Chapter 11 Chemical Reactions 1 All chemical reactions












































































- Slides: 83

Chapter 11 Chemical Reactions 1

All chemical reactions have two parts l Reactants - the substances you start with l Products- the substances you end up with l The reactants turn into the products. l Reactants Products l 2

In a chemical reaction The way atoms are joined is changed l Atoms aren’t created or destroyed. l Can be described several ways l In a sentence – Copper reacts with chlorine to form copper (II) chloride. l In a word equation l Copper + chlorine copper (II) chloride l 3

Symbols used in equations Table 11. 1 l the arrow separates the reactants from the products l Read “reacts to form” l The plus sign = “and” l (s) after the formula -solid l (g) after the formula -gas l (l) after the formula -liquid l 4

Symbols used in equations (aq) after the formula - dissolved in water, an aqueous solution. l used after a product indicates a gas (same as (g)) l ¯ used after a product indicates a solid (same as (s)) l 5

Symbols used in equations indicates a reversible reaction (More later) l shows that heat is supplied to the reaction l is used to indicate a catalyst used in this case, platinum. l 6

What is a catalyst? A substance that speeds up a reaction without being changed by the reaction. l Enzymes are biological or protein catalysts. l 7

Skeleton Equation Uses formulas and symbols to describe a reaction l doesn’t indicate how many. l All chemical equations are sentences that describe reactions. l 8

Convert these to equations l 9 Solid iron (III) sulfide reacts with gaseous hydrogen chloride to form solid iron (II) chloride and hydrogen sulfide gas.

Convert these to equations l 10 Nitric acid dissolved in water reacts with solid sodium carbonate to form liquid water and carbon dioxide gas and sodium nitrate dissolved in water.

The other way l 11 Fe(g) + O 2(g) Fe 2 O 3(s)

The other way l 12 Cu(s) + Ag. NO 3(aq) Ag(s) + Cu(NO 3)2(aq)

Balancing Chemical Equations 13

Balanced Equation Atoms can’t be created or destroyed l All the atoms we start with we must end up with l A balanced equation has the same number of atoms of each element on both sides of the equation. l 14

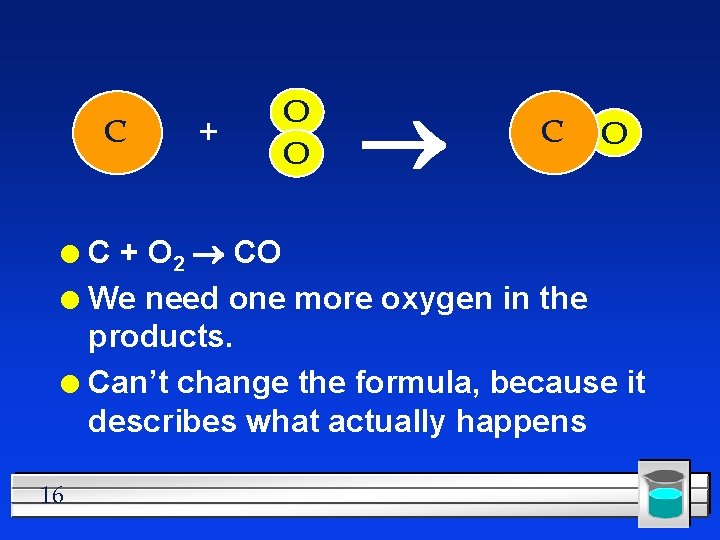
C + O O O C + O 2 CO 2 l This equation is already balanced l What if it isn’t already? l 15

C + O O C + O 2 CO l We need one more oxygen in the products. l Can’t change the formula, because it describes what actually happens l 16

C + O O C O Must be used to make another CO l But where did the other C come from? l Must have started with two C l 2 C + O 2 2 CO l 17

Rules for balancing 1 Write the correct formulas for all the reactants and products 2 Count the number of atoms of each type appearing on both sides 3 Balance the elements one at a time by adding coefficients (the numbers in front) 4 Check to make sure it is balanced. 18

Never Change a subscript to balance an equation. – If you change the formula you are describing a different reaction. – H 2 O is a different compound than H 2 O 2 l Never put a coefficient in the middle of a formula – 2 Na. Cl is okay, Na 2 Cl is not. l 19

