Organic Chemistry Molecular Orbitals and Hybridisation Molecular orbitals

Organic Chemistry Molecular Orbitals and Hybridisation

Molecular orbitals Orbitals can be used to explain bonding between atoms. Atomic orbitals are the volume of space that the electrons of an atom are likely to be found in. H H 1 s atomic orbitals of hydrogen The atomic orbitals containing the valence electrons (outer electrons) are the ones that are important to us.
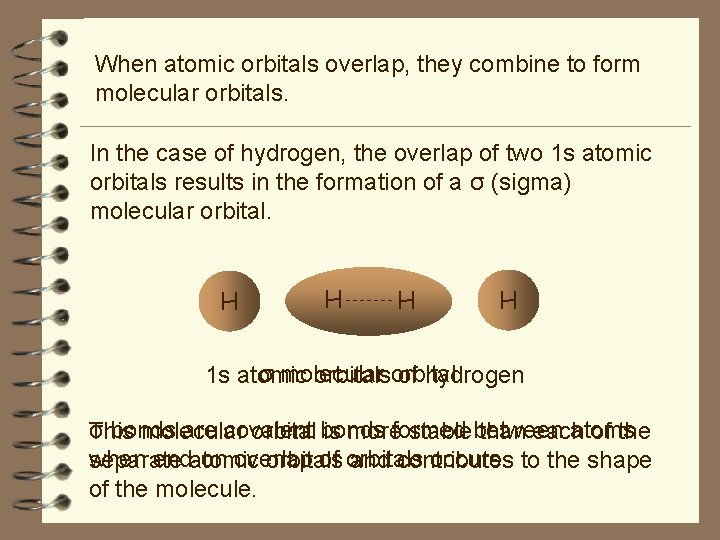
When atomic orbitals overlap, they combine to form molecular orbitals. In the case of hydrogen, the overlap of two 1 s atomic orbitals results in the formation of a σ (sigma) molecular orbital. H H σ molecular 1 s atomic orbitalsorbital of hydrogen σ bonds are covalent atoms This molecular orbital bonds is moreformed stablebetween than each of the when end-on overlap of orbitals occurs. to the shape separate atomic orbitals and contributes of the molecule.
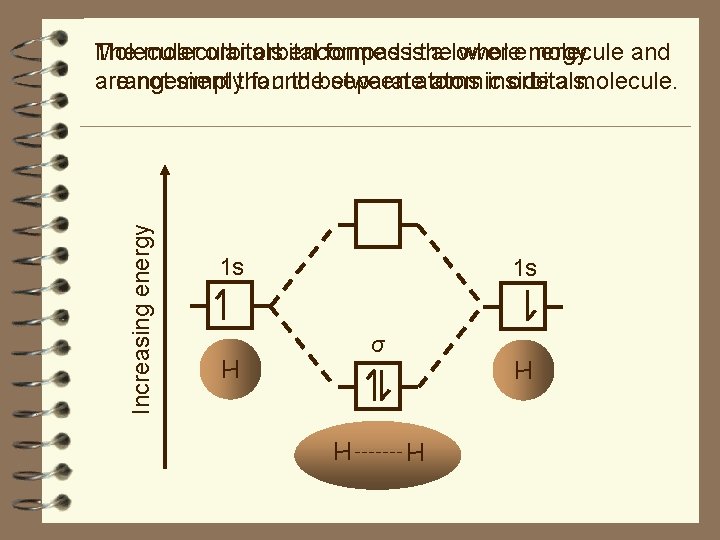
Increasing energy Molecular The molecular orbitals orbital encompass formed isthe a lower wholeenergy molecule and arrangement are not simplythan found thebetween separateatoms atomic inside orbitals. a molecule. 1 s 1 s σ H H

Bonding continuum The shape of the molecular orbital formed from overlapping atomic orbitals will govern the type of intermolecular bonding that is observed.

Non-polar (pure) covalent bonds Non-polar covalent bonds or pure covalent bonds are formed between two atoms of the same element, or two atoms with a very low difference in electronegativity. The molecular orbital formed from overlapping atomic orbitals is symmetrical around a mid-point where the bonding electrons are most likely to be found.

