Hydrocarbons Naming Alkanes Dr Seemal Jelani Chem160 652021

Hydrocarbons Naming Alkanes Dr Seemal Jelani Chem-160 6/5/2021 1
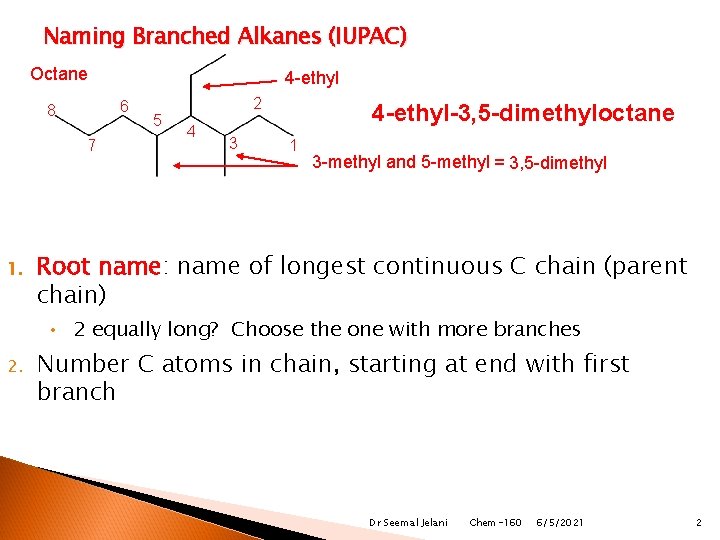
Naming Branched Alkanes (IUPAC) Octane 6 8 7 1. 4 -ethyl 5 2 4 3 4 -ethyl-3, 5 -dimethyloctane 1 3 -methyl and 5 -methyl = 3, 5 -dimethyl Root name: name of longest continuous C chain (parent chain) • 2 equally long? Choose the one with more branches 2. Number C atoms in chain, starting at end with first branch Dr Seemal Jelani Chem-160 6/5/2021 2

1. Identify substituents, give each a number (C it is connected to) • Two or more identical substituents: use prefixes (di-, tri, tetra-, etc. ) 2. List substituents alphabetically before root name • Do not alphabetize prefixes 3. Punctuation: commas separate numbers from each other hyphens separate numbers from names no space between last substituent & root name Dr Seemal Jelani Chem-160 6/5/2021 3
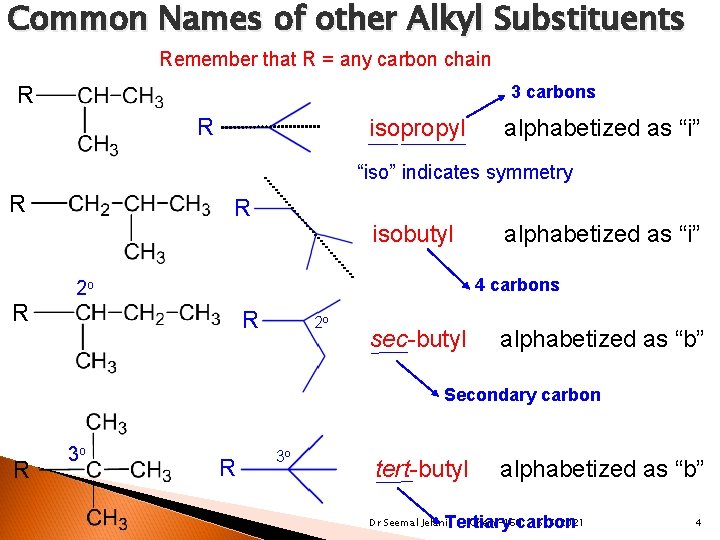
Common Names of other Alkyl Substituents Remember that R = any carbon chain 3 carbons R R isopropyl alphabetized as “i” “iso” indicates symmetry R R R isobutyl alphabetized as “i” 4 carbons 2 o R 2 o sec-butyl alphabetized as “b” Secondary carbon R 3 o tert-butyl alphabetized as “b” Chem-160 6/5/2021 Tertiary carbon Dr Seemal Jelani 4
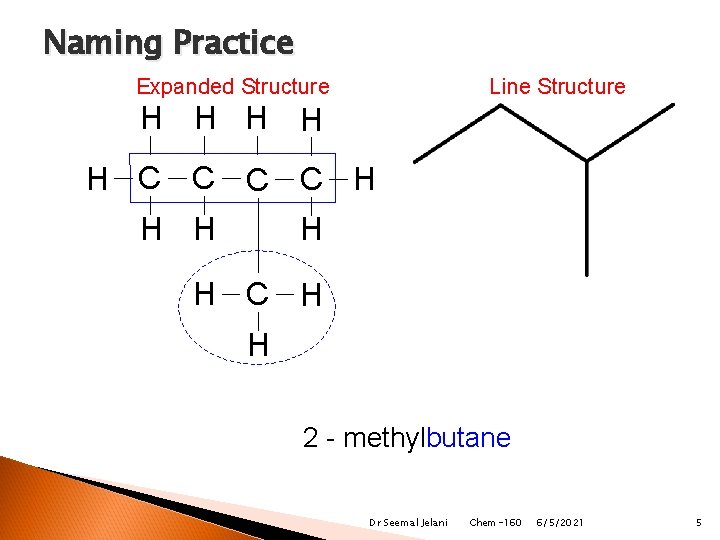
Naming Practice Line Structure Expanded Structure H H H C C H H H C H H butane 2 - methylbutane Dr Seemal Jelani Chem-160 6/5/2021 5
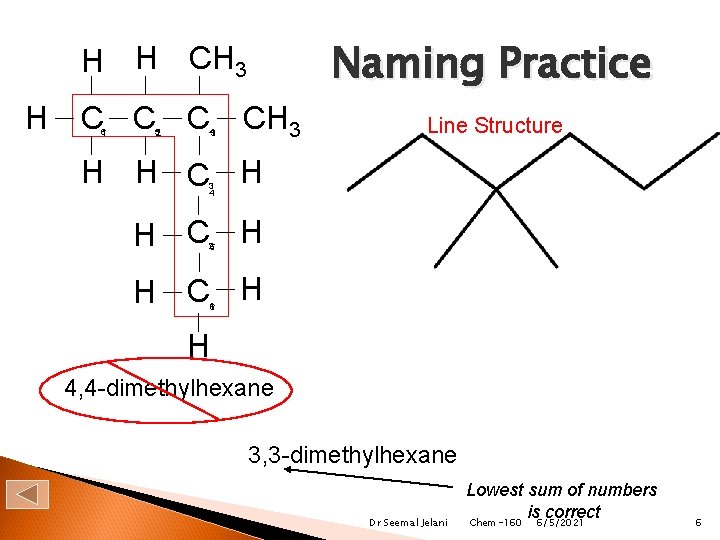
Naming Practice H H CH 3 H C C C 6 1 52 43 CH 3 Line Structure H H C H 3 4 H C H 25 H C H 16 H 4, 4 -dimethylhexane 3, 3 -dimethylhexane Dr Seemal Jelani Lowest sum of numbers is correct Chem-160 6/5/2021 6