Example 2 H 2 + O 2 H 22 H O 2 O R P 4 2 H 2 4 2 O 1 2 The equation Make Changes Also Need Recount changes twice a table theas. O to is the much keep balanced, HH O track in the has of where product reactant the same you number are at of each kind of atom on both sides 20

Example 2 H 2 + O 2 2 H 2 O R P 4 2 H 2 4 2 O 1 2 This is the answer Not this 21

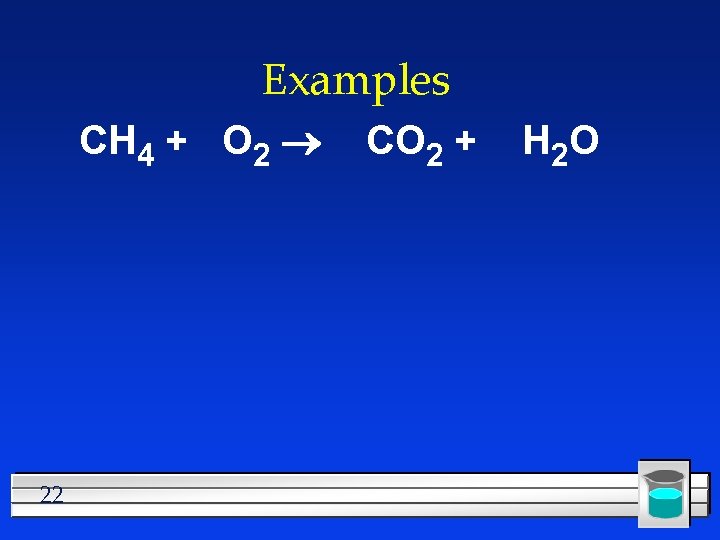
Examples CH 4 + O 2 22 CO 2 + H 2 O

Examples Ag. NO 3 + 23 Cu Cu(NO 3)2 + Ag

Examples Al + N 2 24 Al 2 N 3

Examples P+ 25 O 2 P 4 O 10

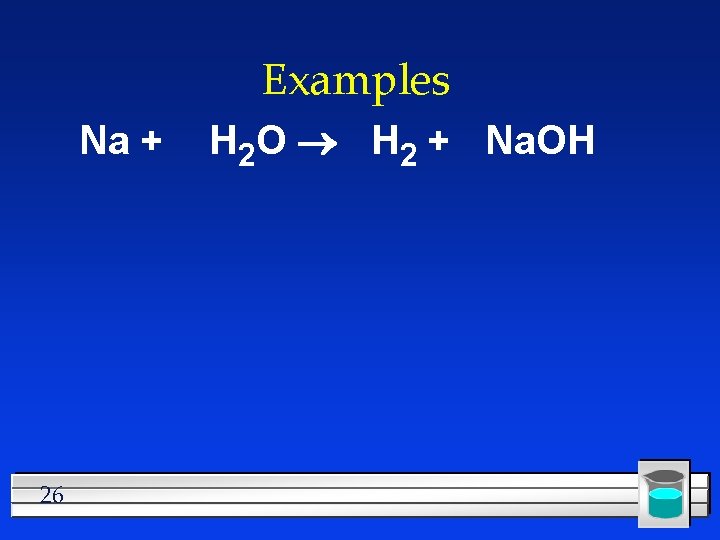
Examples Na + 26 H 2 O H 2 + Na. OH

Techniques If an atom appears more than once on a side, balance it last. l If you fix everything except one element, and it is even on one side and odd on the other, double the first number, then move on from there. l C 4 H 10 + O 2 CO 2 + H 2 O l 27 27

Types of Reactions Predicting the Products 28

Types of Reactions There are too many reactions to remember l Fall into categories. l We will learn 5 types. l Will be able to predict the products. l For some we will be able to predict whether they will happen at all. l Must recognize them by the reactants l 29

#1 Combination Reactions Combine - put together l 2 elements, or compounds combine to make 1 compound. l Ca +O 2 Ca. O l SO 3 + H 2 O H 2 SO 4 l We can predict the products if they are two elements. l Mg + N 2 l 30

