682021 Chem160 1 Covalent Bonds 682021 Chem160 2

6/8/2021 Chem-160 1

Covalent Bonds 6/8/2021 Chem-160 2

LET’S FIRST REVIEW IONIC BONDING 6/8/2021 Chem-160 3

In an IONIC bond, electrons are lost or gained, resulting in the formation of IONS in ionic compounds. K 6/8/2021 F Chem-160 4
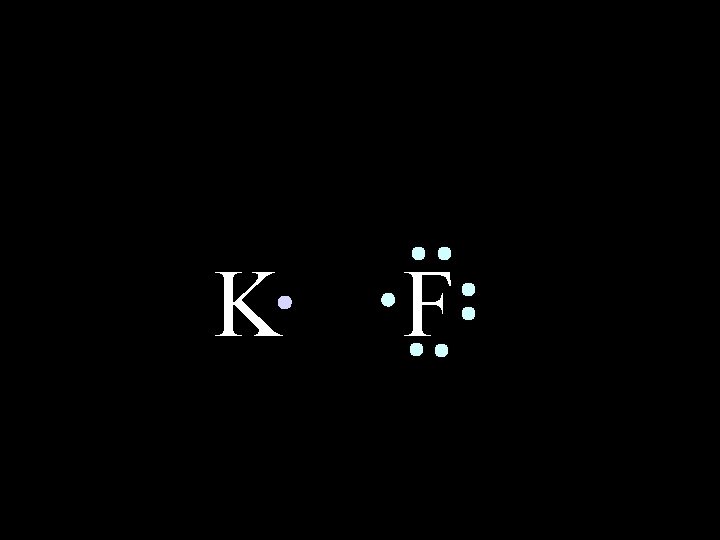
K 6/8/2021 F Chem-160 5
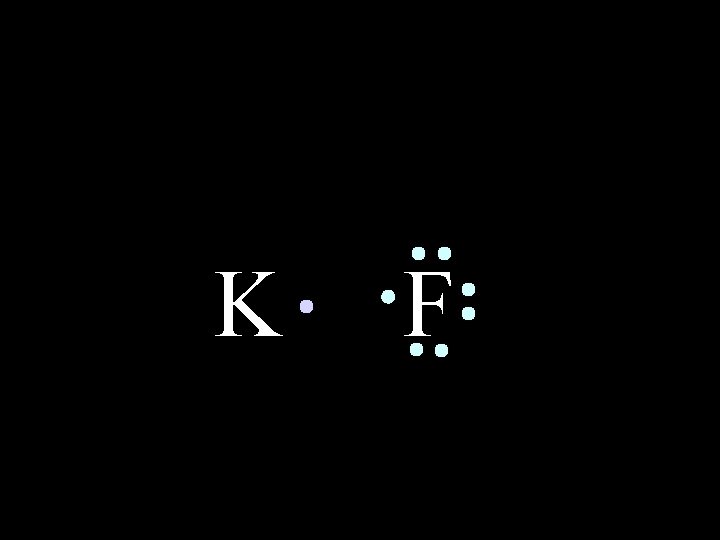
K 6/8/2021 F Chem-160 6

