Thermochemistry AP Chemistry Mr G Thermochemistry The study




















- Slides: 26

Thermochemistry AP Chemistry Mr. G

Thermochemistry- The study of Energy changes in matter. Energy- the capacity to do work or produce heat. 1 st Law of Thermodynamics- All the energy in the universe is constant. It cannot be created nor destroyed. It simply changes forms. Energy comes in many forms: Kinetic(translational) KE = 1/2 m. V 2, Rotational KE = 1/2 Iw 2, In a gas KE = 3/2 n. RT, etc. Potential (U) Energy: Positional PE = mgh, Spring U = ½ Kx 2, etc.

In Thermochemistry: • System(Sys. )- the part of the Universe on which you are focusing in on. (Usually the reaction) • Surroundings(Surr. )-the rest of the Universe. (container, room, air, oceans, mars, etc. ) • Universe(Univ. )-the system(Sys) + the surroundings(Surr. ) We can consider the system as a box: Surroundings System

Energy can enter or leave the system. It must do by work or heat. DE = work x heat Surroundings System

A gas expands and does 45 J of work. It also releases 15 J of heat. Find the change in internal energy. A gas is compressed and gains 78 J of work, while absorbing 88 J heat. A reaction does 63 J of work, while absorbing 27 J or heat. Find DE: Convert to e. V(1 e. V = 1. 60 x 10 -19 J) Convert to ergs(1 J = 1 x 107 erg)

Work-the amount of energy needed to push something a certain distance. • Work = Force x distance • In Chemistry, you have to find the amount of work associated with a gas in a piston. • (isobaric process- pressure is constant)

Finding work of a gas w/ DV (Adiabatic, q=0) • A gas in a piston is compressed from 882 ml to 645 ml against a constant 1. 25 atm. How much work is done & by who? • A gas expands from 22. 4 L to 35. 7 L against a constant 1. 55 atm. How much work is done? • A gas expands by 1. 6 liters, while W= - 472 J. What is the external pressure?
A hot air balloon changes from 4. 00 X 106 L to 4. 50 X 106 L by the addition 1. 3 X 108 J of heat. Find the change in internal energy of the balloon. A monkey pushes down on 3. 00 L piston, until it is half its volume. If the system releases 452 J as heat, what is the change in internal energy?

• AP Chem. Thermodynamics HW: • Write out each question. Box your answers. Work must be shown for credit. This is an INDIVIDUAL, NOT GROUP assignment. • p. 285, #'s: 4, 6 a, 26, 30, 32, 34, 38, 44, 46, 52, 56, 60, 72, 74, 76, 78, 82 • Due on test day.

Diabatic process- heat(q) is exchanged between system and surr. (q = 0) • Diabatic(when heat is exchanged between system and surroundings) processes can occur with or without a temperature change. • W/ temp. change: • q = m. Cp. DT • q = heat(Joules) m = mass Cp = specific heat (J/g mol) DT = Tf – Ti Specific heat- the amount of heat needed to raise the temp. of 1 gram(or mole) of subst. by 1 C o. How much heat is released when 25. 0 grams of water cool from 89 o. C to 35 o. C? (Cp. H 2 O = 4. 184 J/g. Co)

• Iron has a specific heat of 0. 450 J/g. Co. What mass of iron releases 122 J of heat when cooled from 50. 0 o. C to 47. 2 o. C? • How much heat is absorbed by 150. grams H 2 O when as it is warmed from 15. 0 o. C to 32. 5 o. C?

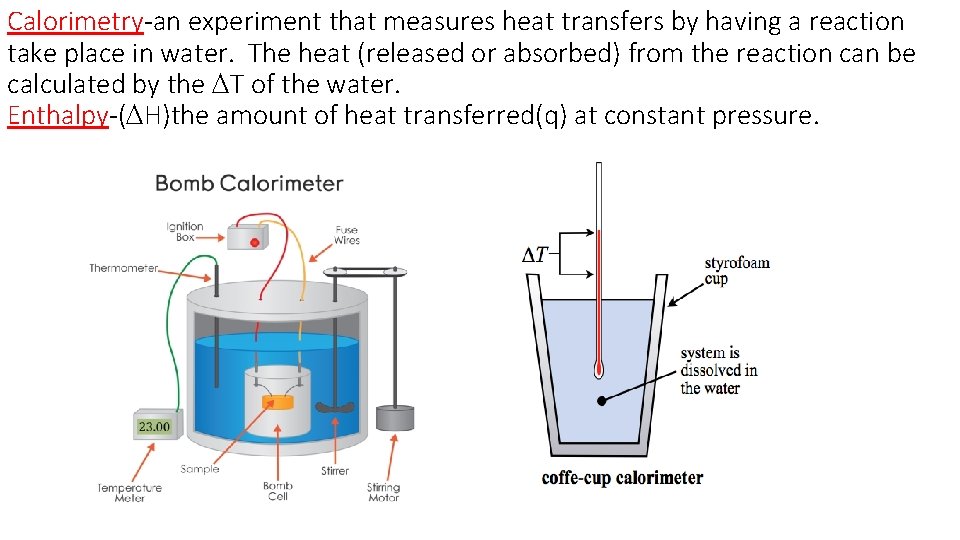
Calorimetry-an experiment that measures heat transfers by having a reaction take place in water. The heat (released or absorbed) from the reaction can be calculated by the DT of the water. Enthalpy-(DH)the amount of heat transferred(q) at constant pressure.

