Thermochemistry Thermo heat Chemistry study of matter Thermochemistry



















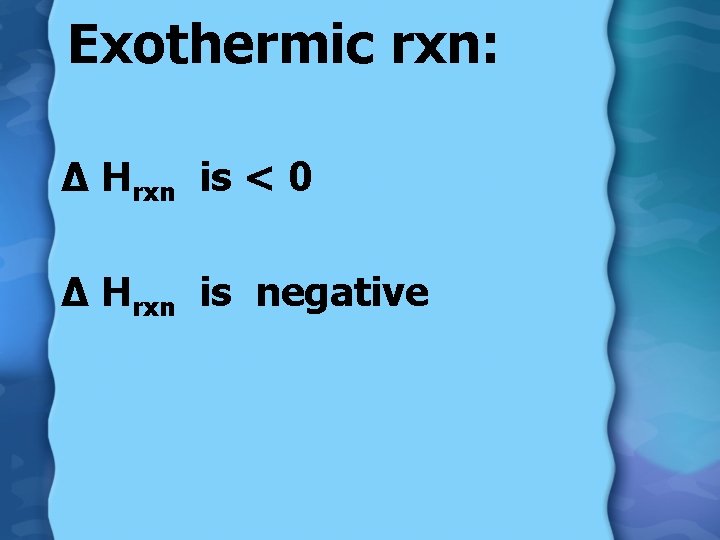

- Slides: 24

Thermochemistry Thermo = heat Chemistry = study of matter

Thermochemistry • Study of heat changes and energy that accompany chemical reactions and phase changes

Review of Energy • Capacity to do work or to create heat and or generate electricity • Types: • Chemical • Nuclear • Thermal • Radiant (light) • Electrical • Mechanical

Law of conservation of Energy • Energy can be converted from one form to another but it cannot be created or destroyed

Forms of energy: • Potential – Stored energy • Kinetic – Energy of motion

Chemical potential energy • Energy stored within the structural units of chemical compounds

Heat is energy (Q) • flows from an area of high heat to an area of low heat • Can be measured in calories, Calories, or Joules • 1000 cal = 1 Cal = 4. 184 J

Heat energy (Q) • System: part of the universe being studied • Surroundings: everything outside of the system • Universe: system + surroundings

Heat energy (Q) Endothermic: • feels cold to touch • temperature decreases • heat absorbed by system • Q = +

Heat energy (Q) Exothermic: • feels warm to touch • temperature increases • heat released by system • Q = -

Specific heat capacity • C or Cp • The amount of energy required to raise the temperature of 1 gram of a substance 1ºC • Water= 4. 18 J/g 0 C • Glass=. 50 J/g 0 C

Specific heat capacity • Measure of a substance’s resistance to temperature change • High=resistant (water) • Low=easy (metals, glass, etc. )

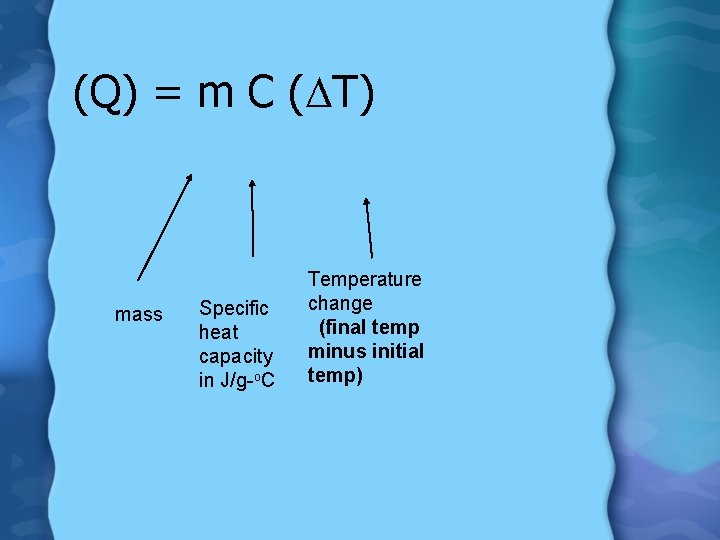
Equation Q = m C ∆T - Q is the heat absorbed or released in Joules -m is mass in grams -C is the specific heat in J/g/ºC -∆T is the temperature change in ºC

(Q) = m C (DT) mass Specific heat capacity in J/g-o. C Temperature change (final temp minus initial temp)

When 1982 g of water underwent a temperature change from 23. 677 o. C to 27. 482 o. C, how much energy in k. J did the water absorb? The specific heat of water is 4. 184 J/(go. C). Show all work!

How much energy in J is required to raise the temperature of 500. 0 g of copper from 22. 8 o. C to 100. 0 o. C? The specific heat of copper is 0. 387 J/g/o. C. Show all work!

