Carbohydrates Dr Seemal Jelani Introduction to Biochemistry 1

Carbohydrates Dr Seemal Jelani Introduction to Biochemistry 1 10/2/2020

Carbohydrates Most abundant Their abundance in human body is low but constitute about 75% by mass of dry plant materials Green (chlorophyll-containing) plants produce carbohydrates via photosynthesis A major source of energy from our diet composed of the elements C, H and O. They also called saccharides, which means “sugars. ” Dr Seemal Jelani Introduction to Biochemistry 2 10/2/2020

Carbohydrates They are produced by photosynthesis in plants. such as glucose are synthesized in plants from CO 2 from the air, H 2 O from the soil, and energy from the sun absorbed in chlorophyll to form carbohydrates and O 2 Oxidized in living cells to produce CO 2, H 2 O, and energy. Dr Seemal Jelani Introduction to Biochemistry 3 10/2/2020

Uses of carbohydrates in plants Cellulose (carbohydrates) serve as structural elements Starch (carbohydrates) provide energy reserves for the plants Dr Seemal Jelani Introduction to Biochemistry 4 10/2/2020

Dietary intake of plant materials is the major carbohydrate source for humans and animals Functions: Carbohydrate oxidation provides energy Carbohydrate storage in the form of glycogen provides a short-term energy reserve Carbohydrates supply carbon atoms for the synthesis of other biochemical substances (proteins, lipids, nucleic acids) Carbohydrate form part of the structural framework of DNA and RNA Dr Seemal Jelani Introduction to Biochemistry 5 10/2/2020
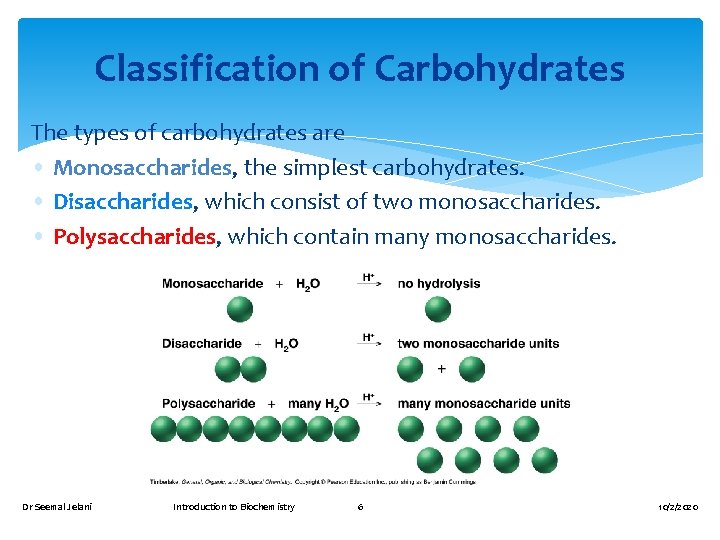
Classification of Carbohydrates The types of carbohydrates are • Monosaccharides, the simplest carbohydrates. • Disaccharides, which consist of two monosaccharides. • Polysaccharides, which contain many monosaccharides. Dr Seemal Jelani Introduction to Biochemistry 6 10/2/2020

General Formula Cn H 2 n On Carbohydrates are Polyhydroxy aldehydes, Polyhydroxy ketones The carbohydrate glucose is Polyhydroxy aldehyde and the carbohydrate fructose is Polyhydroxy ketone Dr Seemal Jelani Introduction to Biochemistry 7 10/2/2020