Write and balance l 31 Ca + Cl 2

Write and balance l 32 Fe + O 2 iron (II) oxide

Write and balance l l Al + O 2 Remember that the first step is to write the formula Then balance l Also called synthesis reaction l 33

Combining two compounds If they tell you it is combination, you will make one product l Two compounds will make a polyatomic ion. l CO 2 + H 2 O → l H 2 O + Cl 2 O 7→ l 34 34

#2 Decomposition Reactions l decompose = fall apart l one reactant falls apart into two or more elements or compounds. l Na. Cl l Ca. CO 3 35 Na + Cl 2 Ca. O + CO 2

#2 Decomposition Reactions Can predict the products if it is a binary compound l Made up of only two elements l Falls apart into its elements l l 36 H 2 O

#2 Decomposition Reactions l 37 Hg. O

#2 Decomposition Reactions If the compound has more than two elements you must be given one of the products l The other product will be from the missing pieces l Ni. CO 3 Ni. O + l H 2 CO 3(aq) CO 2 + l 38

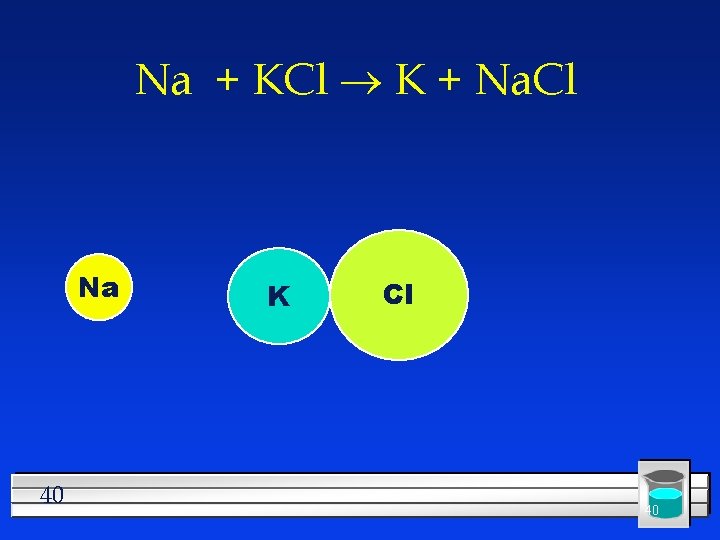
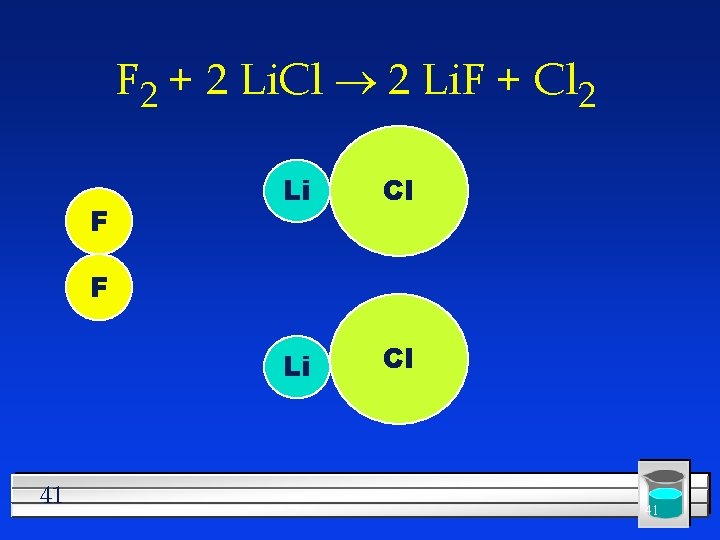
#3 Single Replacement One element replaces another l Reactants must be an element and a compound. l Products will be a different element and a different compound. l Na + KCl K + Na. Cl l F 2 + Li. Cl Li. F + Cl 2 l 39

Na + KCl ® K + Na. Cl Na 40 K Cl 40

F 2 + 2 Li. Cl ® 2 Li. F + Cl 2 F Li Cl F 41 41

#3 Single Replacement Metals replace metals (and hydrogen) l Al + Cu. SO 4 l Zn + H 2 SO 4 l Think of water as HOH l Metals replace one of the H, combine with hydroxide. l Na + HOH l 42

#3 Single Replacement We can tell whether a reaction will happen l Some are more active than other l More active replaces less active l There is a list on page 333 l 43