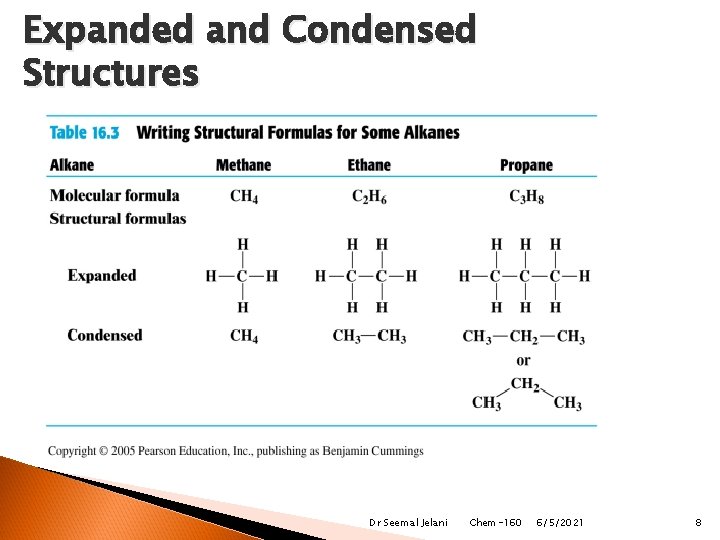
Structural Formulas Alkanes are written with structural formulas that are expanded to show each bond (Lewis Structure). condensed to show each carbon atom and hydrogen atoms attached to that carbon. Expanded (Lewis) H H C Condensed H CH 4 , methane H Dr Seemal Jelani Chem-160 6/5/2021 7
Expanded and Condensed Structures Dr Seemal Jelani Chem-160 6/5/2021 8

Structural Formulas Condensed formulas are written for expanded structural formula by showing each carbon and the attached hydrogen atoms. Expanded Condensed H H │ │ H─C ─C ─ H │ │ H H CH 3─CH 2─CH 3 Dr Seemal Jelani Chem-160 6/5/2021 9

Names of Alkanes The names of alkanes are determined by the IUPAC (International Union of Pure and Applied Chemistry) system. end in –ane. with 1 -4 carbons in a chain use prefixes as follows. Name # Carbons Structural Formula Methane 1 CH 4 Ethane 2 CH 3 Propane 3 CH 3 CH 2 CH 3 Butane 4 CH 3 CH 2 CH 3 Dr Seemal Jelani Chem-160 6/5/2021 10

Names of Alkanes with 5 -10 carbon atoms in a chain use Greek prefixes. Name # Carbons Structural Formula Pentane 5 CH 3 CH 2 CH 2 CH 3 Hexane 6 CH 3 CH 2 CH 2 CH 3 Heptane 7 CH 3 CH 2 CH 2 CH 2 CH 3 Octane 8 CH 3 CH 2 CH 2 CH 2 CH 3 Nonane 9 CH 3 CH 2 CH 2 CH 3 Decane 10 CH 3 CH 2 CH 2 CH 3 Know the 1 st eight alkanes (name and structure) Dr Seemal Jelani Chem-160 6/5/2021 11
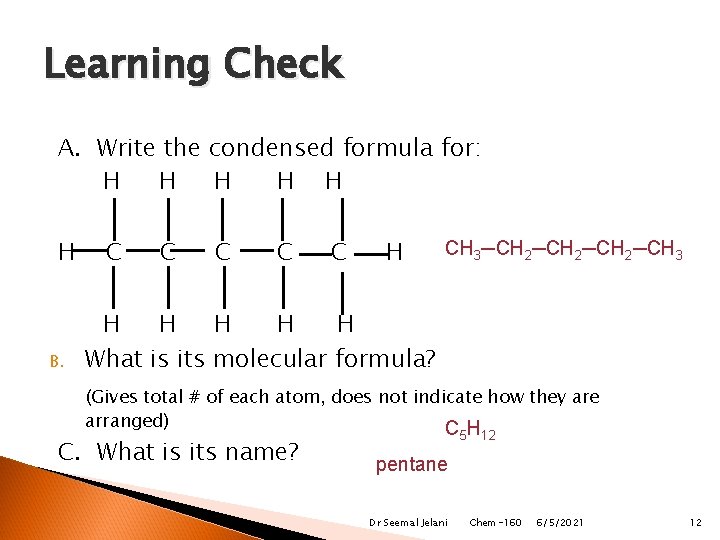
Learning Check A. Write the condensed formula for: H H H B. C C C H CH 3─CH 2─CH 3 H H H What is its molecular formula? (Gives total # of each atom, does not indicate how they are arranged) CH C. What is its name? 5 12 pentane Dr Seemal Jelani Chem-160 6/5/2021 12