• The combustion of 3. 2 grams of ethane in a calorimeter raises the temperature of 800. 0 grams of water from 25. 0 o. C to 49. 8 o. C. • a) write the balanced molecular equation for the reaction: • b) How much heat(q) was released by the reaction? • c) What is the molar enthalpy of the reaction? • d) write the balanced molecular equation including heat: • e) How much heat would burning 42. 5 g ethane release?

• In a coffee cup calorimeter, 100. ml of 1. 00 M HCl is neutralized by 100. ml of 1. 00 M Na. OH. Both solutions were at 24. 2 o. C but warmed to 31. 3 o. C when the reaction was over. a) Write the balanced equation for the rxn: b) How much heat was released by the rxn? c) Find the molar enthalpy of neutralization for the rxn: d) Include the heat into the balanced equation: e) How much heat would be released by neutralizing 25. 0 grams Na. OH?

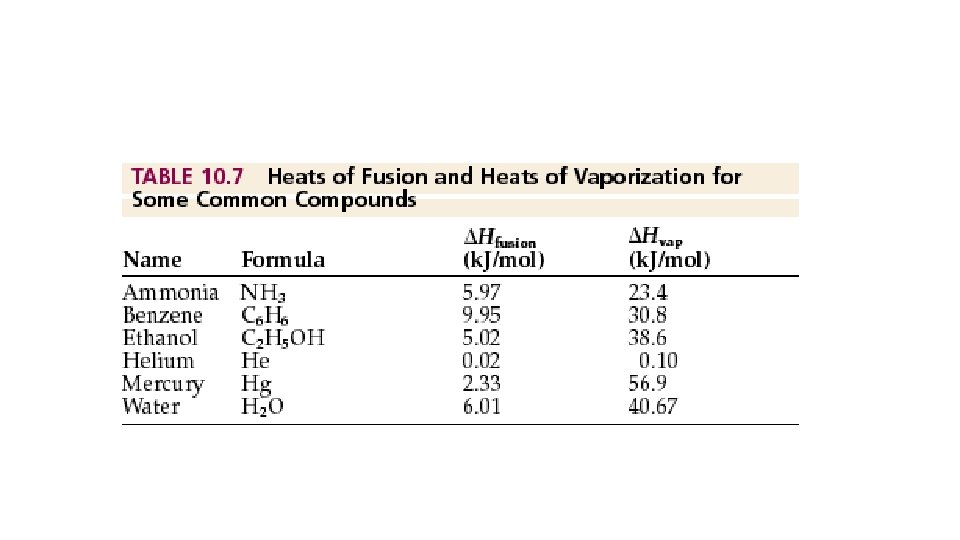
Diabatic process- heat(q) is exchanged between system and surr. (q = 0) • Diabatic processes can occur with or without a temperature change. • W/O temp. change: During a phase change: We will need: the heat of fusion(DHfus)- the amount of heat needed to melt or freeze a subst. Or The heat of vaporization(DHvap) – the amount of heat needed to boil or condense a subst. DHvap >> DHfus (always)

• How much energy is released as 25. 0 grams of water melt? • Boil? You have 25. 0 grams C 6 H 6(benzene). How much heat does it take to melt and boil?
• Calculate the amount of heat needed to heat 35. 0 grams of ice at -12 o. C to 118 o. C

Hot & cold water mixed • 100. 0 grams of water at 25 o. C are mixed with 42. 7 grams of water at 37 o. C. What is the final temp. ? • 125 grams of water at 25 o. C are mixed with 62. 5 grams of water at 37 o. C. What is the final temp. ?

• 98. 5 grams of Al at 87 o. C are dropped into a calorimeter w/ 150. ml of water at 25 o. C. What is the equilibrium temp. of the system?

Hess’s Law-the DH of a process is always the same regardless of the number of steps taken. (state function) • Solve problems from handout

Standard Heat(enthalpy) of formation, DHf o The amount of heat gained/lost when forming 1 mole of compound from each free element, in it’s most stable state(phase and allotrope) • Write the reaction for the formation of each compound: • Na. Cl • H 2 SO 4 • C 2 H 6 • P 2 O 5

Standard Enthalpies of formation • Solve Problems from handout.