Functional groups in carbohydrates Aldehydes CHO Aldose Ketone C=O Ketose Dr Seemal Jelani Introduction to Biochemistry 8 10/2/2020
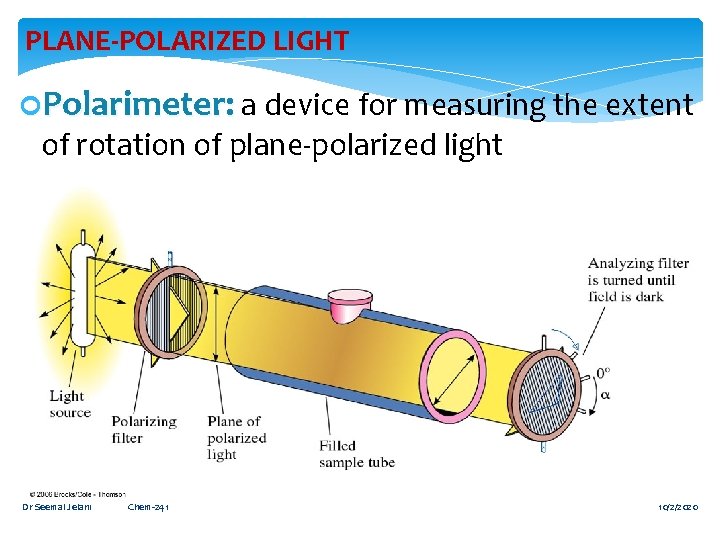
PLANE-POLARIZED LIGHT Polarimeter: a device for measuring the extent of rotation of plane-polarized light Dr Seemal Jelani Chem-241 9 10/2/2020
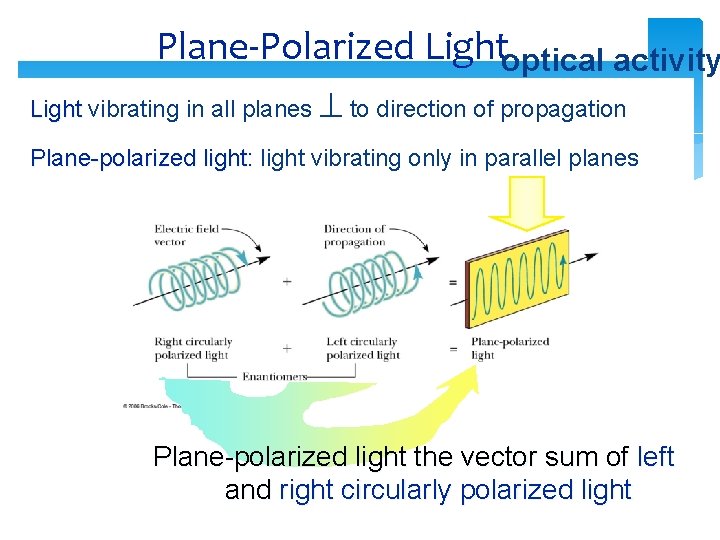
Plane-Polarized Lightoptical activity Light vibrating in all planes to direction of propagation Plane-polarized light: light vibrating only in parallel planes Plane-polarized light the vector sum of left and right circularly polarized light
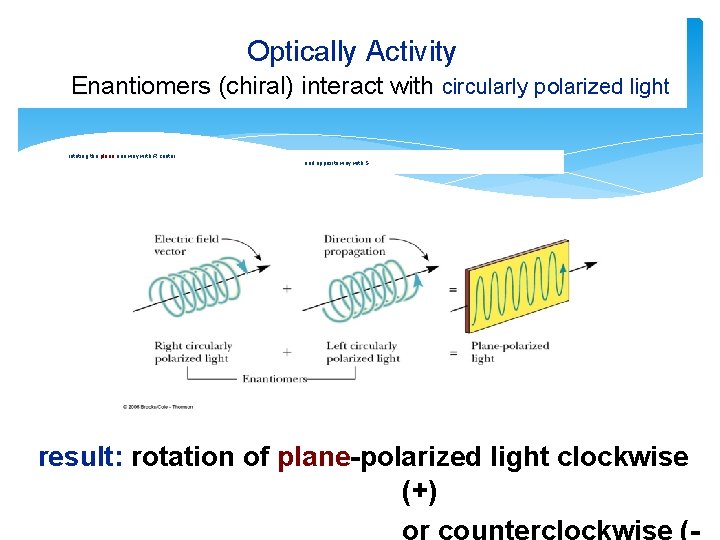
Optically Activity Enantiomers (chiral) interact with circularly polarized light rotating the plane one way with R center and opposite way with S result: rotation of plane-polarized light clockwise (+)
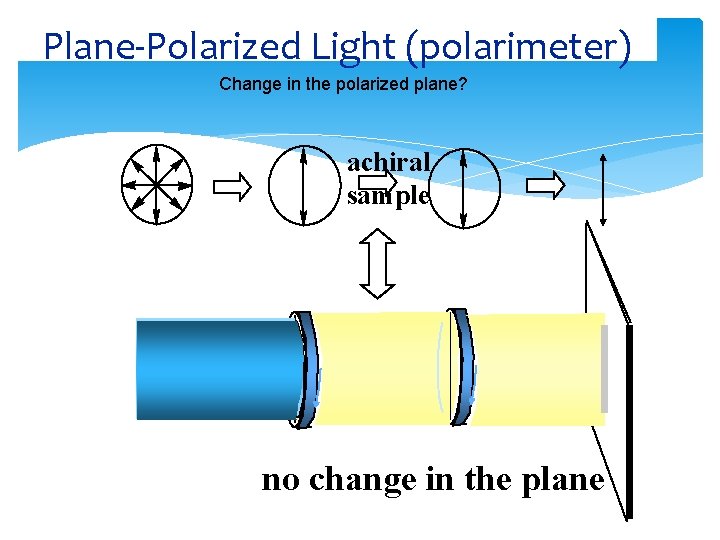
Plane-Polarized Light (polarimeter) Change in the polarized plane? achiral sample no change in the plane
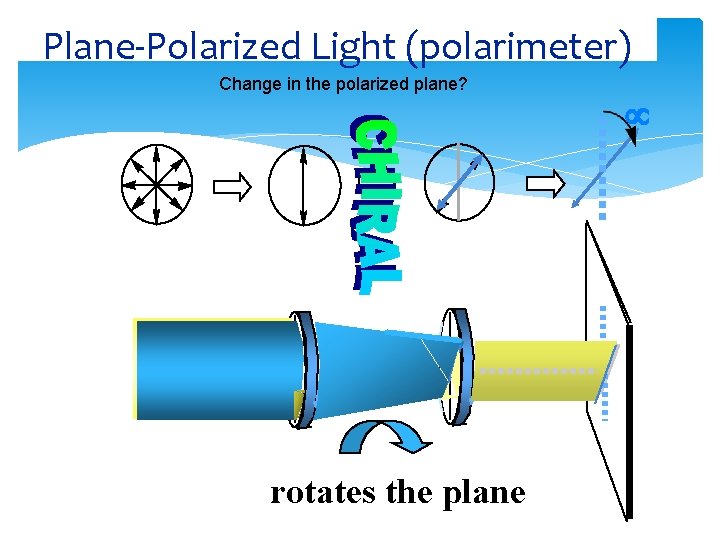
Plane-Polarized Light (polarimeter) Change in the polarized plane? rotates the plane

