Chapters 16 17 Thermochemistry Thermochemistry The study of

























![Equilibrium � Rate of forward reaction = rate of reverse reaction � [products] and Equilibrium � Rate of forward reaction = rate of reverse reaction � [products] and](https://slidetodoc.com/presentation_image_h2/40dd22ab1f185ff66471cf4214ef6a39/image-33.jpg)
- Slides: 33

Chapters 16 & 17 Thermochemistry

Thermochemistry � The study of energy transfers as heat (Q) Heat (Q) -The energy transferred when there is a difference in temperature between two areas -Measured in Joules (J) or kilojoules (k. J)

Temperature � Measure particles of AVERAGE kinetic energy of ◦ Faster particles = higher temps ◦ Slower particles = lower temps � Measured in ℃elsius or Kelvins. � T(°C) = (T(°F) - 32) × 5/9 � K = 273 + ℃

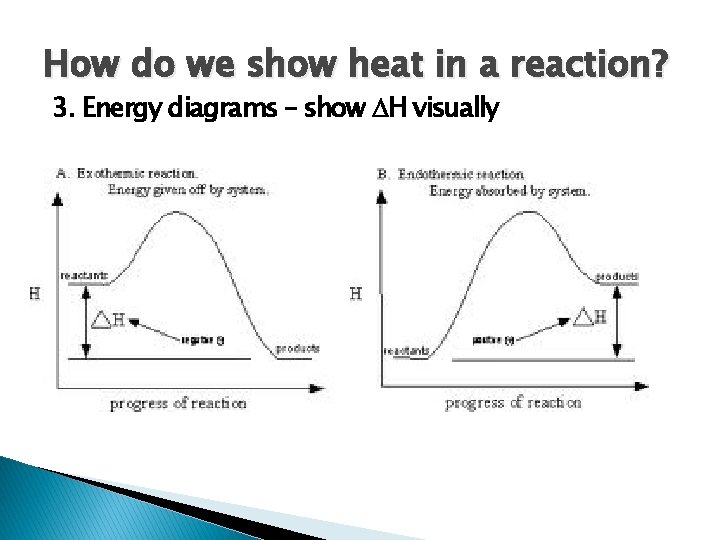
Heat of Reaction � Some reactions release or absorb heat � Exothermic -Heat energy is released (product) -Products lower in energy than reactants -Feels warm � Endothermic -Heat energy is absorbed (reactant) -Products higher in energy than reactants -Feels cold

How do we include heat in a reaction? 1. Show it as a reactant or product: 2 H 2(g) + O 2(g) 2 H 2 O(g) + 483. 6 k. J 2 H 2(g) + O 2(g) Exothermic Endothermic *This can be used as a conversion factor! (2 moles H 2 O = 483. 6 k. J heat)

How do we include heat in a reaction? 2. Write it separately as a H value (enthalpy change) Enthalpy Change, H H = Hproducts – Hreactants - H = exothermic + H = endothermic 2 H 2(g) + O 2(g) 2 H 2 O(g) H = -483. 6 k. J/mol Exothermic 2 H 2 O(g) 2 H 2(g) + O 2(g) H = +483. 6 k. J/mol Endothermic

How do we show heat in a reaction? 3. Energy diagrams – show H visually

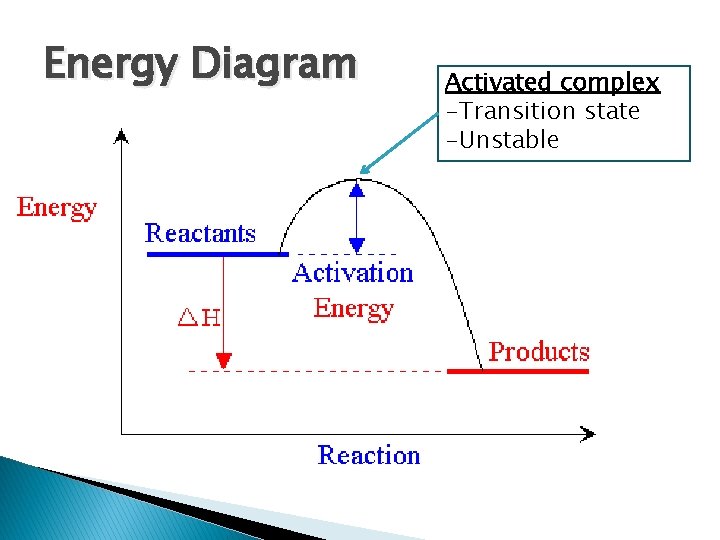
Energy Diagram Activated complex -Transition state -Unstable

