General Chemistry Principles and Modern Applications Petrucci Harwood







![Example 15 -2 What is the concentration at 100 s? [H 2 O 2]i Example 15 -2 What is the concentration at 100 s? [H 2 O 2]i](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-9.jpg)


![Example 15 -3 R 3 = k [Hg. Cl 2]3 m [C 2 O Example 15 -3 R 3 = k [Hg. Cl 2]3 m [C 2 O](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-13.jpg)
![Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42 Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-14.jpg)
![Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42 Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-15.jpg)
![15 -4 Zero-Order Reactions A → products Rrxn = k [A]0 Rrxn = k 15 -4 Zero-Order Reactions A → products Rrxn = k [A]0 Rrxn = k](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-16.jpg)
![Integrated Rate Law -Δ[A] Δt = k Move to the -d[A] infinitesimal dt = Integrated Rate Law -Δ[A] Δt = k Move to the -d[A] infinitesimal dt =](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-17.jpg)




![Testing for a Rate Law Plot [A] vs t. Plot ln[A] vs t. Plot Testing for a Rate Law Plot [A] vs t. Plot ln[A] vs t. Plot](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-26.jpg)














![The Steady State Approximation d[N 2 O 2] dt = k 1[NO]2 – k The Steady State Approximation d[N 2 O 2] dt = k 1[NO]2 – k](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-47.jpg)
![Kinetic Consequences of Assumptions 2 NO(g) d[NO 2] dt = k 1 k 3[NO]2[O Kinetic Consequences of Assumptions 2 NO(g) d[NO 2] dt = k 1 k 3[NO]2[O](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-48.jpg)

![Saturation Kinetics k 1 d[P] k 2 E + S ES → E + Saturation Kinetics k 1 d[P] k 2 E + S ES → E +](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-53.jpg)
![Michaelis-Menten d[P] dt d[P] = k 2[E]0 dt d[P] dt = = k 1 Michaelis-Menten d[P] dt d[P] = k 2[E]0 dt d[P] dt = = k 1](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-54.jpg)

- Slides: 55

General Chemistry Principles and Modern Applications Petrucci • Harwood • Herring 8 th Edition Chapter 15: Chemical Kinetics Philip Dutton University of Windsor, Canada N 9 B 3 P 4 Prentice-Hall © 2002

Contents 15 -1 15 -2 15 -3 15 -4 15 -5 15 -6 15 -7 Prentice-Hall © 2002 The Rate of a Chemical Reaction Measuring Reaction Rates Effect of Concentration on Reaction Rates: The Rate Law Zero-Order Reactions First-Order Reactions Second-Order Reactions Reaction Kinetics: A Summary General Chemistry: Chapter 15 Slide 2 of 55

Contents 15 -8 15 -9 15 -10 15 -11 Theoretical Models for Chemical Kinetics The Effect of Temperature on Reaction Rates Reaction Mechanisms Catalysis Focus On Combustion and Explosions Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 3 of 55

15 -1 The Rate of a Chemical Reaction • Rate of change of concentration with time. 2 Fe 3+(aq) + Sn 2+ → 2 Fe 2+(aq) + Sn 4+(aq) t = 38. 5 s Δt = 38. 5 s [Fe 2+] = 0. 0010 M Δ[Fe 2+] = (0. 0010 – 0) M Rate of formation of Fe 2+= Prentice-Hall © 2002 Δ[Fe 2+] Δt = 0. 0010 M General Chemistry: Chapter 15 38. 5 s = 2. 6 10 -5 M s-1 Slide 4 of 55

Rates of Chemical Reaction 2 Fe 3+(aq) + Sn 2+ → 2 Fe 2+(aq) + Sn 4+(aq) 1 Δ[Fe 3+] Δ[Sn 4+] 1 Δ[Fe 2+] = = Δt Δt 2 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 5 of 55

General Rate of Reaction a. A+b. B→c. C+d. D Rate of reaction = rate of disappearance of reactants 1 Δ[B] 1 Δ[A] ==b Δt a Δt = rate of appearance of products 1 Δ[D] 1 Δ[C] = = d Δt c Δt Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 6 of 55

15 -2 Measuring Reaction Rates H 2 O 2(aq) → H 2 O(l) + ½ O 2(g) 2 Mn. O 4 -(aq) + 5 H 2 O 2(aq) + 6 H+ → 2 Mn 2+ + 8 H 2 O(l) + 5 O 2(g) Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 7 of 55