Classification of Monosaccharides consist of 3 -6 carbon atoms typically. A carbonyl group (aldehyde or ketone). Several hydroxyl groups. 2 types of monosaccharide structures: Aldoses and ketoses Dr Seemal Jelani Introduction to Biochemistry 14 10/2/2020
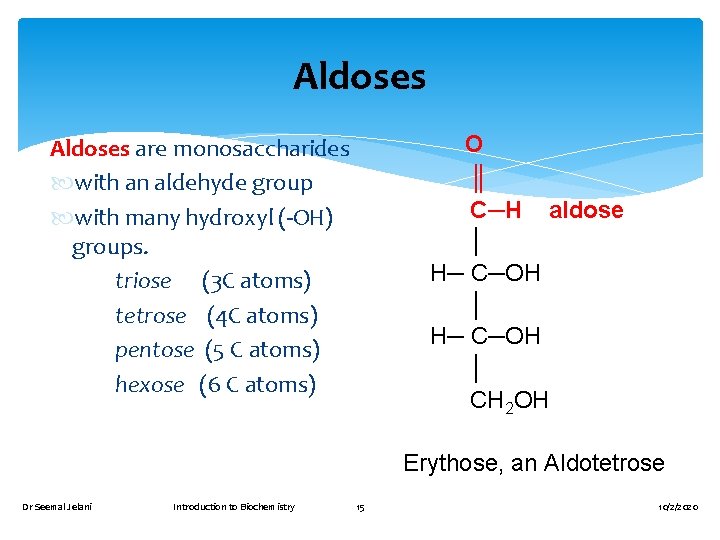
Aldoses O ║ C─H aldose │ H─ C─OH │ CH 2 OH Aldoses are monosaccharides with an aldehyde group with many hydroxyl (-OH) groups. triose (3 C atoms) tetrose (4 C atoms) pentose (5 C atoms) hexose (6 C atoms) Erythose, an Aldotetrose Dr Seemal Jelani Introduction to Biochemistry 15 10/2/2020
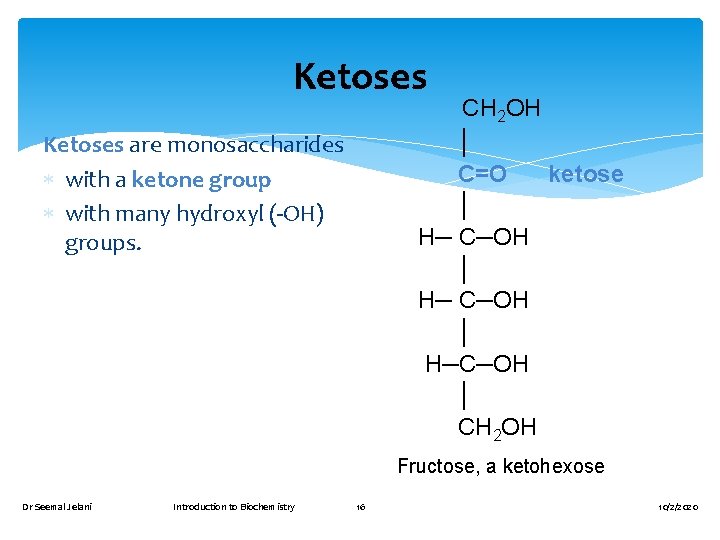
Ketoses CH 2 OH │ C=O ketose │ H─ C─OH │ H─C─OH │ CH 2 OH Ketoses are monosaccharides with a ketone group with many hydroxyl (-OH) groups. Fructose, a ketohexose Dr Seemal Jelani Introduction to Biochemistry 16 10/2/2020

Dr Seemal Jelani Introduction to Biochemistry 17 10/2/2020

Learning Check Identify each as aldo- or keto- and as tetrose, pentose, or hexose: Dr Seemal Jelani Introduction to Biochemistry 18 10/2/2020

Structures of Monosaccharides Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings Dr Seemal Jelani Introduction to Biochemistry 19 10/2/2020

D and L Notations In a Fischer projection, the −OH group on the chiral carbon farthest from the carbonyl group determines an L or D isomer. left is assigned the letter L for the L-form. right is assigned the letter D for the D-form. Dr Seemal Jelani Introduction to Biochemistry 20 10/2/2020
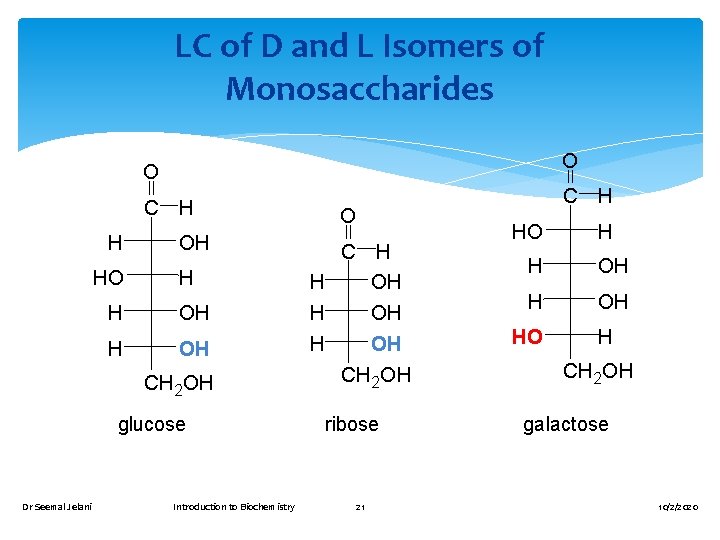
LC of D and L Isomers of Monosaccharides O O C H H HO OH H H OH CH 2 OH glucose Dr Seemal Jelani Introduction to Biochemistry C H O C H H OH OH CH 2 OH ribose 21 HO H H OH HO H CH 2 OH galactose 10/2/2020