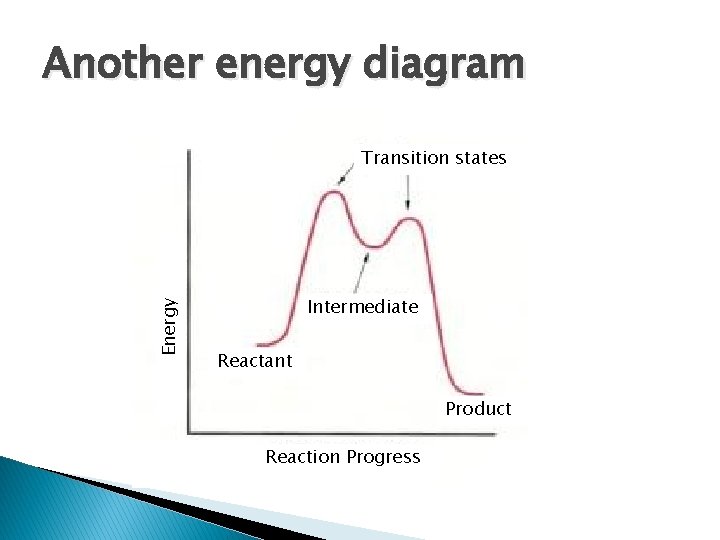
Another energy diagram Energy Transition states Intermediate Reactant Product Reaction Progress

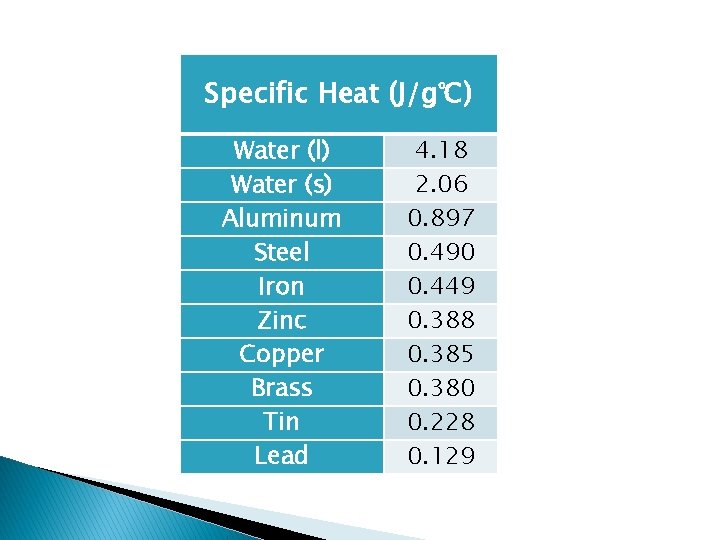
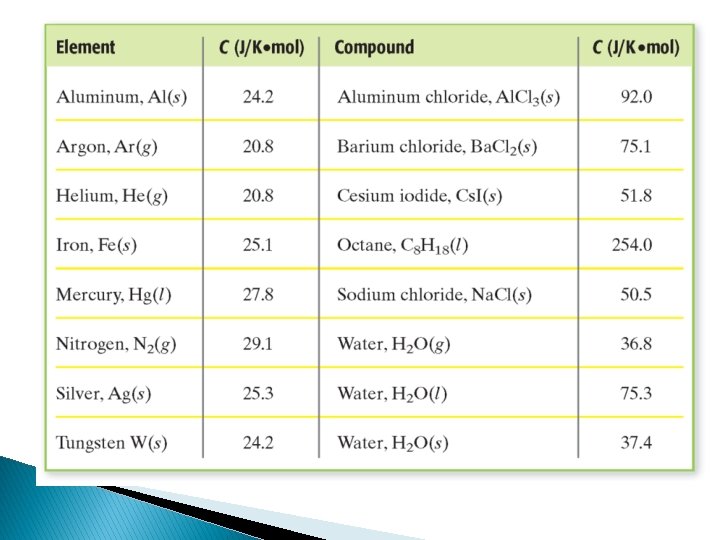
Specific Heat (cp) � Quantity of heat needed to raise the temp of 1 gram of a substance by 1 C or 1 K � Units = J/g C or J/g. K � Different substances have different specific heats � Water – high specific heat; Iron – low specific heat Q Cp = m t Q = Cpm t heat mass change in temperature