Non-polar (pure) covalent bonds Example: fluorine The overlap of two 2 p orbitals results in the formation of a σ orbital. F + F 2 p atomic orbital σ molecular orbital
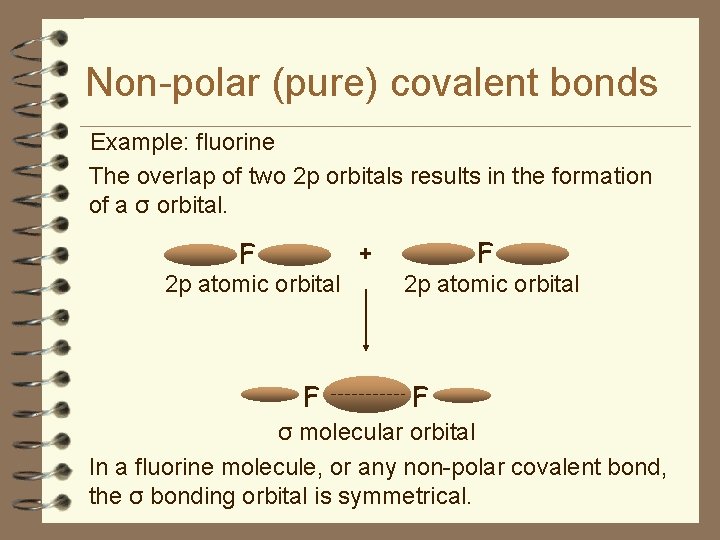
Non-polar (pure) covalent bonds Example: fluorine The overlap of two 2 p orbitals results in the formation of a σ orbital. F 2 p atomic orbital F + F 2 p atomic orbital F σ molecular orbital In a fluorine molecule, or any non-polar covalent bond, the σ bonding orbital is symmetrical.

Polar covalent bonds Since oxygen more difference electronegative thanthe hydrogen, When there is is a large between the molecular orbital be asymmetrical, with electronegativities of formed the two will elements involved in the bonding electrons more likely to will be found around bond, the bonding molecular orbital be the δ– oxygen atom. asymmetrical. Example: water δ– δ+ H O δ+ H

Ionic bonds When ionic bonds form, there is extreme asymmetry and the bonding molecular orbital is almost entirely around one atom.
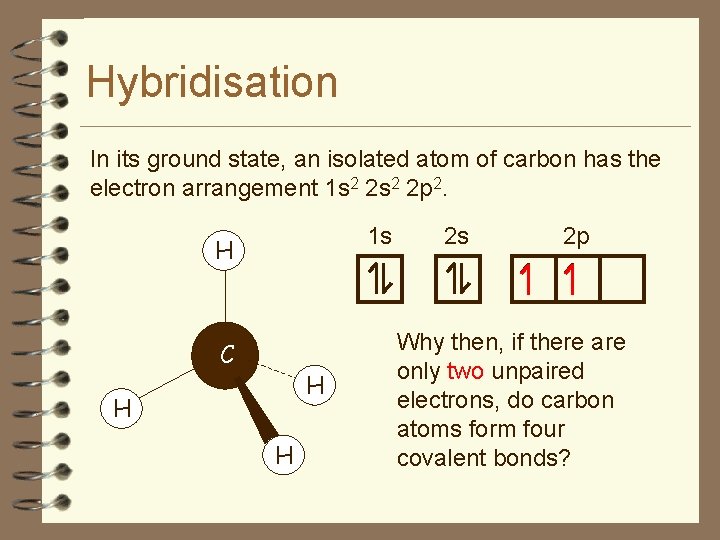
Hybridisation In its ground state, an isolated atom of carbon has the electron arrangement 1 s 2 2 p 2. 1 s H C H H H 2 s 2 p Why then, if there are only two unpaired electrons, do carbon atoms form four covalent bonds?
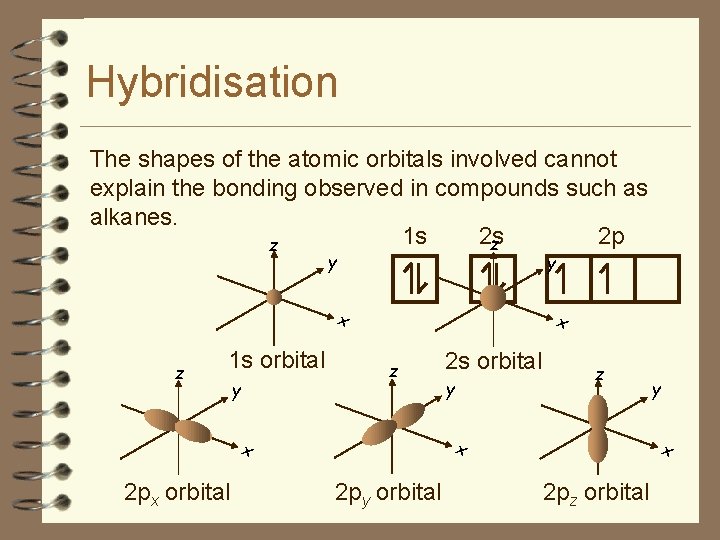
Hybridisation The shapes of the atomic orbitals involved cannot explain the bonding observed in compounds such as alkanes. 1 s 2 s 2 p z z y y x z 1 s orbital y x z y z x x 2 px orbital 2 s orbital 2 py orbital y x 2 pz orbital
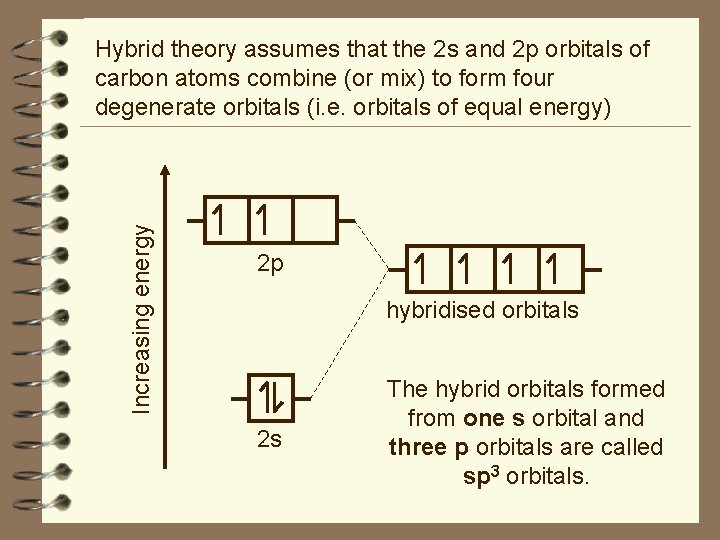
Increasing energy Hybrid theory assumes that the 2 s and 2 p orbitals of carbon atoms combine (or mix) to form four degenerate orbitals (i. e. orbitals of equal energy) 2 p hybridised orbitals 2 s The hybrid orbitals formed from one s orbital and three p orbitals are called sp 3 orbitals.