D-Glucose found in fruits, corn syrup, and honey. an aldohexose with the formula C 6 H 12 O 6 known as blood sugar in the body. The monosaccharide in polymers of starch, cellulose, and glycogen. Dr Seemal Jelani Introduction to Biochemistry 22 10/2/2020
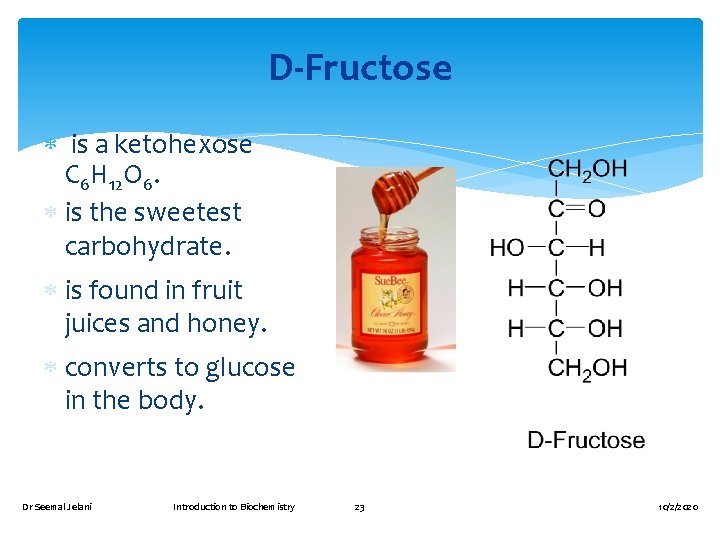
D-Fructose is a ketohexose C 6 H 12 O 6. is the sweetest carbohydrate. is found in fruit juices and honey. converts to glucose in the body. Dr Seemal Jelani Introduction to Biochemistry 23 10/2/2020
Cyclic Structures Cyclic structures are the widespread form of monosaccharides with 5 or 6 carbon atoms. Dr Seemal Jelani Introduction to Biochemistry 24 10/2/2020

The cyclic forms of monosaccharides result from the ability of their carbonyl group to react intramolecularly with a –OH group Cyclic structure is formed when the –OH group on C-5 reacts with the aldehyde group or ketone group The result is a cyclic hemiacetal or cyclic hemiketal Dr Seemal Jelani Introduction to Biochemistry 25 10/2/2020