Specific Heat (J/g℃) Water (l) Water (s) Aluminum Steel Iron Zinc Copper Brass Tin Lead 4. 18 2. 06 0. 897 0. 490 0. 449 0. 388 0. 385 0. 380 0. 228 0. 129


Calorimeter – reaction chamber § Used to measure the energy absorbed or released as heat in a chemical or physical change. § Usually substances are immersed in a known quantity of water. v. Energy given off by the reaction is absorbed by the water, and the Δt of the water is measured. v. From the Δt of the water, it is possible to calculate the Q of the reaction.

Combustion Calorimeter

Examples: pg. ) 534 � Determine the specific heat of a material if a 35 g sample absorbed 96 J as it was heated from 293 K to 313 K.

� If 980 k. J of energy are added to 6. 2 L of water at 291 K, what will the final temperature of the water be?

Calculating ΔH of Reaction � Hess’s Law - the overall enthalpy change in a reaction is equal to the sum of enthalpy changes for the individual steps in the process. � Ex. ) Formation of methane gas: + 2 O 2(g) CO 2(g) + 2 H 2 O(l) ΔH = -393. 5 k. J � C(s) + O 2(g) CO 2(g) ΔH = -285. 8 k. J � 2 H 2(g) + O 2(g) 2 H 2 O(l) ΔH = -890. 8 k. J -----------------------------------------� CH 4(g) � C(s) + 2 H 2(g) CH 4(g) ΔH = ?

Enthalpy Formation �C + O 2 CO 2 ΔH = -393. 5 k. J � H 2+ O 2 H 2 O ΔH = -285. 8 k. J � C 5 H 12+ 8 O 2 5 CO 2 + 6 H 2 O ΔH = +3535. 6 k. J � --------------------C(s) + H 2(g) C 5 H 12(g) ΔH = ? k. J

Combustion of C 8 H 18 � C(s) + O 2(g) CO 2(g) ΔH = -393. 5 k. J � H 2(g) + 0. 5 O 2(g) H 2 O(l) ΔH = -241. 8 k. J � 8 C(s) + 9 H 2(g) C 8 H 18(l) ΔH = -224. 13 k. J --------------------

Driving Forces of Reactions � Not all reactions are spontaneous � Spontaneity depends on 1. Enthalpy ( H) 2. Entropy ( S)

1. Enthalpy ( H) � Lower energy is more favorable (So, exothermic reactions usually “work”) � Exothermic ◦ - H ◦ Heat released (is a product) ◦ Feels hot - H = spontaneous + H = not spontaneous

2. Entropy ( S) � Entropy is a measure of DISORDER in a system � Units of J/mol. K ◦ T = S - S = not spontaneous ◦ gas = S + S = spontaneous ◦ Dissolving = S � More disorder = more favorable/spontaneous 2 NH 4 NO 3(s) 2 N 2 (g) + 4 H 2 O Is this spontaneous? (l) + O 2(g) Yes! Increasing disorder!!