K 6/8/2021 F Chem-160 7
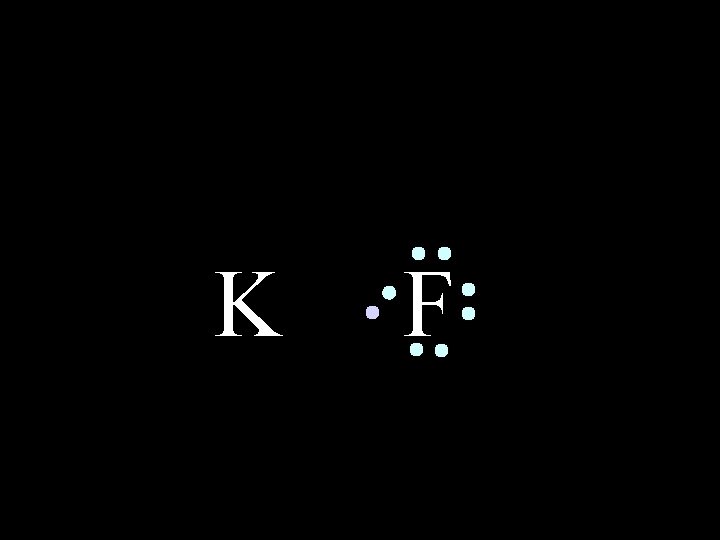
K 6/8/2021 F Chem-160 8
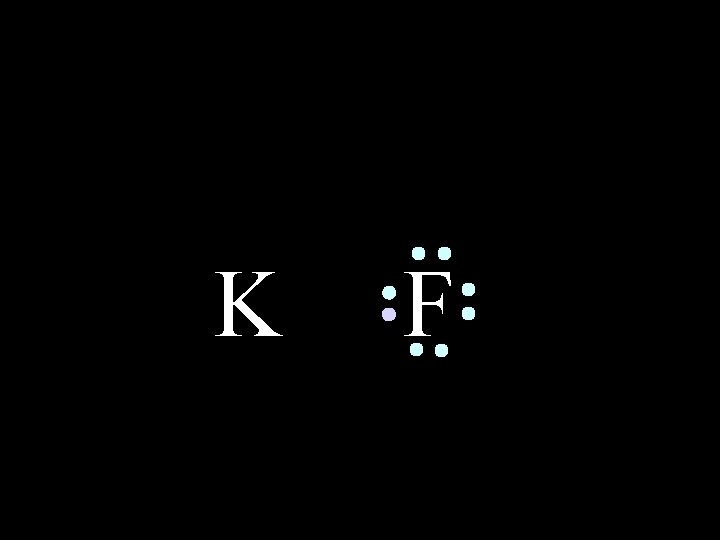
K 6/8/2021 F Chem-160 9
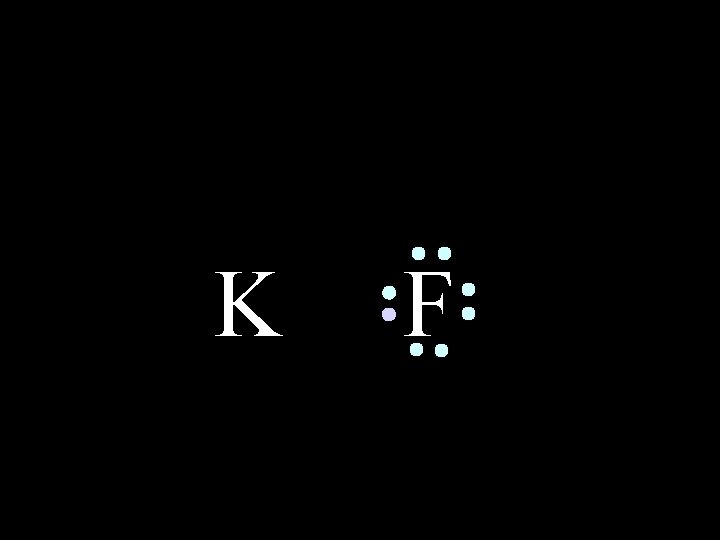
K 6/8/2021 F Chem-160 10
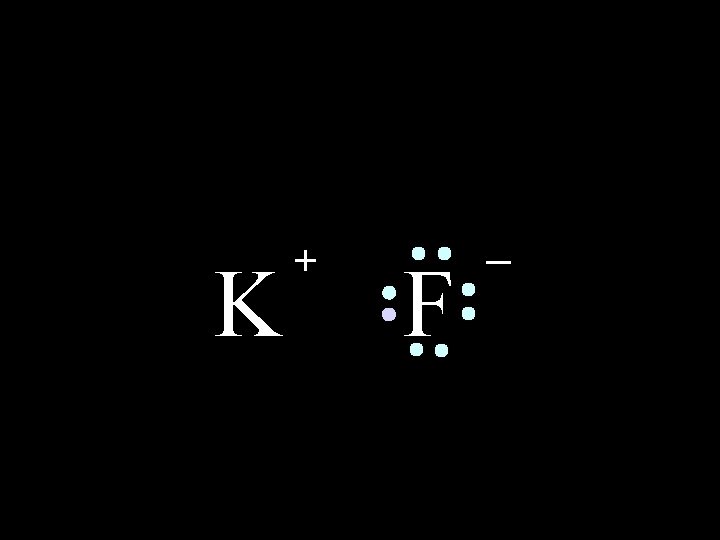
K 6/8/2021 + F Chem-160 _ 11