Some Structures for Butane Dr Seemal Jelani Chem-160 6/5/2021 13

Hexane has Six Carbon Atoms Hexane is an alkane with six carbon atoms in a continuous chain. has a “zig-zag” look because each carbon atom is at the center of a tetrahedron. is represented by a ball-and-stick model as shown below. Dr Seemal Jelani Chem-160 6/5/2021 14

Cycloalkanes They are cyclic have two hydrogen atoms fewer than the open chain. (remember each carbon has 4 bonds) are named by using the prefix cyclo- before the name of the alkane chain with the same number of carbon atoms. Dr Seemal Jelani Chem-160 6/5/2021 15
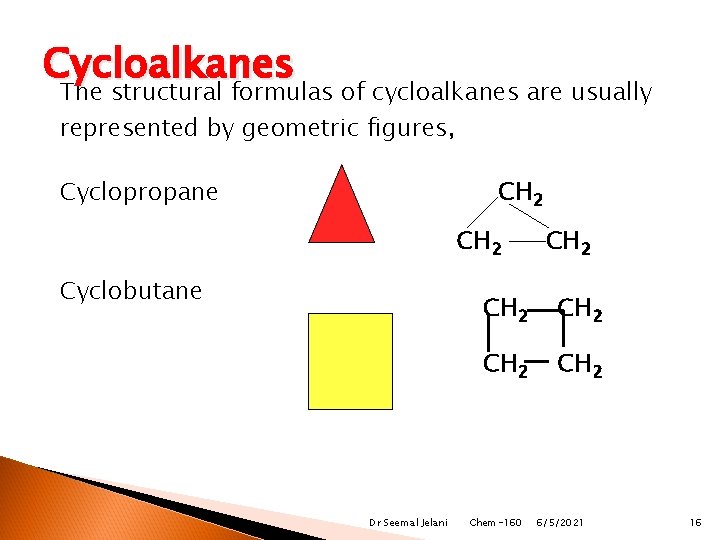
Cycloalkanes The structural formulas of cycloalkanes are usually represented by geometric figures, Cyclopropane CH 2 Cyclobutane Dr Seemal Jelani CH 2 CH 2 Chem-160 6/5/2021 16
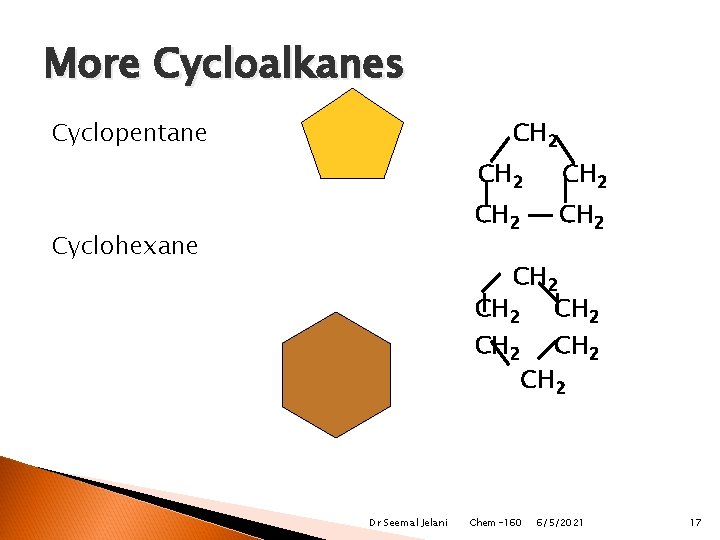
More Cycloalkanes Cyclopentane CH 2 Cyclohexane CH 2 CH 2 Dr Seemal Jelani CH 2 Chem-160 6/5/2021 17
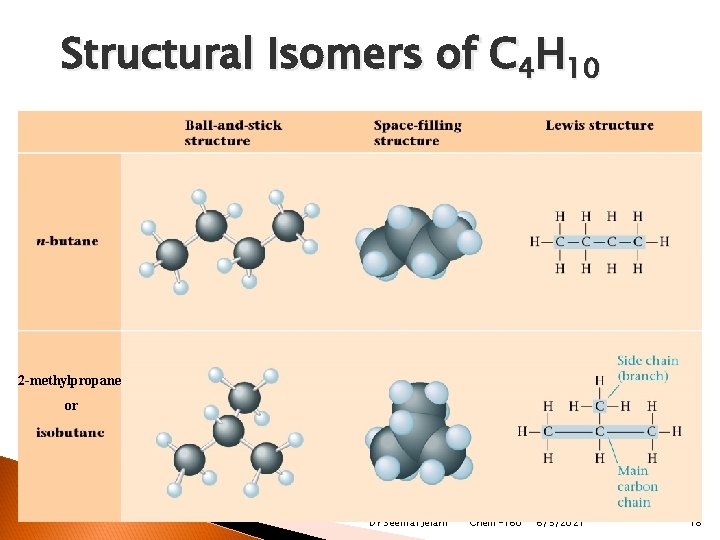
Structural Isomers of C 4 H 10 2 -methylpropane or Dr Seemal Jelani Chem-160 6/5/2021 18

