Chapter 23 Chemical Reactions 1 All chemical reactions



















































- Slides: 54

Chapter 23 Chemical Reactions 1

All chemical reactions l l l 2 A chemical reaction is a change in which one or more substances are converted into new substances. Reactants - the substances you start with and are located on the left side of the reaction arrow Products - the substances you end up with and are located on the right side of the reaction arrow

What occurs when a chemical reaction takes place? Heat is produced/removed l Light is produced l Bubbles are produced l 3 3

Father of Modern Chemistry l l l The French chemist Antoine Lavoisier established that the total mass of the products always equals the total mass of the reactants. This is known as The Law of Conservation of Mass Lavoisier is known today as the father of modern chemistry for his more accurate explanation of the conservation of mass and for describing a common type of chemical reaction called combustion. 4 4

In a chemical reaction l 5 A chemical equation is a way to describe a chemical reaction using chemical formulas and other symbols.

What are coefficients? l l A coefficient is a number used to balance equations in order to maintain the Law of Conservation of Mass. It is placed IN FRONT of a compound and is distributed to all atoms within the compound. 6 6

What is a balanced equation? l l l The formulas in a chemical equation must accurately represent the compounds that react. Fixing this equation requires a process called balancing. The balancing process involves changing coefficients in a reaction to achieve a balanced chemical equation, which has the same number of atoms of each element on both sides of the equation. 7 7

Balancing Equations l http: //www. bozemanscience. com/be ginners-guide-balancing-equations l 8 8

Steps for Balancing l Step 1 Write a chemical equation for the reaction using formulas and symbols. l Step 2 Count the atoms in reactants and products. If polyatomic ions are on both sides of the arrow…leave it together! Mg O 2 Mg O 1 2 1 1 l l 9 9

Steps for Balancing Step 3 Choose coefficients that balance the equation. l Remember, never change subscripts of a correct formula to balance an equation. Coefficients go IN FRONT of compounds and get distributed to the atoms in the compound. l 10 10

Steps for Balancing l Step 4 Recheck the numbers of each atom on each side of the equation and adjust coefficients again if necessary. 11 11

Again…. . Never!! Change a subscript to balance an equation. – If you change the formula you are describing a different reaction. – H 2 O is a different compound than H 2 O 2 l Never put a coefficient in the middle of a formula – 2 Na. Cl is okay, Na 2 Cl is not. l 12

Example 2 H 2 + O 2 2 H 2 O R P 4 2 H 2 4 2 O 1 2 This is the answer Not this 13

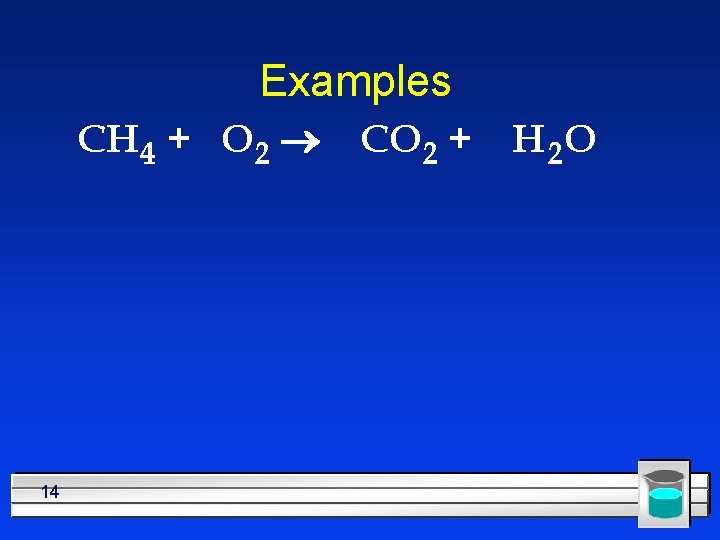
Examples CH 4 + O 2 14 CO 2 + H 2 O

Examples Ag. NO 3 + 15 Cu Cu(NO 3)2 + Ag

Techniques If an atom appears more than once on a side, balance it last. l If you fix everything except one element, and it is even on one side and odd on the other, double the first number, then move on from there. l C 4 H 10 + O 2 CO 2 + H 2 O l 16 16

Types of Chemical Reactions l 17 https: //www. youtube. com/watch? v= g-bi. Rw. AVTV 8

Types of Reactions There are too many reactions to remember l Fall into categories. l We will learn 5 types. l Will be able to predict the products. l For some we will be able to predict whether they will happen at all. l Must recognize them by the reactants l 18