K + F _ The compound potassium fluoride consists of potassium (K+) ions and fluoride (F-) ions 6/8/2021 Chem-160 12

K + F _ The ionic bond is the attraction between the positive K+ ion and the negative F- ion 6/8/2021 Chem-160 13

So what are covalent bonds? 6/8/2021 Chem-160 14

In covalent bonding, atoms still want to achieve a noble gas configuration (the octet rule). 6/8/2021 Chem-160 15

In covalent bonding, atoms still want to achieve a noble gas configuration (the octet rule). But rather than losing or gaining electrons, atoms now share an electron pair. 6/8/2021 Chem-160 16

In covalent bonding, atoms still want to achieve a noble gas configuration (the octet rule). But rather than losing or gaining electrons, atoms now share an electron pair. The shared electron pair is called a bonding pair 6/8/2021 Chem-160 17

Chlorine forms a covalent bond with itself 6/8/2021 Cl 2 Chem-160 18

Cl 6/8/2021 Cl Chem-160 How will two chlorine atoms react? 19

Cl Cl Each chlorine atom wants to gain one electron to achieve an octet 6/8/2021 Chem-160 20

Cl Cl Neither atom will give up an electron – chlorine is highly electronegative. What’s the solution – what can they do to achieve an octet? 6/8/2021 Chem-160 21
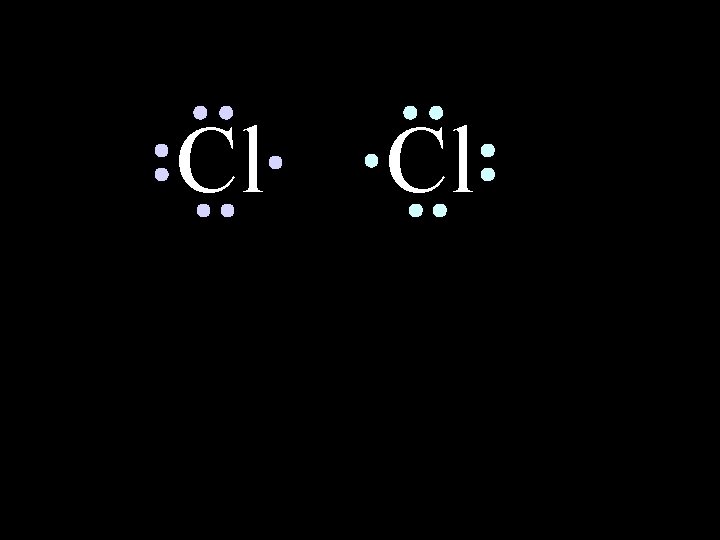
Cl 6/8/2021 Cl Chem-160 22

Cl Cl 6/8/2021 Chem-160 23

Cl Cl 6/8/2021 Chem-160 24

Cl Cl 6/8/2021 Chem-160 25

Cl Cl octet 6/8/2021 Chem-160 26

Cl Cl octet 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 27

Cl Cl The octet is achieved by each atom sharing the electron pair in the middle 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 28

Cl Cl The octet is achieved by each atom sharing the electron pair in the middle 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 29

Cl Cl This is the bonding pair 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 30

Cl Cl It is a single bonding pair 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 31

Cl Cl It is called a SINGLE BOND 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 32

Cl Cl Single bonds are abbreviated with a dash 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 33

Cl Cl This is the chlorine molecule, Cl 2 6/8/2021 circle the electrons for each atom that completes Chem-160 their octets 34