LE 5 -4 Abbreviated ring structure
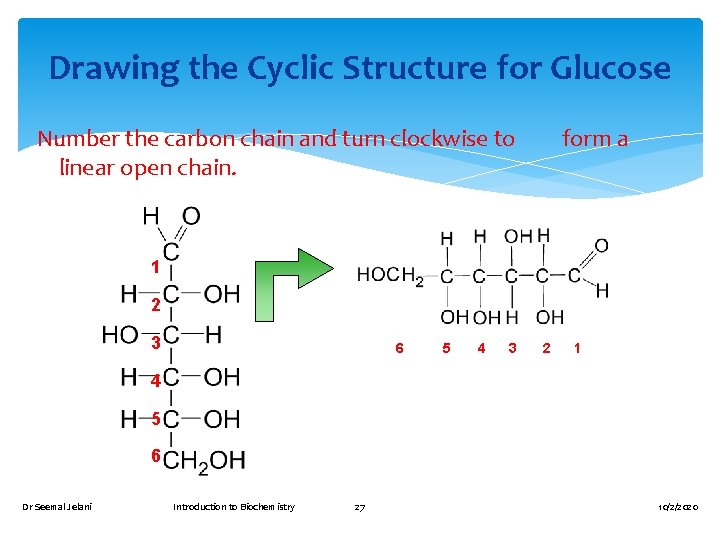
Drawing the Cyclic Structure for Glucose Number the carbon chain and turn clockwise to form a linear open chain. 1 2 3 6 5 4 3 2 1 4 5 6 Dr Seemal Jelani Introduction to Biochemistry 27 10/2/2020
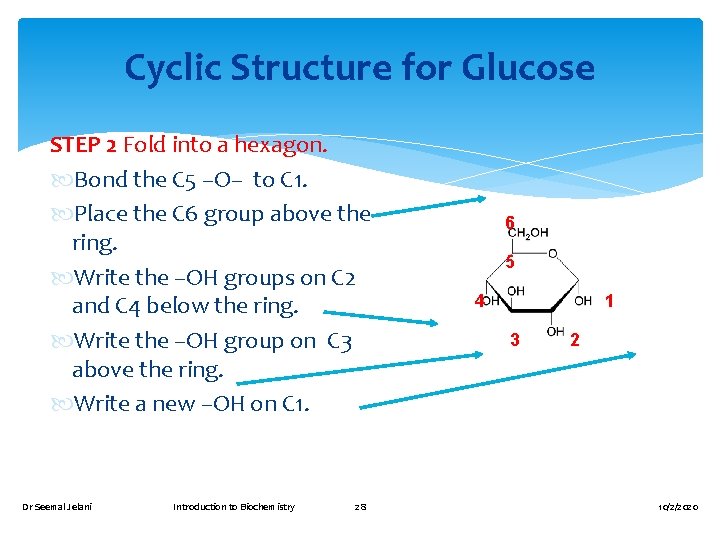
Cyclic Structure for Glucose STEP 2 Fold into a hexagon. Bond the C 5 –O– to C 1. Place the C 6 group above the ring. Write the –OH groups on C 2 and C 4 below the ring. Write the –OH group on C 3 above the ring. Write a new –OH on C 1. Dr Seemal Jelani Introduction to Biochemistry 28 6 5 4 1 3 2 10/2/2020
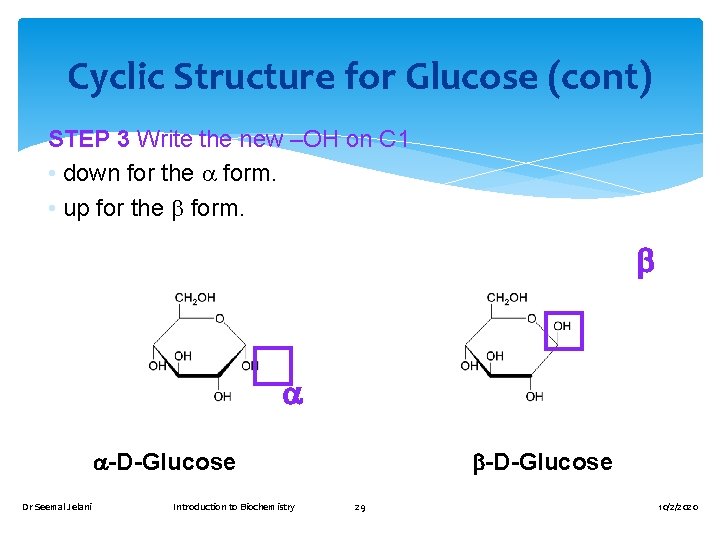
Cyclic Structure for Glucose (cont) STEP 3 Write the new –OH on C 1 • down for the form. • up for the form. -D-Glucose Dr Seemal Jelani Introduction to Biochemistry -D-Glucose 29 10/2/2020
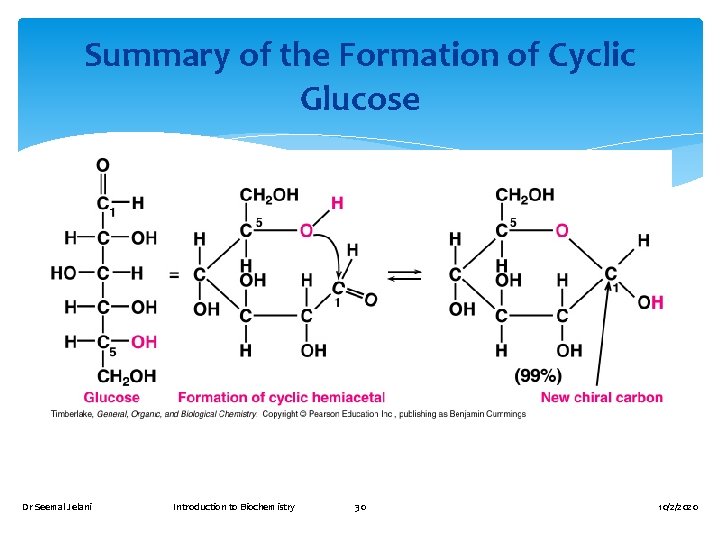
Summary of the Formation of Cyclic Glucose Dr Seemal Jelani Introduction to Biochemistry 30 10/2/2020
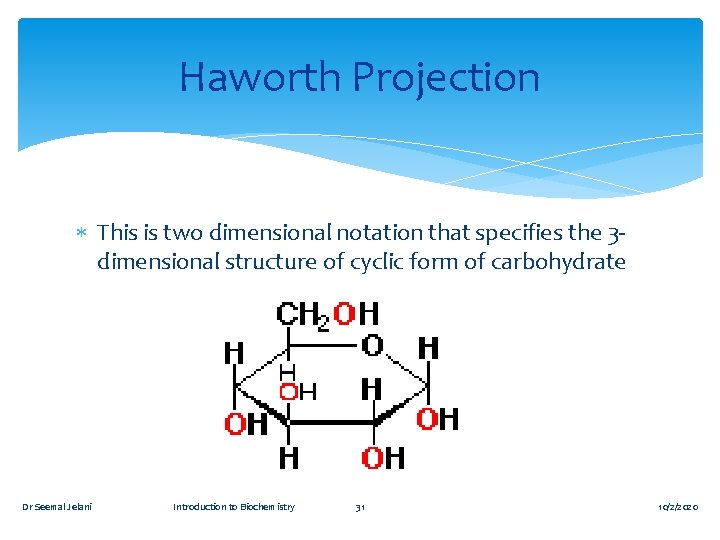
Haworth Projection This is two dimensional notation that specifies the 3 dimensional structure of cyclic form of carbohydrate Dr Seemal Jelani Introduction to Biochemistry 31 10/2/2020

The D and L form of a monosaccharide is determined by the position of the terminal CH 2 OH gp on the highest-numbered ring carbon atom In D-form, this group is positioned above the ring In L-form the terminal CH 2 OH gp is positioned below the ring (not encountered in biological systems) Dr Seemal Jelani Introduction to Biochemistry 32 10/2/2020