Structural Isomer Practice On piece of your own paper, draw AND name ALL of the isomers for the following alkanes: Formulas isomers Pentane Hexane Heptane C 5 H 12 C 6 H 14 C 7 H 16 3 5 9 Dr Seemal Jelani Chem-160 6/5/2021 19
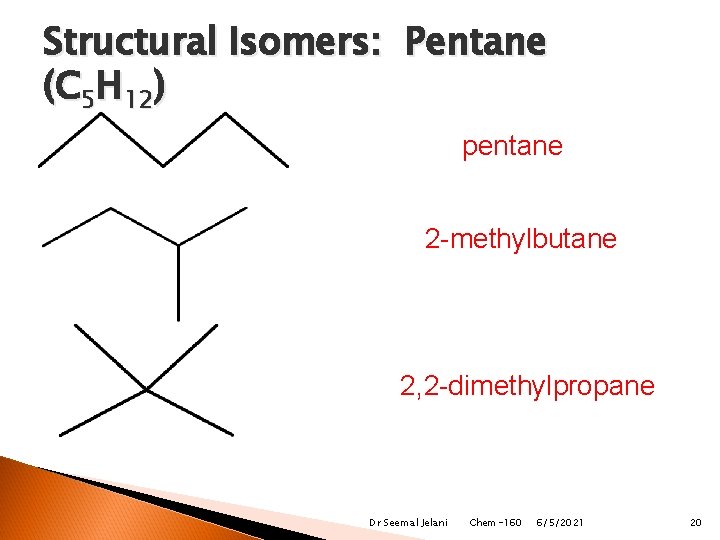
Structural Isomers: Pentane (C 5 H 12) pentane 2 -methylbutane 2, 2 -dimethylpropane Dr Seemal Jelani Chem-160 6/5/2021 20
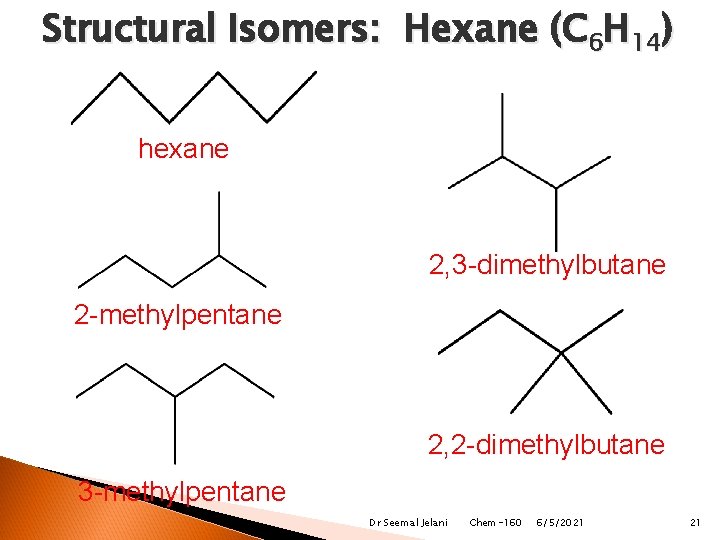
Structural Isomers: Hexane (C 6 H 14) hexane 2, 3 -dimethylbutane 2 -methylpentane 2, 2 -dimethylbutane 3 -methylpentane Dr Seemal Jelani Chem-160 6/5/2021 21
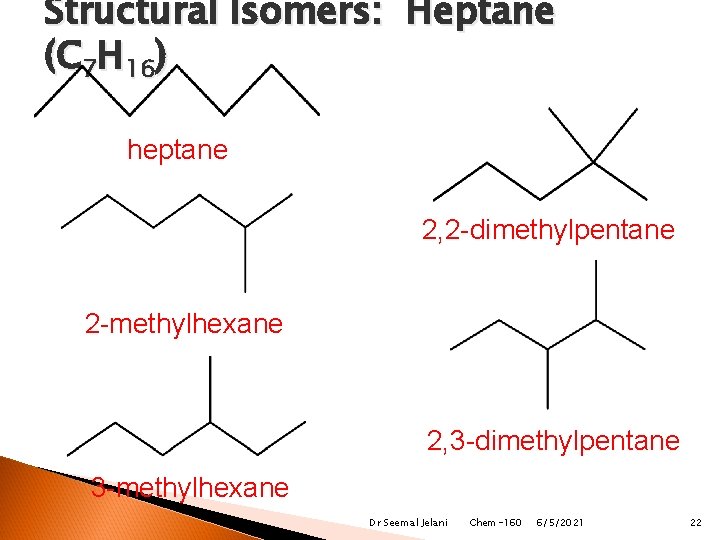
Structural Isomers: Heptane (C 7 H 16) heptane 2, 2 -dimethylpentane 2 -methylhexane 2, 3 -dimethylpentane 3 -methylhexane Dr Seemal Jelani Chem-160 6/5/2021 22
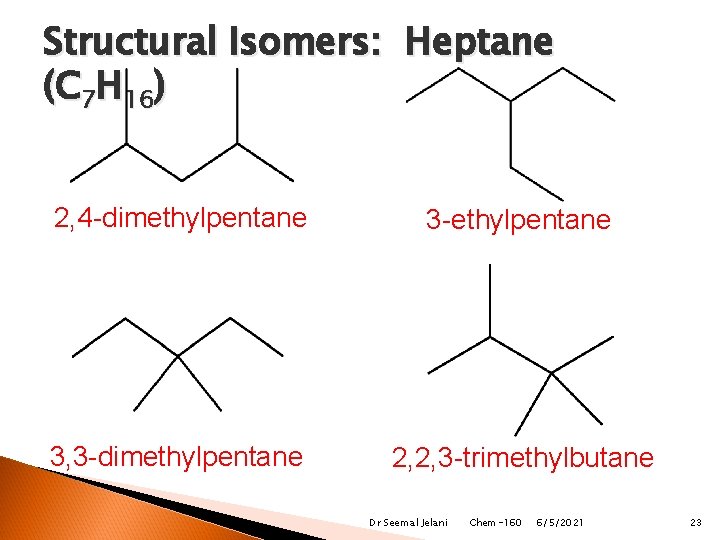
Structural Isomers: Heptane (C 7 H 16) 2, 4 -dimethylpentane 3 -ethylpentane 3, 3 -dimethylpentane 2, 2, 3 -trimethylbutane Dr Seemal Jelani Chem-160 6/5/2021 23
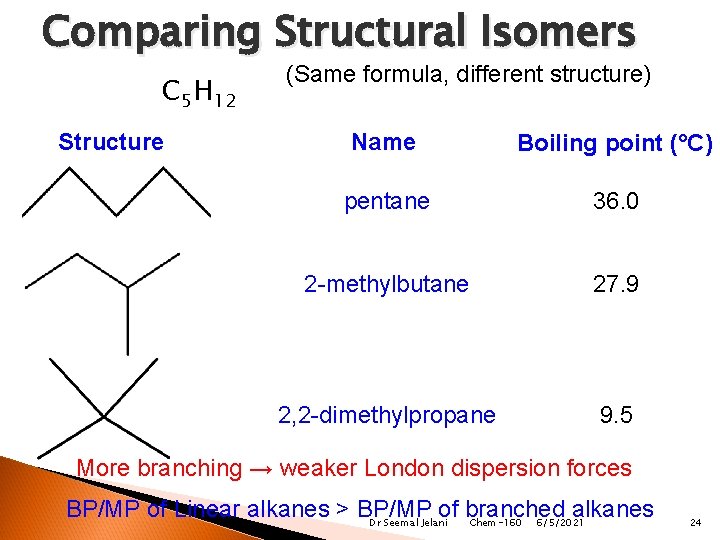
Comparing Structural Isomers C 5 H 12 Structure (Same formula, different structure) Name Boiling point (°C) pentane 36. 0 2 -methylbutane 27. 9 2, 2 -dimethylpropane 9. 5 More branching → weaker London dispersion forces BP/MP of Linear alkanes > BP/MP of branched alkanes Dr Seemal Jelani Chem-160 6/5/2021 24