O 2 Oxygen is also one of the diatomic molecules 6/8/2021 Chem-160 35

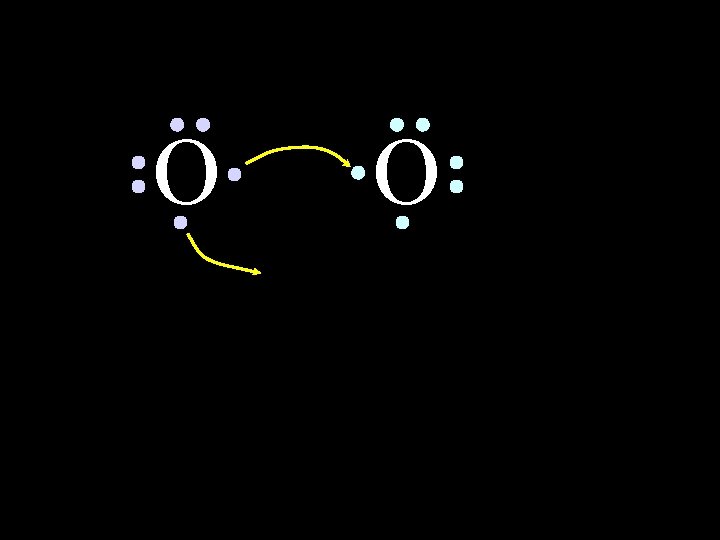
O O How will two oxygen atoms bond? 6/8/2021 Chem-160 36