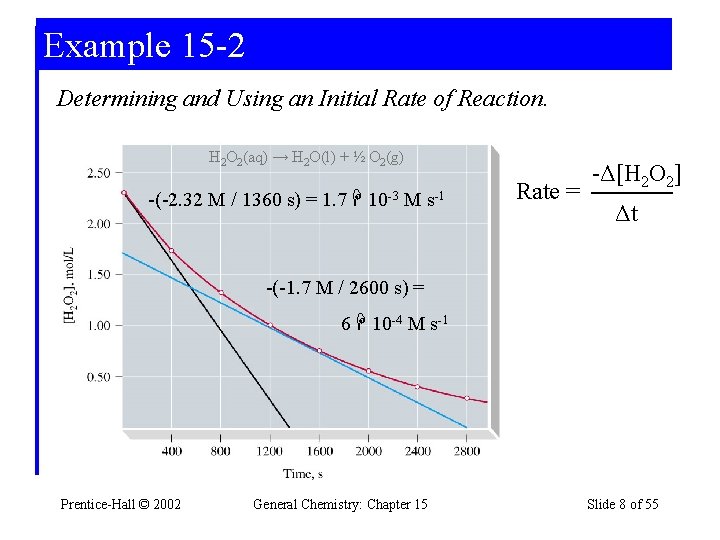
Example 15 -2 Determining and Using an Initial Rate of Reaction. H 2 O 2(aq) → H 2 O(l) + ½ O 2(g) -(-2. 32 M / 1360 s) = 1. 7 10 -3 M s-1 Rate = -Δ[H 2 O 2] Δt -(-1. 7 M / 2600 s) = 6 10 -4 M s-1 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 8 of 55
![Example 15 2 What is the concentration at 100 s H 2 O 2i Example 15 -2 What is the concentration at 100 s? [H 2 O 2]i](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-9.jpg)
Example 15 -2 What is the concentration at 100 s? [H 2 O 2]i = 2. 32 M Rate = 1. 7 10 -3 M s-1 = - Δ[H 2 O 2] Δt -Δ[H 2 O 2] = -([H 2 O 2]f - [H 2 O 2]i) = 1. 7 10 -3 M s-1 Δt [H 2 O 2]100 s – 2. 32 M = -1. 7 10 -3 M s-1 100 s [H 2 O 2]100 s = 2. 32 M - 0. 17 M = 2. 17 M Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 9 of 55

15 -3 Effect of Concentration on Reaction Rates: The Rate Law a A + b B …. → g G + h H …. Rate of reaction = k [A]m[B]n …. Rate constant = k Overall order of reaction = m + n + …. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 10 of 55

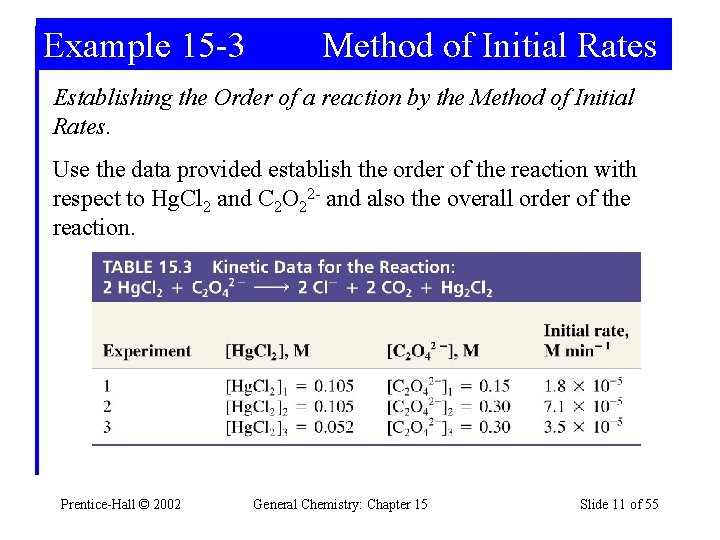
Example 15 -3 Method of Initial Rates Establishing the Order of a reaction by the Method of Initial Rates. Use the data provided establish the order of the reaction with respect to Hg. Cl 2 and C 2 O 22 - and also the overall order of the reaction. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 11 of 55