α and β configurations is determined by the position of the –OH gp on carbon no 1 relative to the CH 2 OH In β-configuration both of these gps point in the same direction In α-configuration the two gps point in opposites direction Dr Seemal Jelani Introduction to Biochemistry 33 10/2/2020
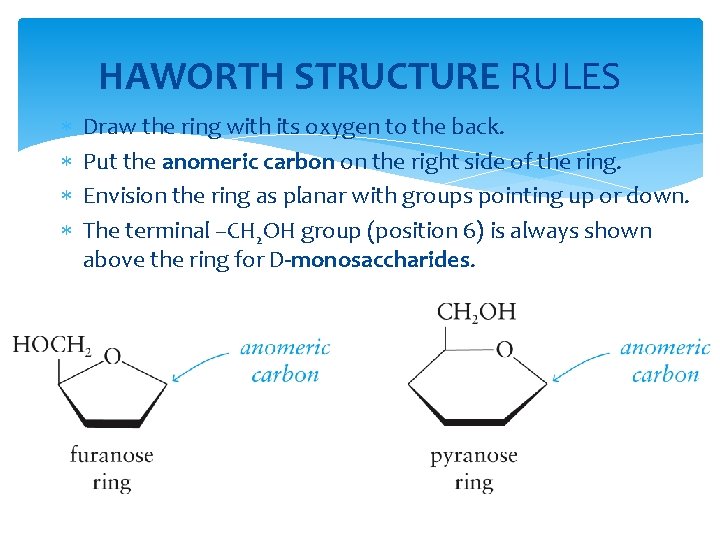
HAWORTH STRUCTURE RULES Draw the ring with its oxygen to the back. Put the anomeric carbon on the right side of the ring. Envision the ring as planar with groups pointing up or down. The terminal –CH 2 OH group (position 6) is always shown above the ring for D-monosaccharides.

Anomeric Carbon The anomeric carbon is a stereocenter Dr Seemal Jelani Introduction to Biochemistry 35 10/2/2020
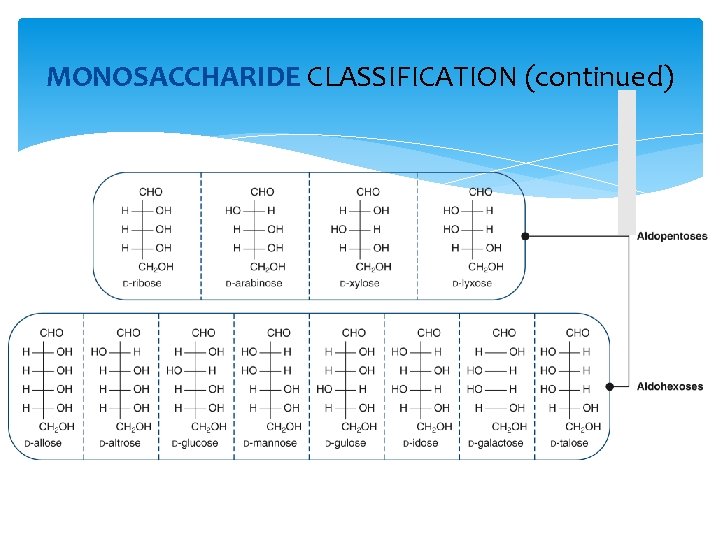
MONOSACCHARIDE CLASSIFICATION (continued)
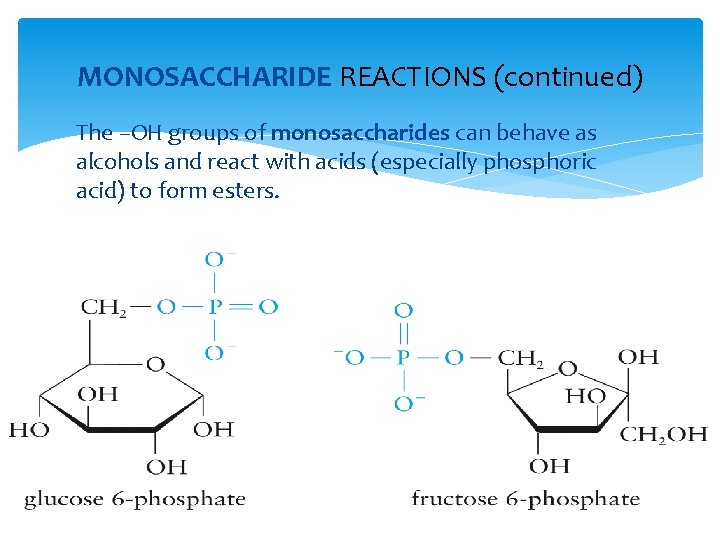
MONOSACCHARIDE REACTIONS (continued) The –OH groups of monosaccharides can behave as alcohols and react with acids (especially phosphoric acid) to form esters.
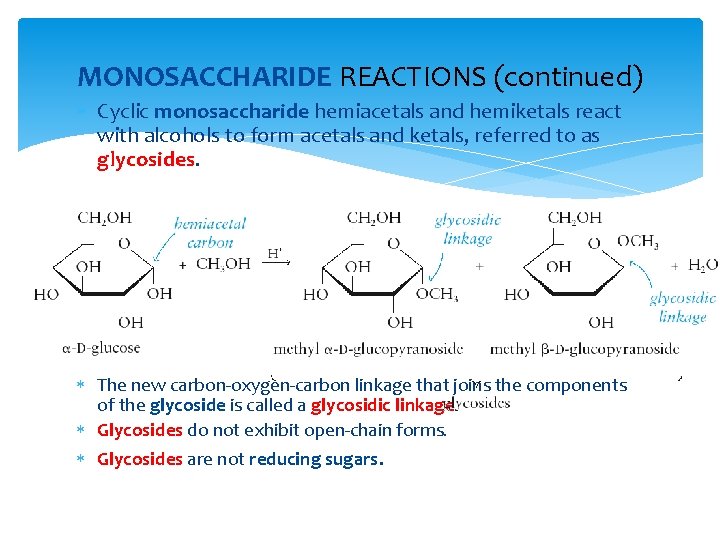
MONOSACCHARIDE REACTIONS (continued) Cyclic monosaccharide hemiacetals and hemiketals react with alcohols to form acetals and ketals, referred to as glycosides. The new carbon-oxygen-carbon linkage that joins the components of the glycoside is called a glycosidic linkage. Glycosides do not exhibit open-chain forms. Glycosides are not reducing sugars.