O O Each atom has two unpaired electrons 6/8/2021 Chem-160 37
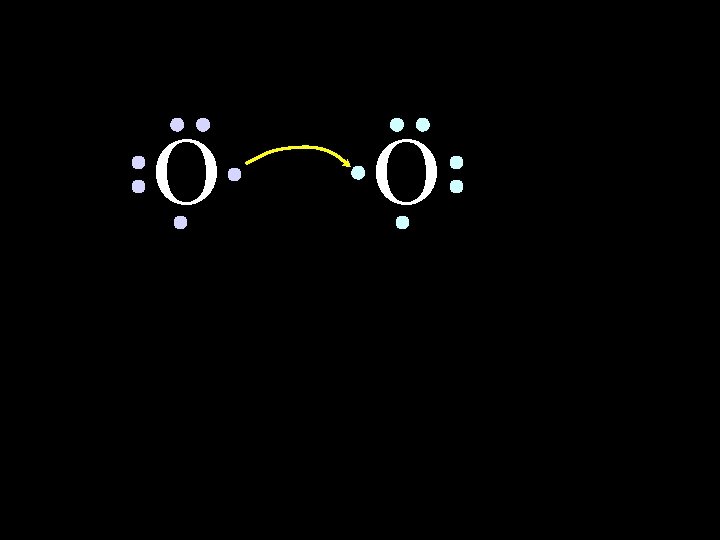
O 6/8/2021 O Chem-160 38
O 6/8/2021 O Chem-160 39
O 6/8/2021 O Chem-160 40
O 6/8/2021 O Chem-160 41
O 6/8/2021 O Chem-160 42
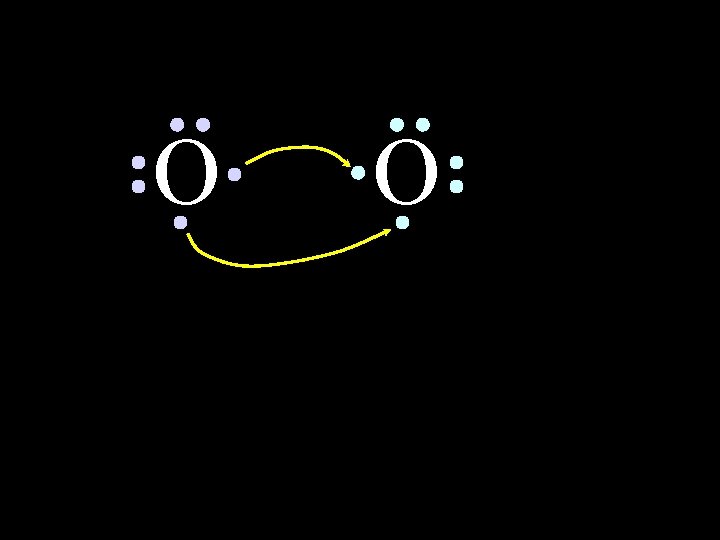
O 6/8/2021 O Chem-160 43
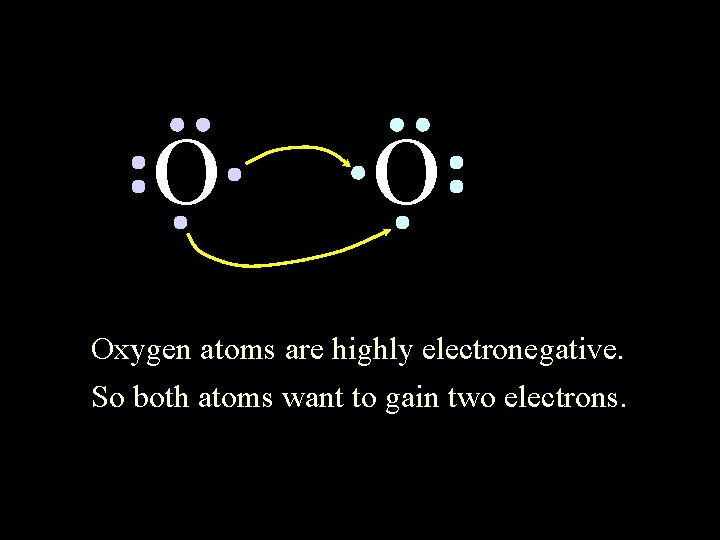
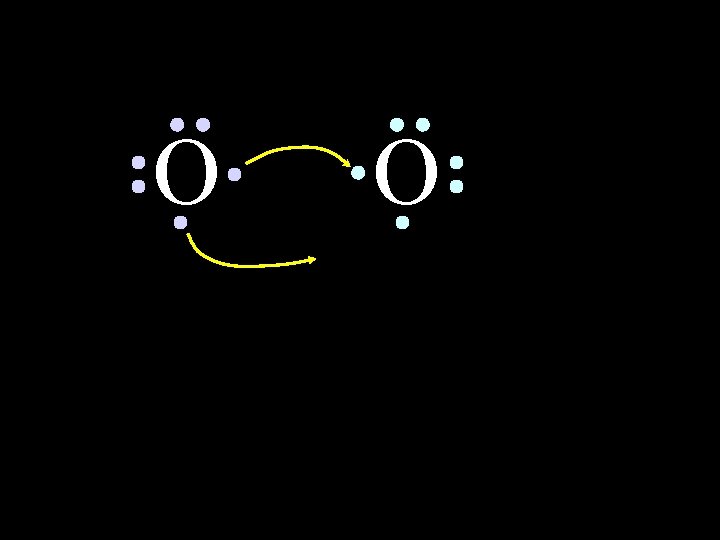
O O Oxygen atoms are highly electronegative. So both atoms want to gain two electrons. 6/8/2021 Chem-160 44
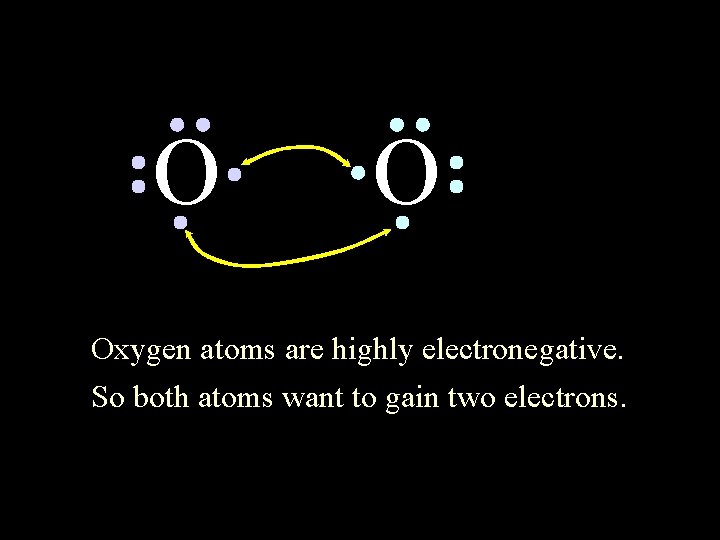
O O Oxygen atoms are highly electronegative. So both atoms want to gain two electrons. 6/8/2021 Chem-160 45