What if H and S compete? � Put them together!! � Use Gibb’s Free Energy calculation

Gibb’s Free Energy G � Combines H and S to determine if a reaction will be spontaneous � G = H - T S - G = spontaneous + G = not spontaneous

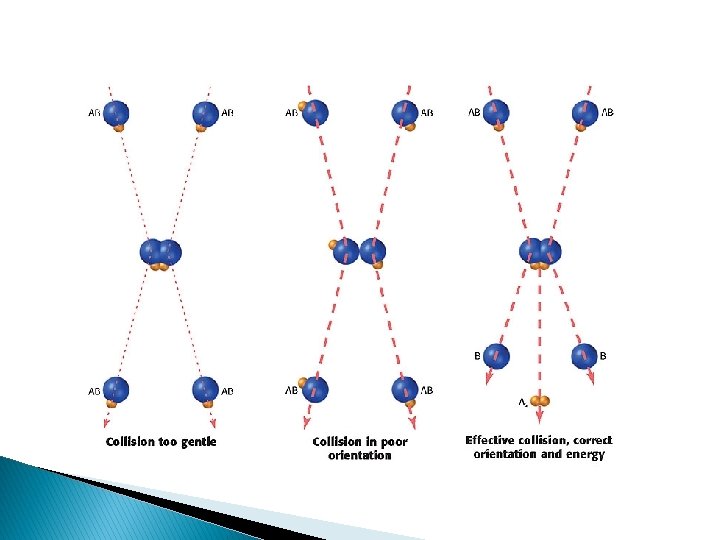
Collision Theory � Collision Theory ◦ Particles must collide with enough energy and force at a proper orientation in order for a reaction to occur � Activation energy, Ea : - Energy required for a reaction to occur


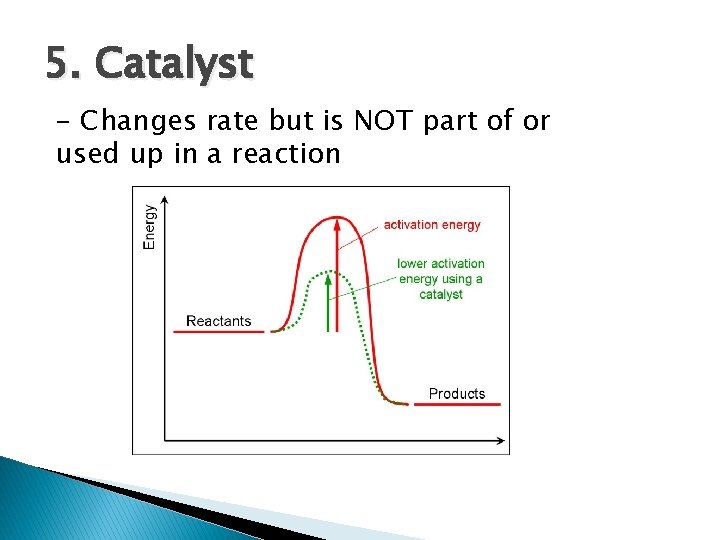
Reaction Rate � Influenced 1. ◦ 2. ◦ 3. ◦ 4. ◦ 5. by 5 factors: Nature of the reactants Bond strength, size of molecules, etc. Surface Area surface area, rate Temperature T, KE, collisions, rate Concentration [ ], rate Catalyst – lowers activation energy

5. Catalyst - Changes rate but is NOT part of or used up in a reaction

Chapter Review �Pg 552 �#’s 1, 3 -4, 7 -10, 12, 23 -25 �Due day of test

Keq – Equilibrium Constant � Gives a picture of what side of an equilibrium reaction is favored Keq � H 2 [products] = [reactants] + I 2 <--> 2 HI Keq [HI]2 = [H 2] X [I 2] Equilibrium expression Keq > 1 = Products favored Keq < 1 = Reactants favored

Le. Chatelier’s Principle � If a system at equilibrium is disturbed, the equilibrium will shift to relieve the stress ◦ PCl 5 (g) + heat What happens if: Cl 2? heat? PCl 3? Add a catalyst? PCl 3 (g) + Cl 2 (g)

Reversible reaction � Products can react to form reactants
![Equilibrium Rate of forward reaction rate of reverse reaction products and Equilibrium � Rate of forward reaction = rate of reverse reaction � [products] and](https://slidetodoc.com/presentation_image_h2/40dd22ab1f185ff66471cf4214ef6a39/image-33.jpg)
Equilibrium � Rate of forward reaction = rate of reverse reaction � [products] and [reactants] is unchanged � Is dynamic (always changing) � Sometimes favors one side: HBr + H 2 O H 3 O+ + Br. H 2 CO 3 + H 2 O H 3 O+ + HCO 3 -