -ene
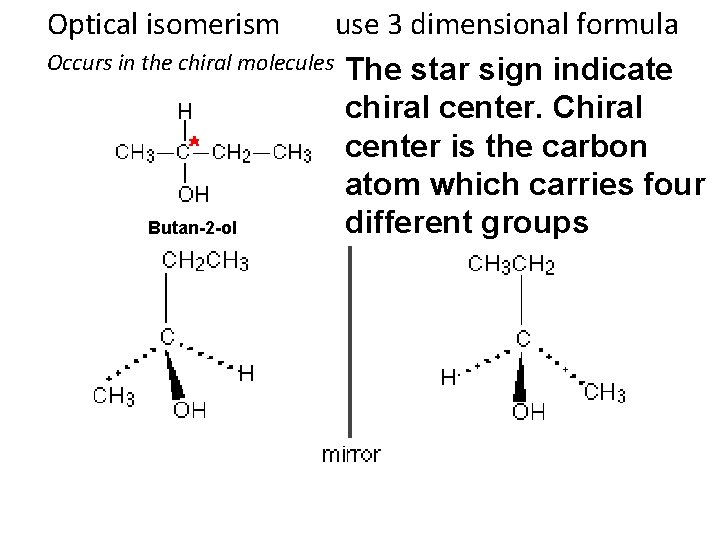
Optical isomerism use 3 dimensional formula Occurs in the chiral molecules The star sign indicate chiral center. Chiral center is the carbon atom which carries four different groups Butan-2 -ol

Reactions Routes Write the reactions routes to convert: a. Propene to propanol – pass propene and steam over heated phosphoric acid catalyst under pressure b. Propene to propanal – pass propene and steam over heated phosphoric acid catalyst under pressure untill propanol forms – Distill on propanol to acidified potassium dichromate
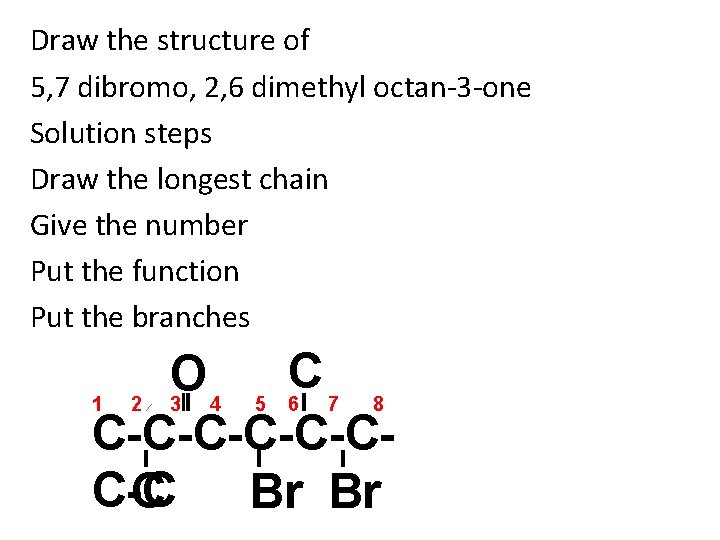
Draw the structure of 5, 7 dibromo, 2, 6 dimethyl octan-3 -one Solution steps Draw the longest chain Give the number Put the function Put the branches C O 1 2 3 4 5 6 7 8 C-C-C-CC-C C Br Br
- Slides: 36