Chapter 17 Thermochemistry Thermochemistry The study of energy











- Slides: 13

Chapter 17 Thermochemistry

Thermochemistry The study of energy changes that occur during chemical reactions and changes in state.

Thermochemistry Energy: the ability to do work or to supply heat Work is the force applied to move an object. ENERGY (Joule or calorie) = HEAT (J or cal) 1 cal = 4. 18 J Chemical Potential Energy: stored energy in chemicals

Heat vs. Temp. Heat and temperature are concepts that are often confused

Heat vs. Temp. Amount of energy in a system Degree of hotness or coldness of an object Form of energy Measures the average kinetic energy of molecules Flows from a warmer object to a cooler one Usually stays within one area It is measured in Joules or It is measured in degrees calories

Calorie vs. calorie Calorie = refers to food (dietary) Calorie = 1 kilocalorie = 1, 000 calories calorie= a unit of heat EXAMPLE: Snickers Bar = 250 Calories = 250 kilocalories = 250, 000 calories

Specific Heat Capacity The amount of heat it takes to raise the temperature of 1 g of a substance 1 o. C q =m ΔT Cp q =heat energy (J or cal) m =mass (g) ΔT =(Tf – Ti) = o. C Cp = specific heat = J = heat (J) g x o. C mass (g) x (Tf – Ti)

Q =m Δ T Cp q = g o. C J g x o. C When units are canceled, q = Joules

Specific Heat Capacity The amount of heat it takes to raise the temperature of 1 g of a substance 1 o. C For example: WATER VS. METAL -Higher the specific heat: the less fluctuation of heat and therefore the less fluctuation of the temp. of the object. Water Iron 4. 18 J/(g x o. C) 0. 46 J/(g x o. C) High Low

Exothermic vs. Endothermic Exothermic: a process in which heat is released to the surrounding - The system loses heat as the surroundings heat up - q has a negative value because the system is losing heat Endothermic: a process in which the system gains heat as the surroundings cool down - Heat flows into a system - q has a positive value because the system is gaining heat

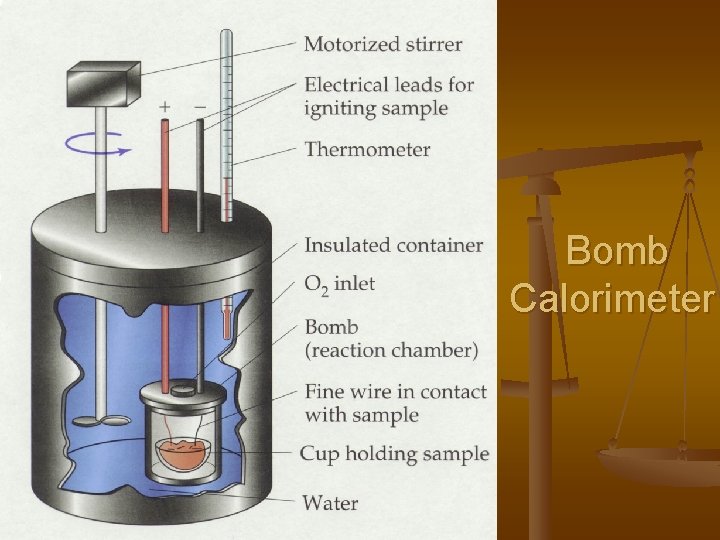
Calorimetry To measure specific heat capacity a bomb calorimeter is used. Calorimetry is based on the principle: Law of Conservation of Energy

Bomb Calorimeter

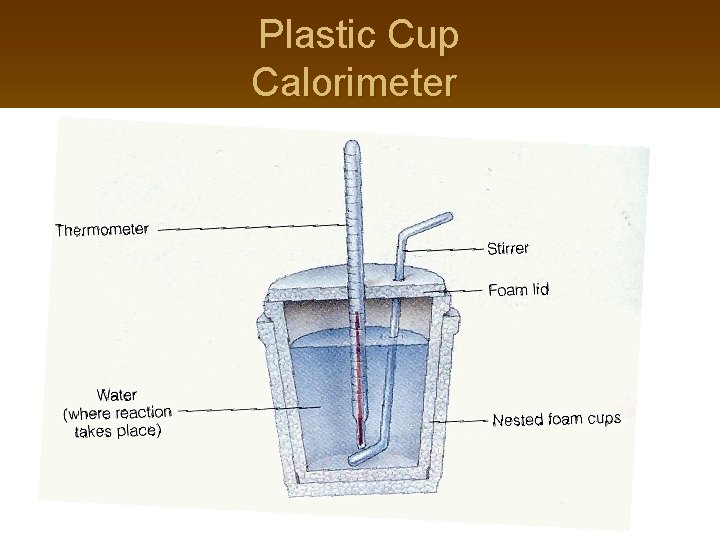
Plastic Cup Calorimeter