Structural Isomer Practice Formulas C 4 H 9 I C 3 H 6 Cl 2 C 5 H 11 Br C 4 H 8 Cl 2 # isomers 4 4 8 9 Dr Seemal Jelani Chem-160 6/5/2021 25
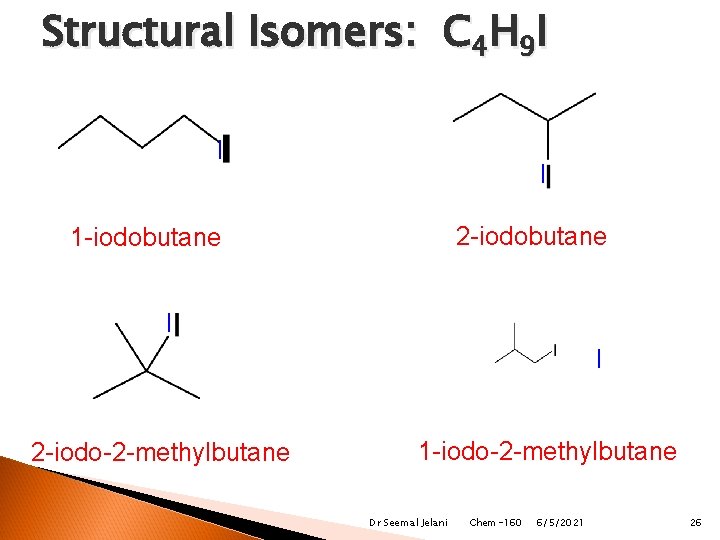
Structural Isomers: C 4 H 9 I I I 2 -iodobutane 1 -iodobutane I I 2 -iodo-2 -methylbutane 1 -iodo-2 -methylbutane Dr Seemal Jelani Chem-160 6/5/2021 26
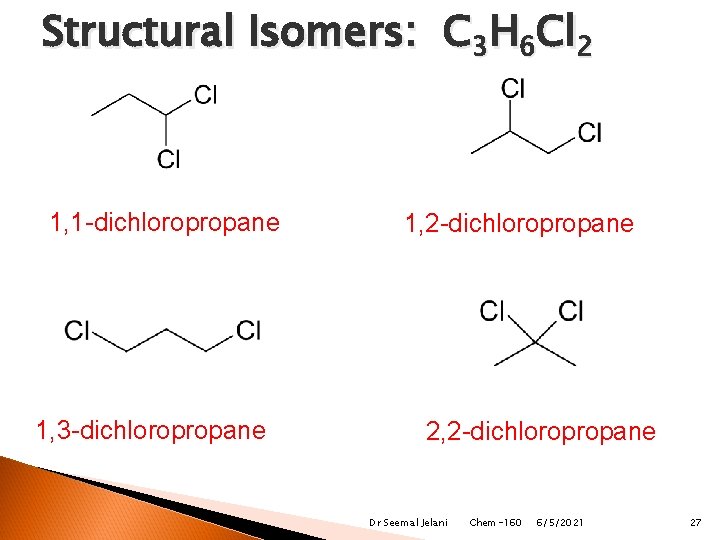
Structural Isomers: C 3 H 6 Cl 2 1, 1 -dichloropropane 1, 3 -dichloropropane 1, 2 -dichloropropane 2, 2 -dichloropropane Dr Seemal Jelani Chem-160 6/5/2021 27
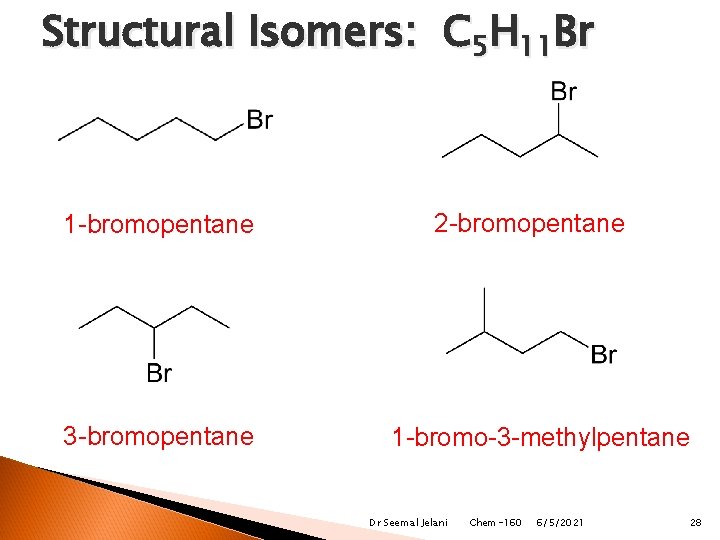
Structural Isomers: C 5 H 11 Br 1 -bromopentane 3 -bromopentane 2 -bromopentane 1 -bromo-3 -methylpentane Dr Seemal Jelani Chem-160 6/5/2021 28