Reactions of Monosaccharides Oxidation Reduction Glycoside formation Phosphate ester formation Amino sugar formation Dr Seemal Jelani Introduction to Biochemistry 39 10/2/2020

Oxidation Yield three different types of oxidation products Oxidizing agent used to determined the product Weak oxidizing Agent: Tollen's Reagent Benedict Solution Reducing Sugars Is a carbohydrate that gives a positive test with TR, FS and BS Dr Seemal Jelani Introduction to Biochemistry 40 10/2/2020

Strong oxidizing agent oxidizes both end of monosaccharide i. e Terminal pri-alcohol and carbonyl group to give dicarboxylic acid Such polyhydroxy dicarboxylic acids are known as – aric acids The oxidation of glucose gives Glucaric acid Dr Seemal Jelani Introduction to Biochemistry 41 10/2/2020

Reduction Carbonyl group present in a monosaccharide ( aldose, ketose) can be reduced to a Hydroxyl group using Hydrogen as a reducing group Product is called Sugar Alcohol D- Glucitol is also known as D-sorbitol These are used as moisturizing agents in foods and cosmetics because of their affinity for water Dr Seemal Jelani Introduction to Biochemistry 42 10/2/2020

Glycoside Formation Cyclic forms of monosaccharides are hemiacetals and hemiketals react with alcohols to form Acetals and Ketals The general name for monosaccharide acetals and Ketals is Glycoside It is an Acetal or a Ketal formed from a cyclic monosaccharide Dr Seemal Jelani Introduction to Biochemistry 43 10/2/2020

Glycoside produced from glucose is Glycoside produced from galactose is Galactoside Exist in α and β forms Named as by listing alkyl or aryl group attached to the oxygen followed by the name of a monosaccharide involved with the suffix-ide Dr Seemal Jelani Introduction to Biochemistry 44 10/2/2020

Disaccharides Dr Seemal Jelani Introduction to Biochemistry 45 10/2/2020

Important Disaccharides A disaccharide consists of two monosaccharides. Monosaccharides Disaccharide glucose + glucose maltose + H 2 O glucose + galactose + H 2 O glucose + fructose sucrose + H 2 O Dr Seemal Jelani Introduction to Biochemistry 46 10/2/2020

Disaccharides Monosaccharide + Monosaccharide (Functioning as A hemiacetal or an alcohol) hemiketal) Disaccharide + H 2 O glycoside Dr Seemal Jelani Introduction to Biochemistry 47 10/2/2020

Maltose is A disaccharide also known as malt sugar. Composed of two D-glucose molecules. Obtained from the hydrolysis of starch. Used in cereals, candies, and brewing. Found in both the - and β - forms. The Glycosidic linkage between the two glucose units is called (1 -4) linkage Dr Seemal Jelani Introduction to Biochemistry 48 10/2/2020

Glycosidic Formation Cyclic forms of monosaccharides are hemiacetals and hemiketals, they react with alcohols to form acetals and Ketals The bond that links two monosaccharides of a disaccharide together is called a Glycosidic linkage A Glycosidic linkage is the carbon-Oxygen-carbon bond that joins the two components of Glycoside together Dr Seemal Jelani Introduction to Biochemistry 49 10/2/2020
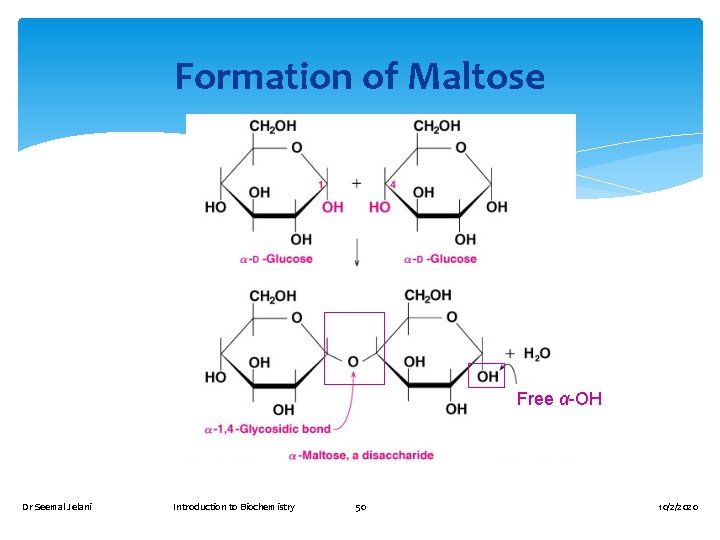
Formation of Maltose Free α-OH Dr Seemal Jelani Introduction to Biochemistry 50 10/2/2020