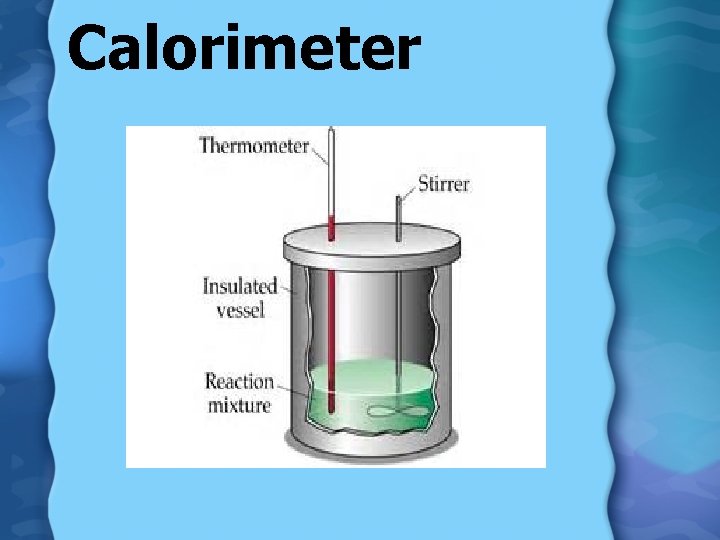
Measuring heat exchange Calorimeterinsulated device used for measuring the amount of heat absorbed or released during a reaction

Calorimeter

Calorimeter In a closed system: Heat loss = Heat gain Q lost = Q gained m. C ∆T = m. C∆T

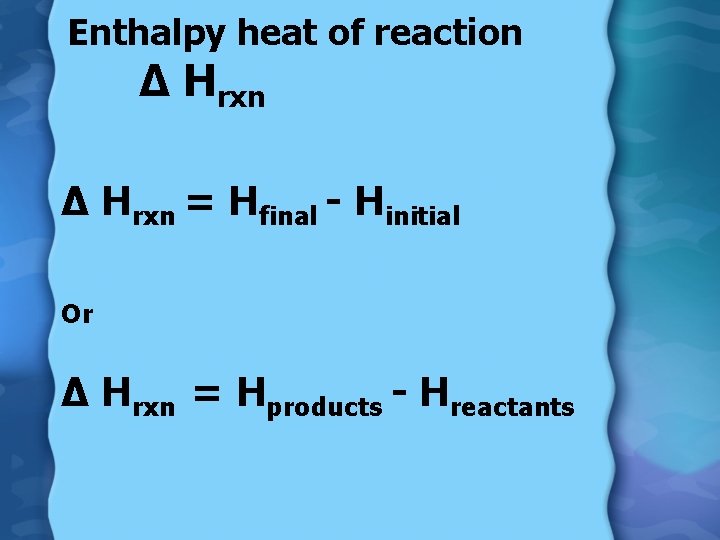
Enthalpy • (∆H) heat content of a system at constant pressure • Can measure changes in enthalpy during a reaction

Enthalpy heat of reaction ∆ Hrxn = Hfinal - Hinitial Or ∆ Hrxn = Hproducts - Hreactants

Thermochemical Equations • Used to calculate and express heat changes. • The physical states of the reactants and products in this equation is important! • Example: CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(l) ∆H = -890. 2 k. J
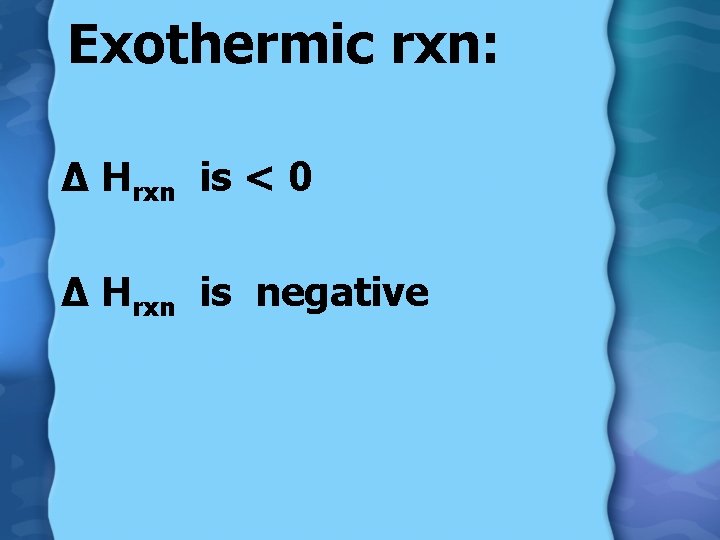
Exothermic rxn: ∆ Hrxn is < 0 ∆ Hrxn is negative

Endothermic rxn: ∆ Hrxn is > 0 ∆ Hrxn is positive