#1 Combustion Reaction A compound composed of only C H and maybe O is reacted with oxygen l. If the combustion is complete, the products will be CO 2 and H 2 O. l. If the combustion is incomplete, the products will be CO and H 2 O. lor just C and H 2 O. l. O 2 will always be the second reactant 19

#1 Combustion Reaction CH 4 + O 2 ______ + ______ l Write and Balance l 20 20

#2 Synthesis Reactions Combine - put together l 2 elements, or compounds combine to make 1 compound. l Ca +O 2 Ca. O l SO 3 + H 2 O H 2 SO 4 l We can predict the products if they are two elements. l Mg + N 2 l 21

Write and balance l 22 Al + O 2 l Remember that the first step is to write the formula l Then balance

#3 Decomposition Reactions 23 l decompose = fall apart l one reactant falls apart into two or more elements or compounds. l Na. Cl l Ca. CO 3 Na + Cl 2 Ca. O + CO 2

#3 Decomposition Reactions Can predict the products if it is a binary compound l Made up of only two elements l Falls apart into its elements l l 24 H 2 O

#3 Decomposition Reactions l 25 Hg. O

#4 Single Replacement One element replaces another l Reactants must be an element and a compound. l Products will be a different element and a different compound. l Na + KCl K + Na. Cl l F 2 + Li. Cl Li. F + Cl 2 l 26

#4 Single Replacement Metals replace metals (and hydrogen) l Al + Cu. SO 4 l Zn + H 2 SO 4 l Think of water as HOH l Metals replace one of the H, combine with hydroxide. l Na + HOH l 27

#4 Single Replacement We can tell whether a reaction will happen l Some are more active than other l More active replaces less active l l l 28 The activity series chart lets us Know whether or not a reaction will occur

#4 Single Replacement There is a list on page 732 l Higher on the list replaces lower. l If the element by itself is higher, it happens, l if element by itself is lower, it doesn’t l 29

#4 Single Replacement l Note the * H can be replaced in acids by everything higher l Only the first 4 (Li - Na) react with water. l 30

l 31 #4 Single Replacement Al + HCl

l 32 #4 Single Replacement Pb + KCl

#4 Single Replacement l 33 What does it mean that Ag is on the bottom of the list?

#4 Single Replacement Nonmetals can replace other nonmetals l Limited to F 2 , Cl 2 , Br 2 , I 2 l The order of activity is that on the periodic table. l Higher replaces lower. l F 2 + HCl l Br 2 + KCl l 34

#5 Double Replacement Two things replace each other. l Reactants must be two ionic compounds or acids. l Usually in aqueous solution l Na. OH + Fe. Cl 3 l The positive ions change place. l Na. OH + Fe. Cl 3 Fe 3+ OH- + Na +Cll Na. OH + Fe. Cl 3 Fe(OH)3 + Na. Cl l 35

#5 Double Replacement Will only happen if one of the products – doesn’t dissolve in water and forms a solid – or is a gas that bubbles out. – or is a covalent compound usually water. l Polyatomic ions don’t change from side to side l 36

Complete and balance assume all of the reactions take place. l Ca. Cl 2 + Na. OH l Cu. Cl 2 + K 2 S l KOH + Fe(NO 3)3 l 37

How to recognize which type 38

Rates of Reaction A dynamic explosion is an example of a rapid chemical reaction. l Most chemical reactions proceed more slowly, but all chemical reactions release or absorb energy. l 39 39

Rates of Reaction l l l As previously learned, atoms and molecules must bump into each other before a product can be formed. They also need a minimum amount of energy to kick start the reaction. This minimum amount of energy needed to start a reaction is called activation energy. 40 40

Rates of Reaction l 41 http: //ed. ted. com/lessons/activation -energy-kickstarting-chemicalreactions-vance-kite

Rates of Reaction When most chemical reactions take place, some chemical bonds in the reactants are broken, which requires energy. l In order for products to be produced, new bonds must form. Bond formation releases energy. l 42 42

Rates of Reaction Chemical reactions that release energy are called exergonic (ek sur GAH nihk) reactions. l In these reactions less energy is required to break the original bonds than is released when new bonds form. l l As a result, some form of energy, such as light or heat is given off by the reaction. 43 43