an sp 3 hybridised orbital The sp 3 orbitals formed are all half-filled, with the electron far more likely to be found in the larger lobe. Since electrons repel each other, the four sp 3 hybridised orbitals surrounding a central carbon atom result in a familiar tetrahedral shape, with a maximum possible angle between each orbital of 109. 5°.

Alkanes In methane, all four hybrid orbitals are used to form σ bonds between the central carbon atom and hydrogen atoms. H C H H H

Alkanes Carbon-to-carbon single bonds in alkanes resultoverlap from σ bonds are covalent bonds formed by end-on 3 orbitals forming σ bonds. overlapping sporbitals of two atomic and since bonds must lie along the line joining both atoms, there will be free rotation around these orbitals. H H C C H H σ bond H H
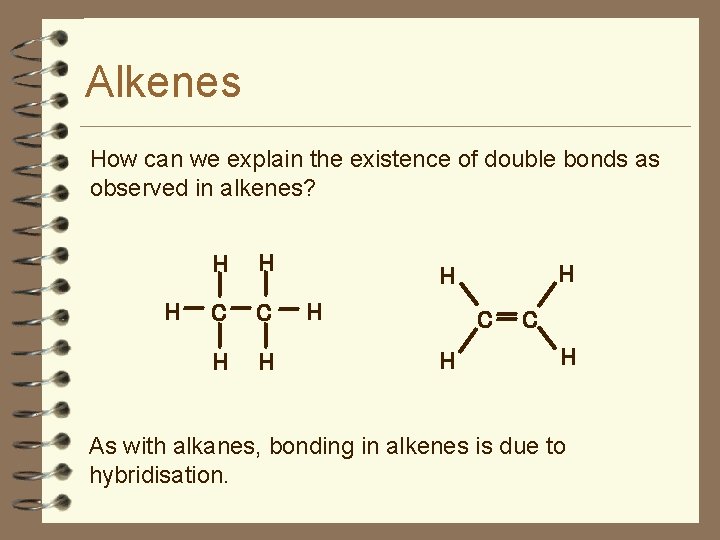
Alkenes How can we explain the existence of double bonds as observed in alkenes? H H H C C H H H C H As with alkanes, bonding in alkenes is due to hybridisation.
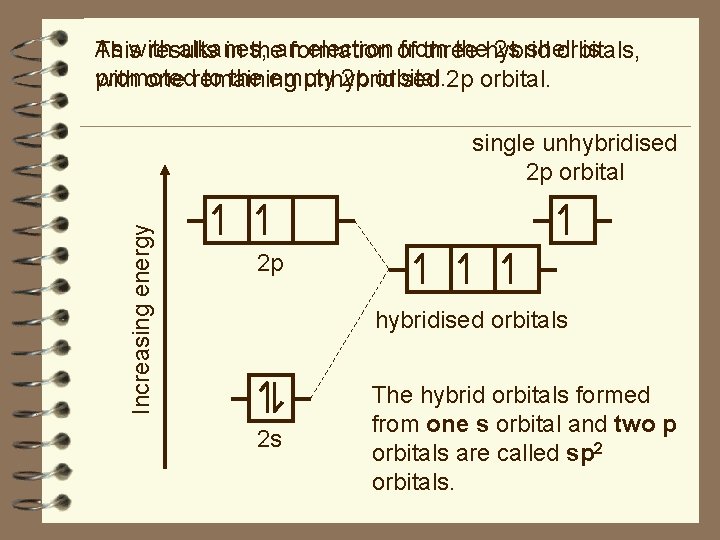