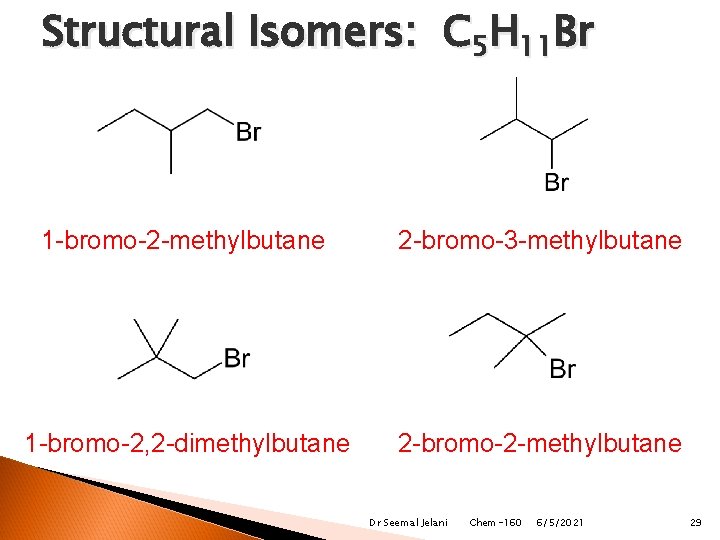
Structural Isomers: C 5 H 11 Br 1 -bromo-2 -methylbutane 2 -bromo-3 -methylbutane 1 -bromo-2, 2 -dimethylbutane 2 -bromo-2 -methylbutane Dr Seemal Jelani Chem-160 6/5/2021 29
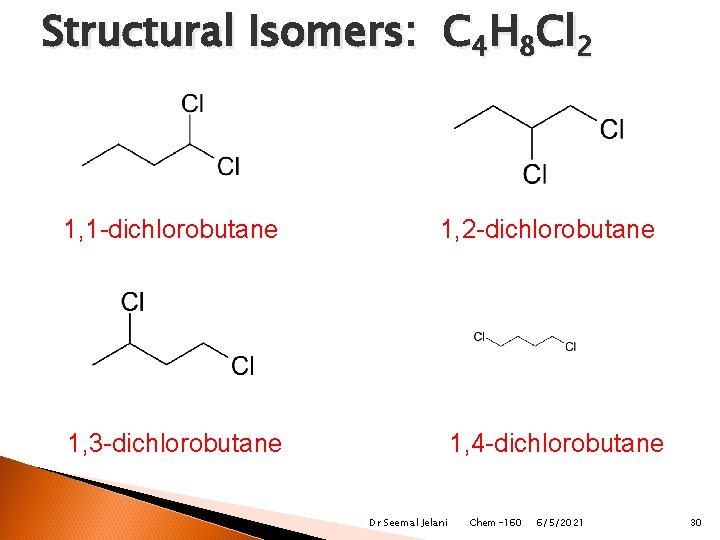
Structural Isomers: C 4 H 8 Cl 2 1, 1 -dichlorobutane 1, 2 -dichlorobutane 1, 4 -dichlorobutane 1, 3 -dichlorobutane Dr Seemal Jelani Chem-160 6/5/2021 30
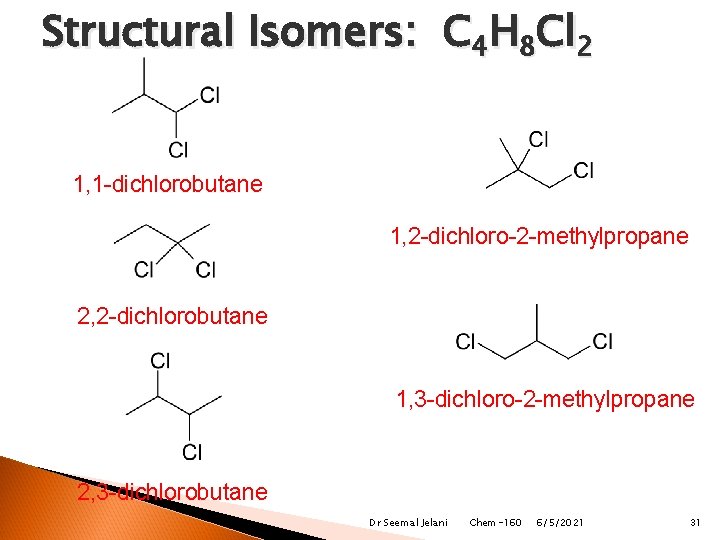
Structural Isomers: C 4 H 8 Cl 2 1, 1 -dichlorobutane 1, 2 -dichloro-2 -methylpropane 2, 2 -dichlorobutane 1, 3 -dichloro-2 -methylpropane 2, 3 -dichlorobutane Dr Seemal Jelani Chem-160 6/5/2021 31