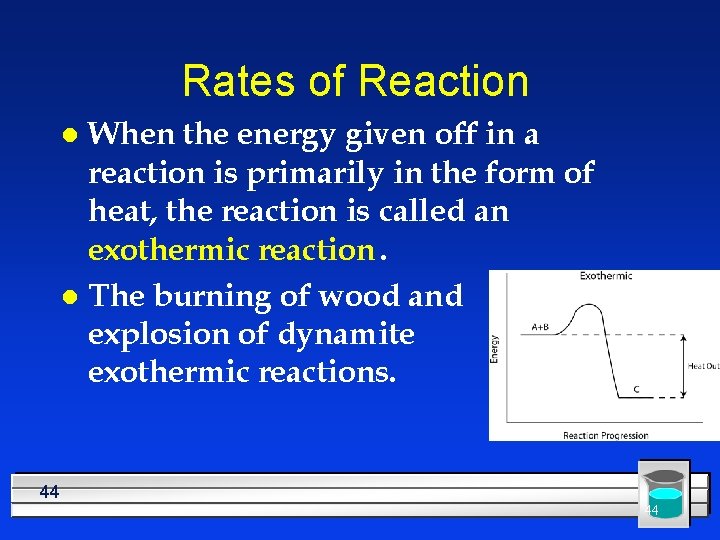
Rates of Reaction When the energy given off in a reaction is primarily in the form of heat, the reaction is called an exothermic reaction. l The burning of wood and the explosion of dynamite are exothermic reactions. l 44 44

Rates of Reaction Sometimes a chemical reaction requires more energy to break bonds than is released when new ones are formed. l These reactions are called endergonic reactions. l The energy absorbed can be in the form of light, heat or electricity. l 45 45

Rates of Reaction When the energy needed is in the form of heat, the reaction is called an endothermic reaction. l Some reactions are so endothermic that they can cause water to freeze. l 46 46

Rates of Reaction l l The rate of reaction is the speed at which reactants are consumed and products are produced in a given reaction. There are 6 things that affect rates of reaction: Temperature, Concentration, Surface Area, Agitation, Pressure and Catalysts/Inhibitors. 47 47

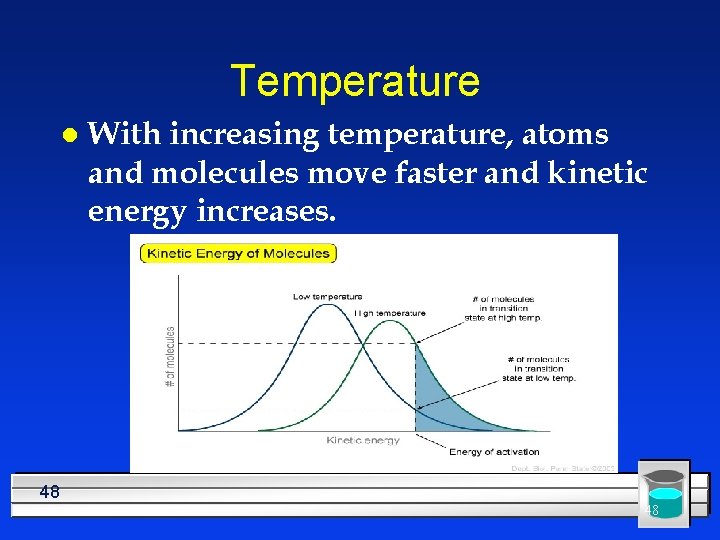
Temperature l With increasing temperature, atoms and molecules move faster and kinetic energy increases. 48 48

Concentration The closer atoms and molecules are to each other, the greater the chance of collision. l Increasing the amount (concentration) of a substance increases the chance of atoms/molecules bumping into each other increases the reaction rate. l 49 49

Surface Area l l When atoms/molecules bump into each other, only their surfaces collide. When a substance is divided into smaller pieces increases the surface area more collisions can occur increases the rate of reaction 50 50

Agitation is a fancy word for stirring l Agitation is a physical process that allows reactants to mix increasing collisions increasing rate of reaction l 51 51

Pressure l Increasing the pressure of gases, molecules have less room to move around increases the concentration increases the amount of collisions increases the rate of reactions 52 52

Catalysts and Inhibitors Some reactions proceed too slowly to be useful. l To speed them up, a catalyst can be added. l A catalyst is a substance that speeds up a chemical reaction without being permanently changed itself. l 53 53

Catalysts and Inhibitors l l l At times, it is worthwhile to prevent certain reactions from occurring. Substances called inhibitors are used to slow down a chemical reaction. One thing to remember when thinking about catalysts and inhibitors is that they do not change the amount of product produced. They only change the rate of production. 54 54