As alkanes, electron of from thehybrid 2 s shell is Thiswith results in theanformation three orbitals, promoted to the empty 2 p orbital. with one remaining unhybridised Increasing energy single unhybridised 2 p orbital 2 p hybridised orbitals 2 s The hybrid orbitals formed from one s orbital and two p orbitals are called sp 2 orbitals.
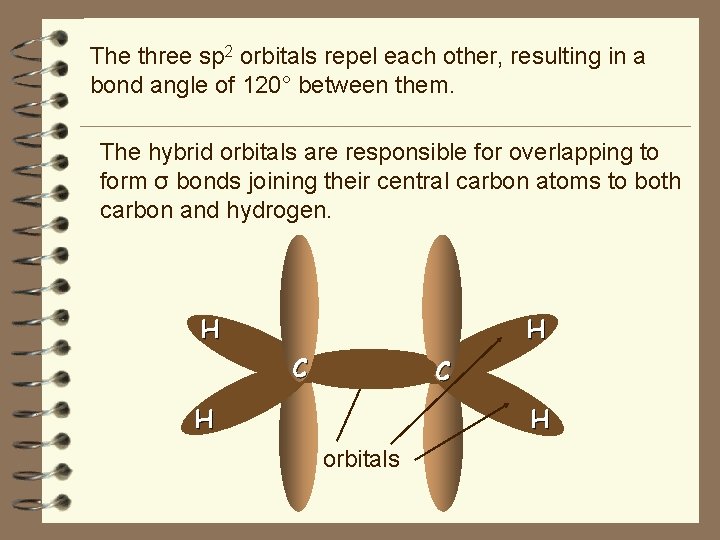
The three sp 2 orbitals repel each other, resulting in a bond angle of 120° between them. The hybrid orbitals are responsible for overlapping to form σ bonds joining their central carbon atoms to both carbon and hydrogen. H H C H sp 2 orbitals
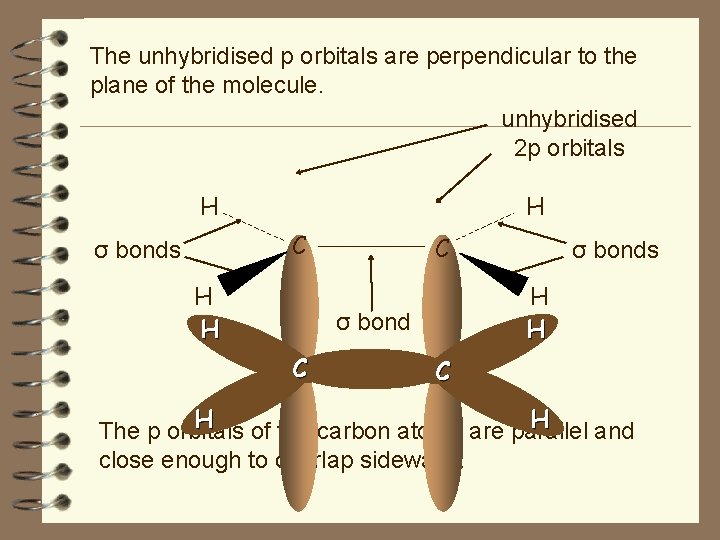
The unhybridised p orbitals are perpendicular to the plane of the molecule. unhybridised 2 p orbitals H H C σ bonds H H C H H σ bond C σ bonds C H H The p orbitals of the carbon atoms are parallel and close enough to overlap sideways.
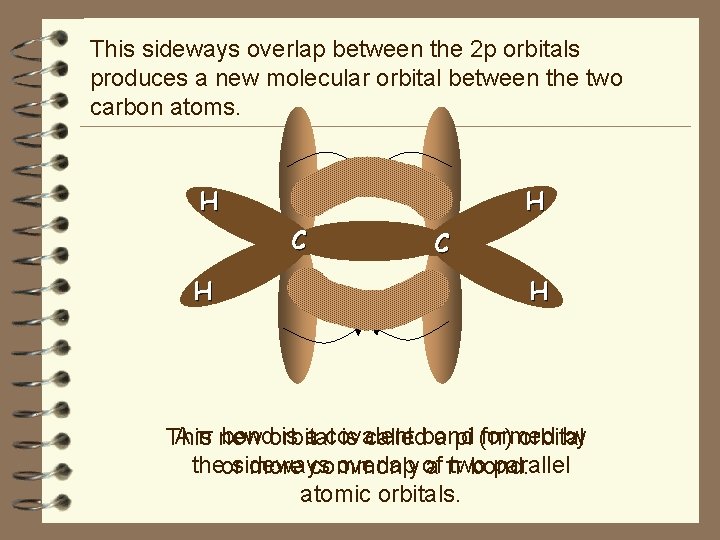
This sideways overlap between the 2 p orbitals produces a new molecular orbital between the two carbon atoms. H H C H A π new bondorbital is a covalent formed by This is calledbond a pi (π) orbital theorsideways overlap of two parallel more commonly aπ bond. atomic orbitals.