Properties of Alkanes Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings Dr Seemal Jelani Chem-160 6/5/2021 32

Some Properties of Alkanes are nonpolar. insoluble in water. less dense than water. flammable in air. Dr Seemal Jelani Chem-160 6/5/2021 33

Some Properties of Alkanes with 1 -4 carbon atoms are methane, propane, and butane. gases at room temperature. used as heating fuels. Dr Seemal Jelani Chem-160 6/5/2021 34

Some Properties of Alkanes with 5 -8 carbon atoms are liquids at room temperature. pentane, hexane, heptane, and octane. very volatile. used to make gasoline. Alkanes with 9 -17 carbon atoms are liquids at room temperature have higher boiling points. are found in kerosene, diesel, and jet fuels. Dr Seemal Jelani Chem-160 6/5/2021 35

Some Properties of Alkanes with 5 -8 carbon atoms are liquids at room temperature. pentane, hexane, heptane, and octane. very volatile. used to make gasoline. Alkanes with 9 -17 carbon atoms are liquids at room temperature have higher boiling points. are found in kerosene, diesel, and jet fuels. Dr Seemal Jelani Chem-160 6/5/2021 36

Chemical properties Dr Seemal Jelani Chem-160 6/5/2021 37

Reactions of Alkanes Combustion exothermic reaction alkanes used as fuel source 13/ 4 CO 2 + ___ 5 H 2 O 2 ___ C 4 H 10 + ___ Incomplete Combustion with insufficient O 2 produces CO • Poor ventilation, cigarettes 9/ 4 CO + ___ C 4 H 10 + ___ 5 H 2 O 2 ___ CO is poisonous because it binds to the hemoglobin in the blood, preventing the absorption of O 2 Dr Seemal Jelani Chem-160 6/5/2021 38

Combustion In excess oxygen alkanes burn according to the following equation: Cn. H 2 n+2 +(1. 5 n+0. 5)O 2 n. CO 2 +(n+1)H 2 O This is called complete combustion, however if there is insufficient oxygen present incomplete combustion can occur. Cn. H 2 n+2 +(n+0. 5)O 2 n CO +(n+1)H 2 O With even less oxygen soot is formed. Dr Seemal Jelani Chem-160 6/5/2021 39

Combustion In combustion reactions, alkanes react with oxygen. CO 2, H 2 O and energy are produced. Alkane + O 2 H 2 O + heat Dr Seemal Jelani Chem-160 CO 2 + 6/5/2021 40

Radical Halogenation Terms Mechanism ◦ How the reaction occurs through multiple steps (most reactions actually occur in many steps) Chain Reaction ◦ Reactions that occur on their own after some initiating event Free Radicals ◦ Atoms that have one free electron—highly reactive Dr Seemal Jelani Chem-160 6/5/2021 41

Radical Halogenation Terms Initiation Step ◦ Step where a bond is split by heat/light, producing free radicals Propagation Step ◦ Step where free radicals react with non-radicals, producing more free radicals and continuing the “chain reaction” Termination Step ◦ Step where free radicals react with each other, producing non-radicals and terminating the “chain reaction” Dr Seemal Jelani Chem-160 6/5/2021 42
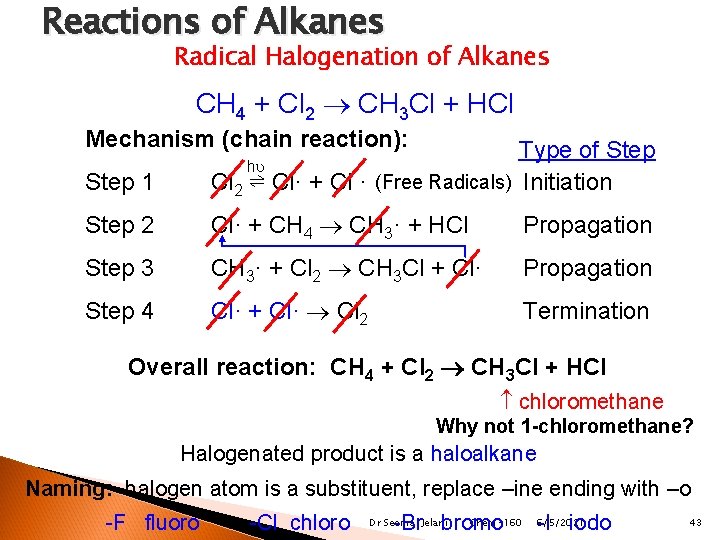
Reactions of Alkanes Radical Halogenation of Alkanes CH 4 + Cl 2 CH 3 Cl + HCl Mechanism (chain reaction): Step 1 Type of Step Cl 2 ⇌ Cl· + Cl · (Free Radicals) Initiation Step 2 Cl· + CH 4 CH 3· + HCl Propagation Step 3 CH 3· + Cl 2 CH 3 Cl + Cl· Propagation Step 4 Cl· + Cl· Cl 2 Termination h Overall reaction: CH 4 + Cl 2 CH 3 Cl + HCl chloromethane Why not 1 -chloromethane? Halogenated product is a haloalkane Naming: halogen atom is a substituent, replace –ine ending with –o -F fluoro -Cl chloro Chem-160 6/5/2021 -Br bromo -I iodo Dr Seemal Jelani 43