O 6/8/2021 O Chem-160 46
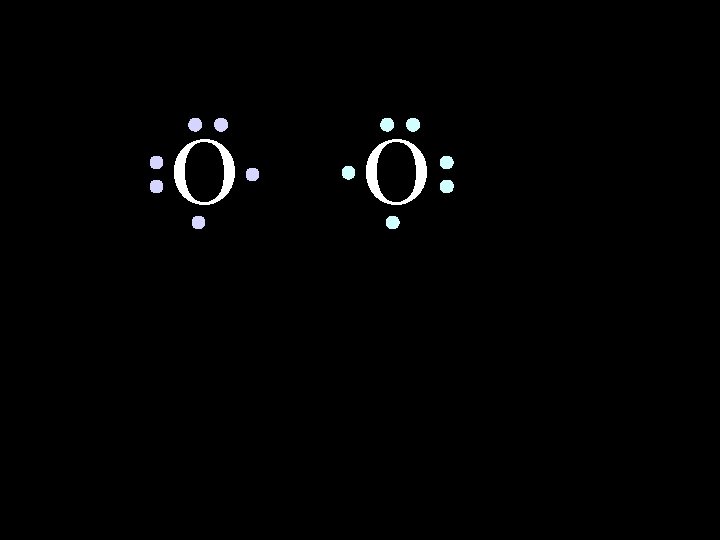
O 6/8/2021 O Chem-160 47
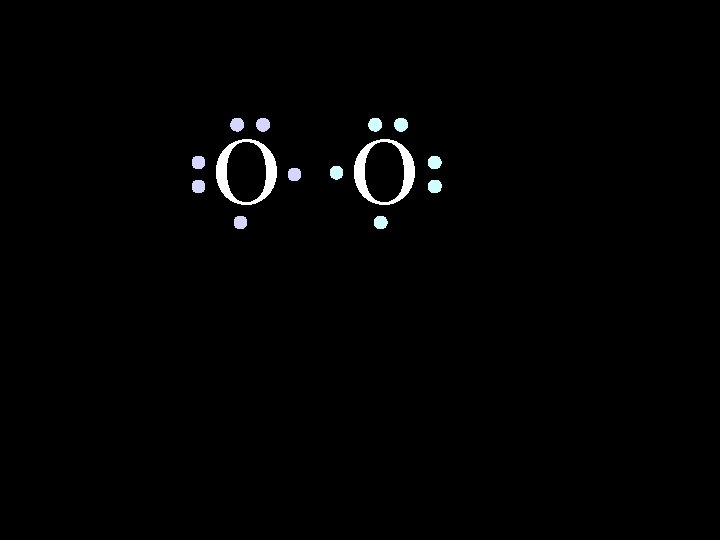
O O 6/8/2021 Chem-160 48