H σ and π bonds H C C H H Looking at information comparing σ and π bonds, we can see that double bonds are stronger than single bonds, but not twice as strong. This is because the sideways overlap (π bond) is weaker than the end-on overlap (σ bond). Bonding orbitals present Bond length Mean bond enthalpy C 1σ 154 pm 370 k. J mol– 1 C C 1σ+1π 134 pm 602 k. J mol– 1 C C 1σ+2π 121 pm 835 k. J mol– 1 Bond type C

Aromatic hydrocarbons Aromatic compounds differ to other hydrocarbons as they contain delocalised electrons. Example: benzene (C 6 H 6) Chemists initially represented H a molecule of benzene as H H C shown here. C C H C H H However, contrary to what might be expected from this structure, benzene is a very stable, saturated structure that does not undergo addition reactions.
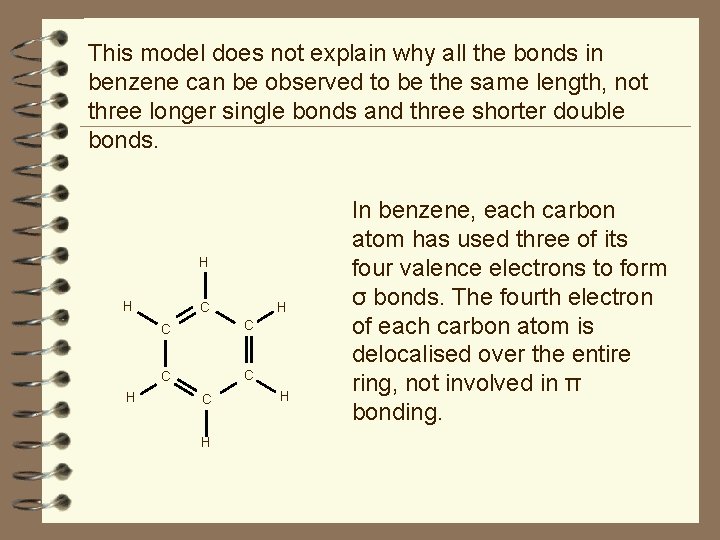
This model does not explain why all the bonds in benzene can be observed to be the same length, not three longer single bonds and three shorter double bonds. H H C C C H H In benzene, each carbon atom has used three of its four valence electrons to form σ bonds. The fourth electron of each carbon atom is delocalised over the entire ring, not involved in π bonding.