Glycosidic Formation Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings Dr Seemal Jelani Introduction to Biochemistry 51 10/2/2020

Polysaccharides Are polymers of D-glucose. Include Amylose and Amylopectin, starches made of α-D-glucose. Include glycogen (animal starch in muscle), which is made of α-D-glucose. Include cellulose (plants and wood), which is made of β-D-glucose. Dr Seemal Jelani Introduction to Biochemistry 52 α-D-Glucose 10/2/2020
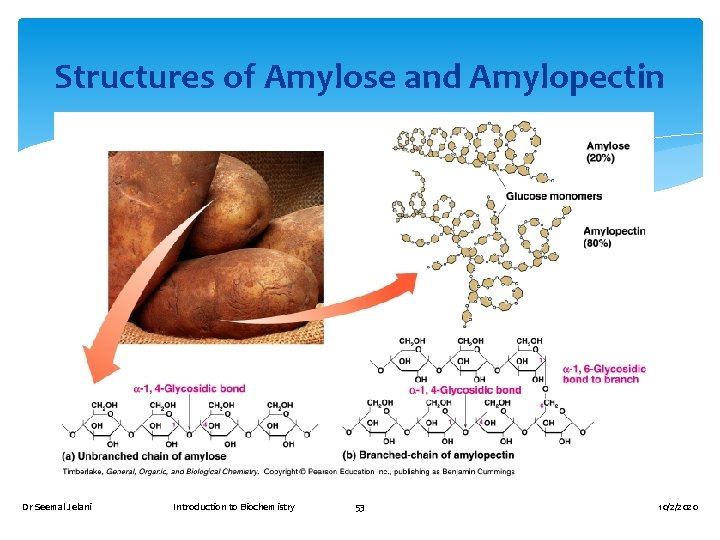
Structures of Amylose and Amylopectin Dr Seemal Jelani Introduction to Biochemistry 53 10/2/2020
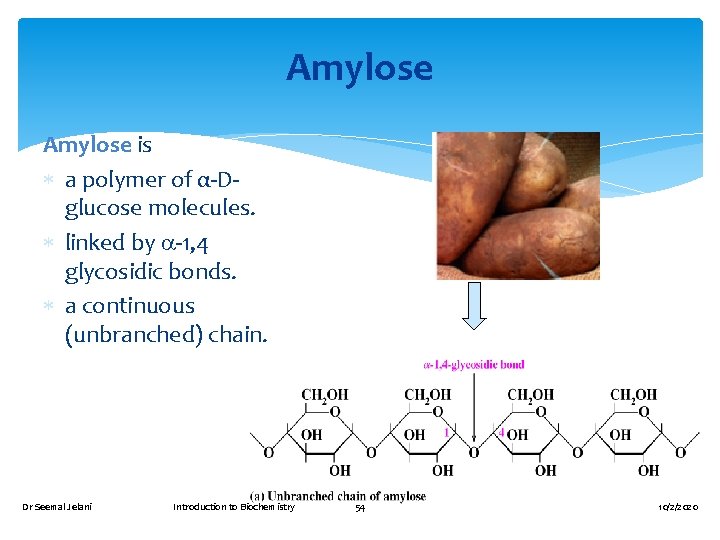
Amylose is a polymer of α-Dglucose molecules. linked by -1, 4 glycosidic bonds. a continuous (unbranched) chain. Dr Seemal Jelani Introduction to Biochemistry 54 10/2/2020
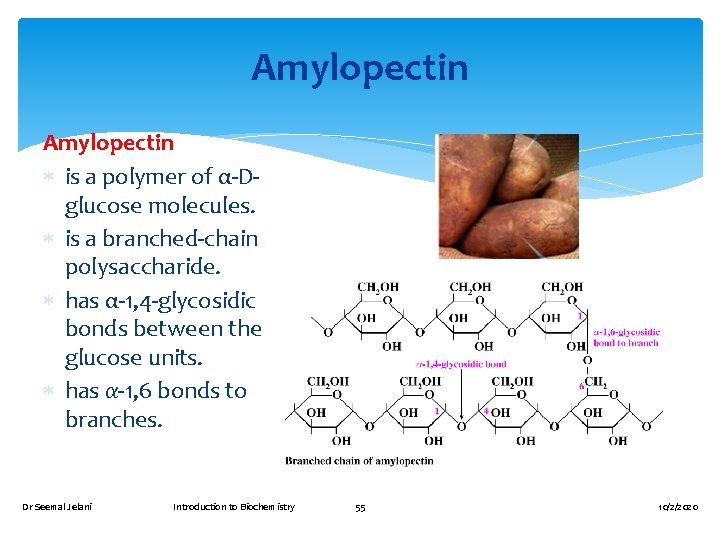
Amylopectin is a polymer of α-Dglucose molecules. is a branched-chain polysaccharide. has α-1, 4 -glycosidic bonds between the glucose units. has α-1, 6 bonds to branches. Dr Seemal Jelani Introduction to Biochemistry 55 10/2/2020

Cellulose is a polysaccharide of glucose units in unbranched chains. has β-1, 4 -glycosidic bonds. cannot be digested by humans because humans cannot break down β-1, 4 -glycosidic bonds. Dr Seemal Jelani Introduction to Biochemistry 56 10/2/2020
- Slides: 56