O O 6/8/2021 Chem-160 49
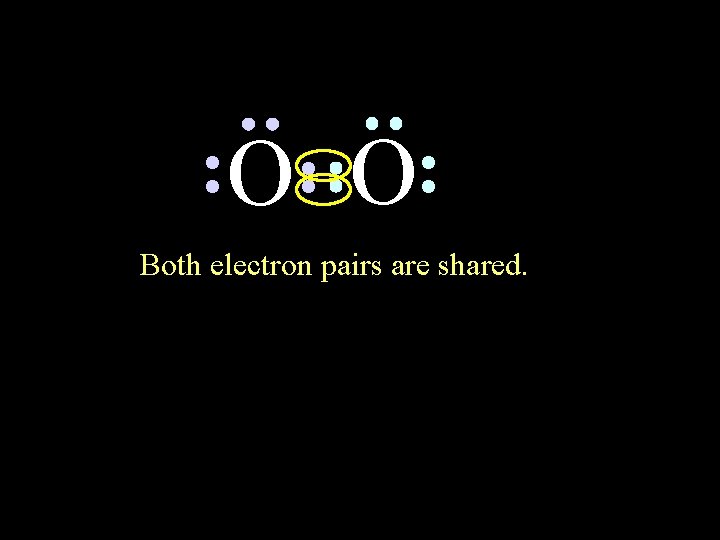
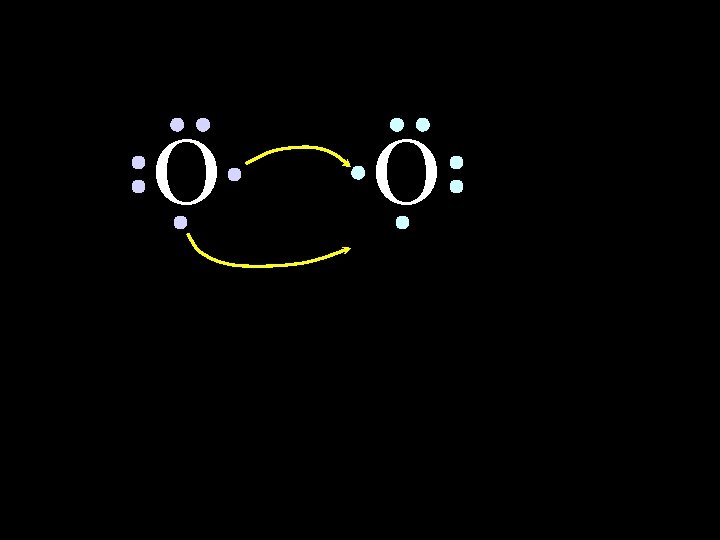
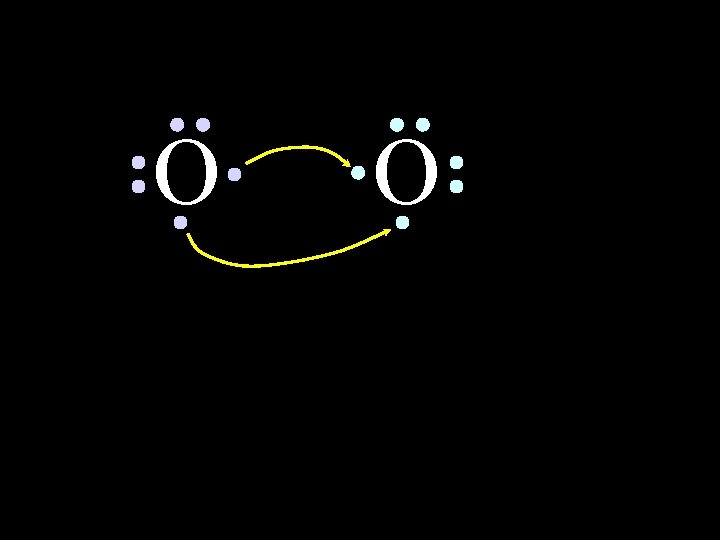
O O Both electron pairs are shared. 6/8/2021 Chem-160 50

O O 6 valence electrons plus 2 shared electrons = full octet 6/8/2021 Chem-160 51

O O 6 valence electrons plus 2 shared electrons = full octet 6/8/2021 Chem-160 52

O O two bonding pairs, making a double bond 6/8/2021 Chem-160 53

O O O =O For convenience, the double bond can be shown as two dashes. 6/8/2021 Chem-160 54

O =O This is the oxygen molecule, O 2 6/8/2021 Chem-160 55
6/8/2021 Chem-160 56
- Slides: 56