Halogen substitutes for hydrogen in alkane →multiple results Cl Cl 2 CH 4 Compound CH 3 Cl CH 2 Cl 2 CHCl 3 CCl 4 2 CH 3 Cl 2 CH 2 Cl 2 IUPAC Name Chloromethane Dichloromethane Trichloromethane Tetrachloromethane 2 CHCl 3 CCl 4 Common Name Methyl chloride Methylene chloride Chloroform Carbon tetrachloride All are liquids at room temperature • Heavy Cl atoms increase LDF • Polar C-Cl bonds – can have polar molecules Dr Seemal Jelani Chem-160 6/5/2021 44

Textbook Resource Chemistry: An Intro to General, Organic and Biological Chemistry by Timberlake (Green/Tan Book) Dr Seemal Jelani Chem-160 6/5/2021 45
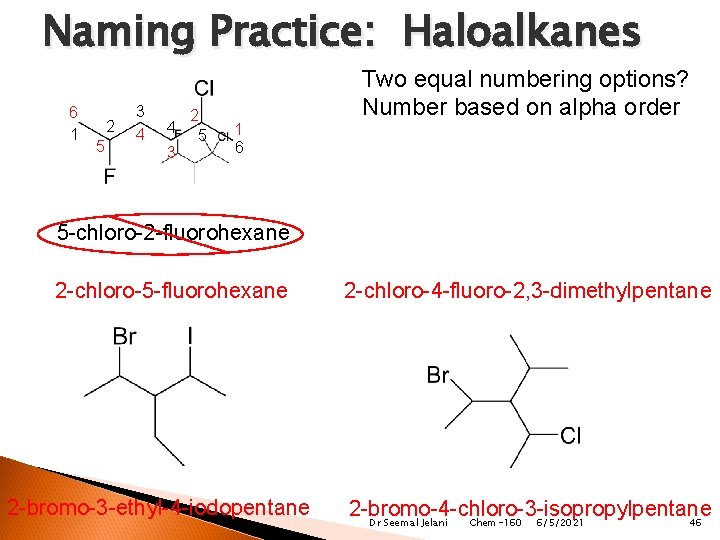
Naming Practice: Haloalkanes 6 1 2 5 3 4 4 3 2 5 1 6 Two equal numbering options? Number based on alpha order 5 -chloro-2 -fluorohexane 2 -chloro-5 -fluorohexane 2 -bromo-3 -ethyl-4 -iodopentane 2 -chloro-4 -fluoro-2, 3 -dimethylpentane 2 -bromo-4 -chloro-3 -isopropylpentane Dr Seemal Jelani Chem-160 6/5/2021 46
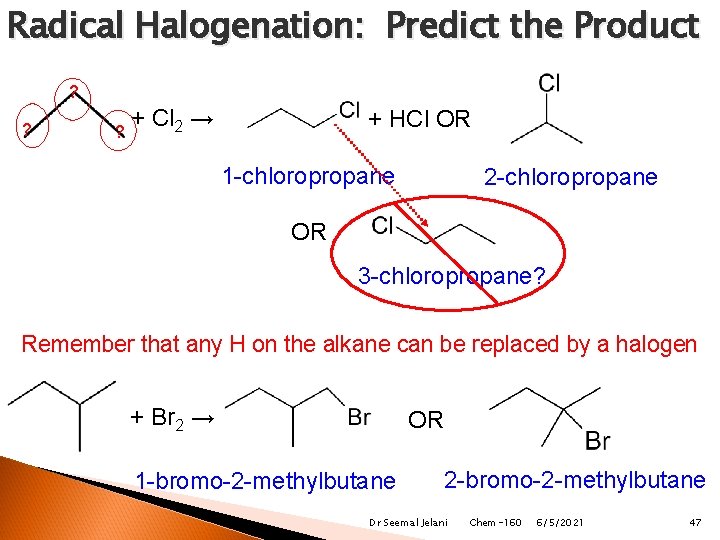
Radical Halogenation: Predict the Product ? ? ? + Cl 2 → + HCl OR 1 -chloropropane 2 -chloropropane OR 3 -chloropropane? Remember that any H on the alkane can be replaced by a halogen + Br 2 → OR 1 -bromo-2 -methylbutane 2 -bromo-2 -methylbutane Dr Seemal Jelani Chem-160 6/5/2021 47
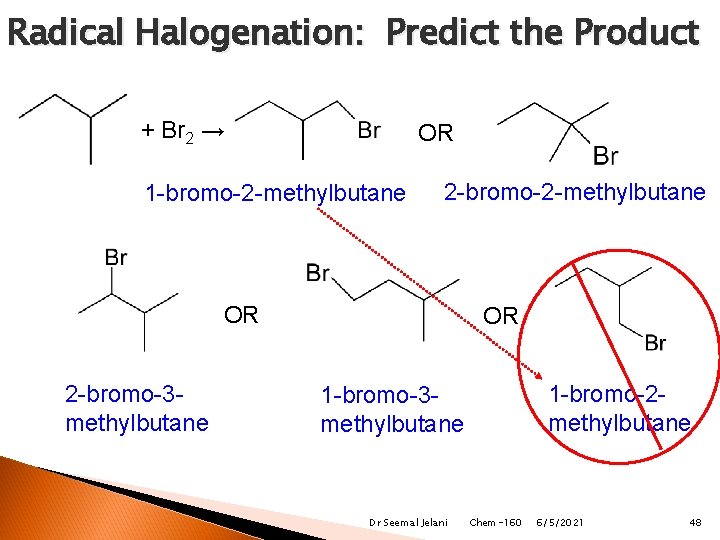
Radical Halogenation: Predict the Product + Br 2 → OR 1 -bromo-2 -methylbutane 2 -bromo-2 -methylbutane OR 2 -bromo-3 methylbutane OR 1 -bromo-2 methylbutane 1 -bromo-3 methylbutane Dr Seemal Jelani Chem-160 6/5/2021 48
- Slides: 48