#3 Single Replacement There is a list on page 333 l Higher on the list replaces lower. l If the element by itself is higher, it happens, l if element by itself is lower, it doesn’t l 44

#3 Single Replacement l Note the * H can be replaced in acids by everything higher l Only the first 4 (Li - Na) react with water. l 45

l 46 #3 Single Replacement Al + HCl

l 47 #3 Single Replacement Fe + Cu. SO 4

l 48 #3 Single Replacement Pb + KCl

l 49 #3 Single Replacement Al + H 2 O

#3 Single Replacement l 50 What does it mean that Ag is on the bottom of the list?

#3 Single Replacement Nonmetals can replace other nonmetals l Limited to F 2 , Cl 2 , Br 2 , I 2 l The order of activity is that on the table. l Higher replaces lower. l F 2 + HCl l Br 2 + KCl l 51

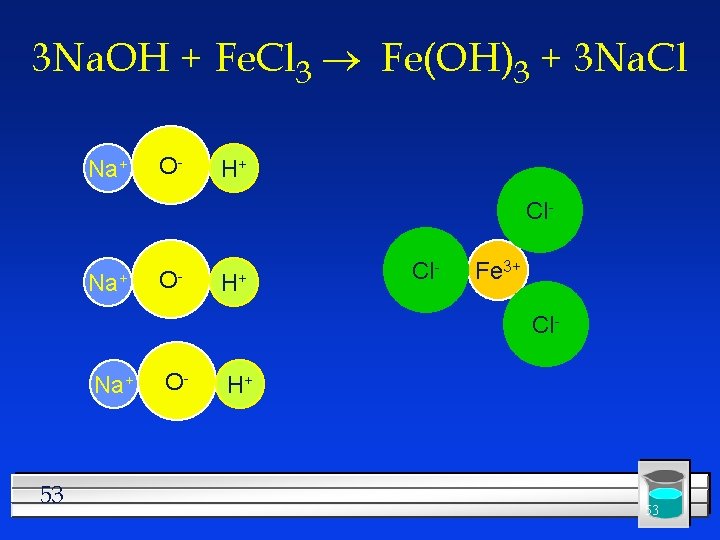
#4 Double Replacement Two things replace each other. l Reactants must be two ionic compounds or acids. l Usually in aqueous solution l Na. OH + Fe. Cl 3 l The positive ions change place. l Na. OH + Fe. Cl 3 Fe 3+OH- + Na+Cll Na. OH + Fe. Cl 3 Fe(OH)3 + Na. Cl l 52

3 Na. OH + Fe. Cl 3 ® Fe(OH)3 + 3 Na. Cl Na+ O- H+ Cl- Fe 3+ Cl- Na+ 53 O- H+ 53

#4 Double Replacement Will only happen if one of the products – doesn’t dissolve in water and forms a solid – or is a gas that bubbles out. – or is a covalent compound usually water. l Polyatomic ions don’t change from side to side l 54

Complete and balance assume all of the reactions take place. l Ca. Cl 2 + Na. OH l Cu. Cl 2 + K 2 S l KOH + Fe(NO 3)3 l 55

Complete and balance l KOH + Fe(NO 3)3 l H 3 PO 4 + Ca(OH)2 56 56

How to recognize which type Look at the reactants l E for element l C for compound l. E+E Combination l. C Decomposition l E + C Single replacement l C + C Double replacement l 57

Last Type Combustion l A compound composed of only C H and maybe O is reacted with oxygen l If the combustion is complete, the products will be CO 2 and H 2 O. l If the combustion is incomplete, the products will be CO and H 2 O. l or just C and H 2 O. l O 2 will always be the second reactant l 58

Examples l Complete combustion of C 4 H 10 l Incomplete combustion of C 4 H 10 59

Examples l Complete combustion of C 6 H 12 O 6 l Incomplete combustion of C 2 H 6 O 60

Ionic Compounds and acids Fall apart into ions when they dissolve l That’s why they conduct electricity when dissolved. l So when we write them as (aq) they are really separated l Na. Cl(aq) is really Na+(aq) and Cl-(aq) l K 2 SO 4 (aq) is really K+(aq) and SO 42 -(aq) l 61 61