The σ bonding can be described as existing between six sp 2 hybridised orbitals. There are six C–C σ bonds in the molecule and so each carbon atom has two σ bonds to adjacent carbon atoms. H H C C C C H H H Every carbon atom also has a σ bond to a hydrogen.
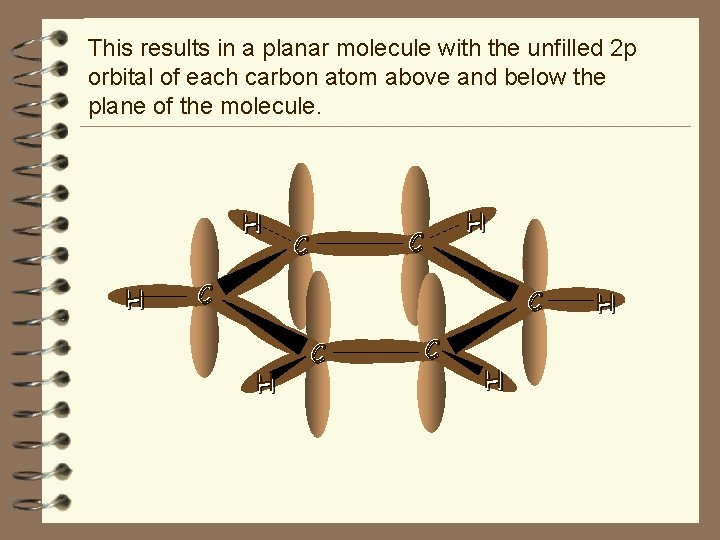
This results in a planar molecule with the unfilled 2 p orbital of each carbon atom above and below the plane of the molecule. H H H C C H H
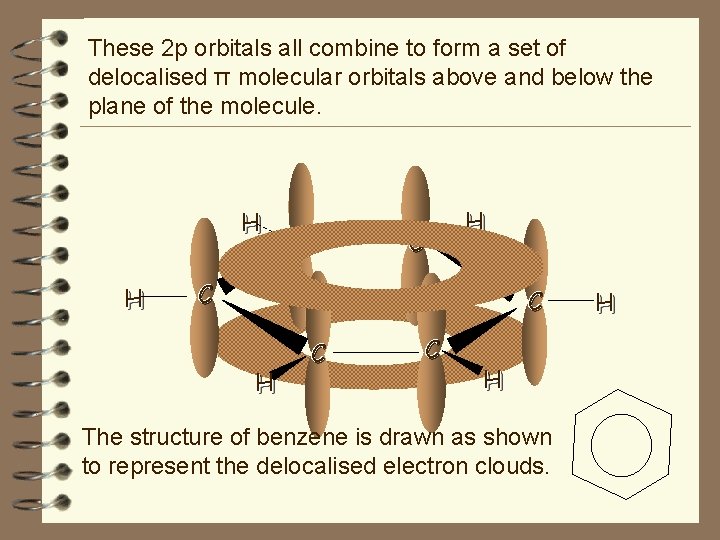
These 2 p orbitals all combine to form a set of delocalised π molecular orbitals above and below the plane of the molecule. H H H C C H The structure of benzene is drawn as shown to represent the delocalised electron clouds. H
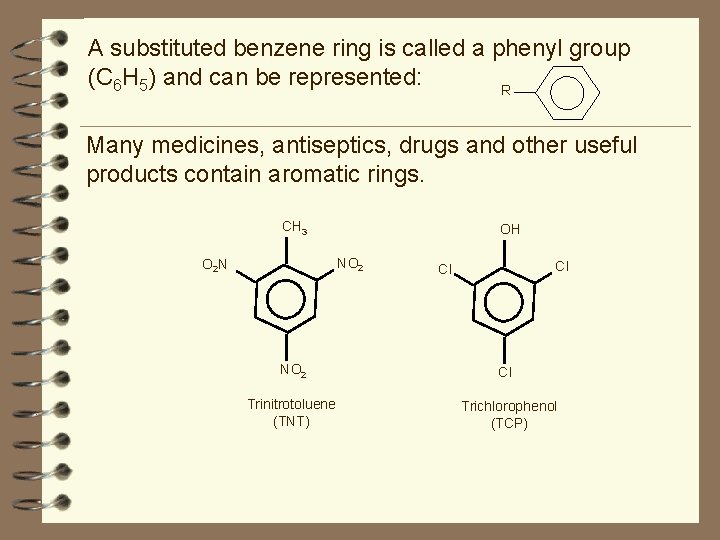
A substituted benzene ring is called a phenyl group (C 6 H 5) and can be represented: R Many medicines, antiseptics, drugs and other useful products contain aromatic rings. CH 3 OH NO 2 O 2 N Cl Cl NO 2 Cl Trinitrotoluene (TNT) Trichlorophenol (TCP)
- Slides: 28