Reactions in aqueous solutions Many reactions happen in solution l Makes it so the ions separate so they can interact. l Solids, liquids, and gases are not separated, only aqueous l 62 62

Complete Ionic Equation Every aqueous compound is written as separate ions l Solids, liquids and gases as whole compounds l Mg. Cl 2(aq) + Pb. SO 4(aq) → Mg. SO 4(aq) + Pb. Cl 2(s) l Is really l Mg 2+(aq) + Cl-(aq) + Pb 2+(aq) + SO 4(aq) → Mg 2+(aq) + SO 4(aq) + Pb. Cl 2(s) l 63 63

Write the complete ionic equation for 3+ -(aq) Br(aq) l Fe Fe. Br 3(aq) + +(aq) -(aq) → OH +K KOH(aq) +(aq) -(aq) + Fe(OH) (s) K Br KBr 3 + + 64 → + + 64

Write the complete ionic equation for l Ca. Cl 2(aq) + Mg. SO 4(aq) → Ca. SO 4(s) + Mg. Cl 2(aq) 65 65

Write the complete ionic equation for l Ba(OH)2(aq) + H 2 SO 4(aq) → Ba. SO 4(s) + HOH(l) 66 66

The complete ionic equation is Fe 3+(aq)+ Br-(aq) + K+(aq) +OH-(aq) → K+(aq) +Br-(aq) + Fe(OH)3(s) l K+ and Br- don’t change. l They are spectator ions l Could be eliminated l Fe 3+(aq) +OH-(aq) →Fe(OH)3(s) l This is what really changes l 67 67

Net ionic equation Shows only those particles that change before and after. l Eliminate spectator ions l Needs to be balanced in terms of both mass and charge l Fe 3+(aq) +OH-(aq) →Fe(OH)3(s) l Fe 3+(aq) +3 OH-(aq) →Fe(OH)3(s) l 68 68

Write the net ionic equation l 69 HCl (aq) + Ba(OH)2 (aq) → Ba. Cl 2(s) + HOH (l) 69

Write the net ionic equation l 70 Al + Fe. SO 4(aq) → Al 2(SO 4)3(aq) + Fe 70

Write the net ionic equation l 71 Cl 2(s) + Na. I(aq) → Na. Cl(aq) + I 2(s) 71

Write the net ionic equation l 72 K 2 CO 3(aq) + Mg. I 2(aq) → Mg. CO 3(s) + KI(aq) 72

Net ionic equations l 73 Written for single and double replacement. 73

Predicting precipitates Solids formed from aqueous solution. l You can predict them if you know some general rules for solubility. l 74 74

These things are soluble 1. Salts with alkali metals and ammonium 2. Salts of nitrates and chlorates 3. Salts of sulfates except Ag+, Pb 2+, Hg 22+, Ba 2+, and Sr 2+ 4. Salts of chlorides except Ag+, Pb 2+, and Hg 22+ 75 75

These things are insoluble 5. Carbonates, phosphates, chromates, sulfides, and hydroxides l Unless they fall under rule # 1 76 76

Is it soluble? Li. Br l Ba(NO 3)2 l Ca. SO 4 l Pb. Cl 2 l Ca. CO 3 l K 2 CO 3 l Cd(Cl. O 3)2 l 77 77

Is there a reaction? For double replacement- has to make gas, solid or water. l Water from an acid- H+ and a hydroxide- OH- makes HOH l Solids- from solubility rules l Exchange ions and see if something is insoluble l 78 78

Is there a reaction? l Mg. SO 4 + Na. OH → l H 2 SO 4 + KOH → l K 3 PO 4 + Fe. F 3→ 79 79

Chapter 7 Summary 80

An equation Describes a reaction l Must be balanced to follow the Law of Conservation of Mass l Can only be balanced by changing the coefficients. l Has special symbols to indicate state, and if catalyst or energy is required. l 81

Reactions Come in 5 types. l Can tell what type they are by the reactants. l Single Replacement happens based on the activity series l Double Replacement happens if the product is a solid, water, or a gas. l 82

The Process 1. Determine the type by looking at the reactants. 2. Put the pieces next to each other based on type 3. Use charges to write the formulas – Elements get 2? 4. Use coefficients to balance the equation. 83