Example 15 -3 Notice that concentration changes between reactions are by a factor of 2. Write and take ratios of rate laws taking this into account. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 12 of 55
![Example 15 3 R 3 k Hg Cl 23 m C 2 O Example 15 -3 R 3 = k [Hg. Cl 2]3 m [C 2 O](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-13.jpg)
Example 15 -3 R 3 = k [Hg. Cl 2]3 m [C 2 O 42 -]3 n R 2 = k [Hg. Cl 2]2 m [C 2 O 42 -]2 n = k (2[Hg. Cl 2]3)m [C 2 O 42 -]3 n R 2 = R 3 k [Hg. Cl 2]3 m [C 2 O 42 -]3 n k 2 m [Hg. Cl 2]3 m [C 2 O 42 -]3 n R 2 2 m. R 3 = = = 2. 0 m 2 n R 3 k [Hg. Cl 2]3 [C 2 O 4 ]3 R 3 2 m = 2. 0 therefore m = 1. 0 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 13 of 55
![Example 15 3 R 2 k Hg Cl 221 C 2 O 42 Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-14.jpg)
Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42 -]2 n = k (0. 105) (0. 30)n R 1 = k [Hg. Cl 2]11 [C 2 O 42 -]1 n = k (0. 105) (0. 15)n k (0. 105) (0. 30)n R 2 = R 1 k (0. 105) (0. 15)n R 2 = R 1 (0. 30)n (0. 15)n = 2 n 7. 1 10 -5 = = 3. 94 -5 1. 8 10 2 n = 3. 98 therefore n = 2. 0 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 14 of 55
![Example 15 3 R 2 k Hg Cl 221 C 2 O 42 Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-15.jpg)
Example 15 -3 R 2 = k [Hg. Cl 2]21 [C 2 O 42 -]22 First order + Prentice-Hall © 2002 Second order General Chemistry: Chapter 15 = Third Order Slide 15 of 55
![15 4 ZeroOrder Reactions A products Rrxn k A0 Rrxn k 15 -4 Zero-Order Reactions A → products Rrxn = k [A]0 Rrxn = k](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-16.jpg)
15 -4 Zero-Order Reactions A → products Rrxn = k [A]0 Rrxn = k [k] = mol L-1 s-1 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 16 of 55
![Integrated Rate Law ΔA Δt k Move to the dA infinitesimal dt Integrated Rate Law -Δ[A] Δt = k Move to the -d[A] infinitesimal dt =](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-17.jpg)
Integrated Rate Law -Δ[A] Δt = k Move to the -d[A] infinitesimal dt = k And integrate from 0 to time t [A]t - d[A] [A]0 t = k dt 0 -[A]t + [A]0 = kt [A]t = [A]0 - kt Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 17 of 55

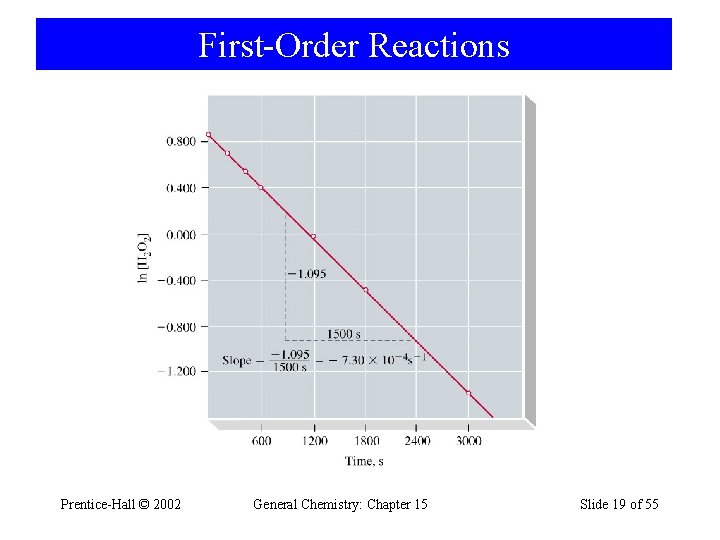
15 -5 First-Order Reactions H 2 O 2(aq) → H 2 O(l) + ½ O 2(g) d[H 2 O 2 ] = -k [H 2 O 2] dt [k] = s-1 [A]t t d[H 2 O 2 ] [H O ] = - k dt [A] 0 2 2 0 ln Prentice-Hall © 2002 [A]t [A]0 = -kt ln[A]t = -kt + ln[A]0 General Chemistry: Chapter 15 Slide 18 of 55

First-Order Reactions Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 19 of 55

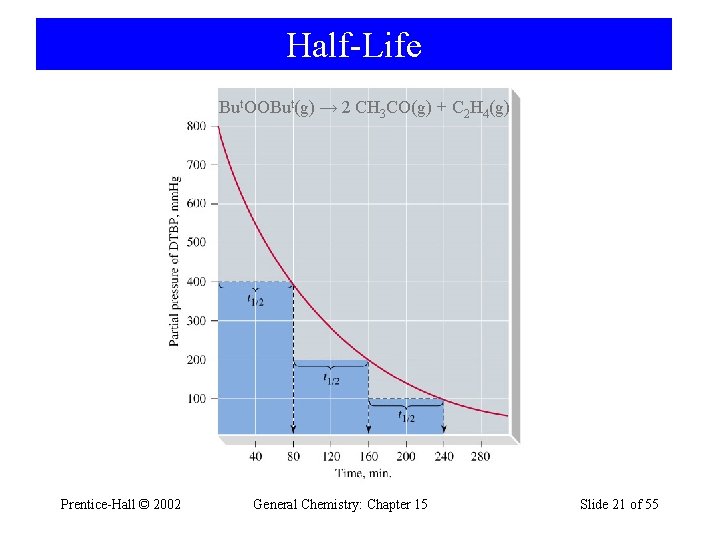
Half-Life • t½ is the time taken for one-half of a reactant to be consumed. ln ln [A]t [A]0 = -kt ½[A]0 = -kt½ [A]0 - ln 2 = -kt½ ln 2 0. 693 t½ = = k k Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 20 of 55

Half-Life But. OOBut(g) → 2 CH 3 CO(g) + C 2 H 4(g) Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 21 of 55

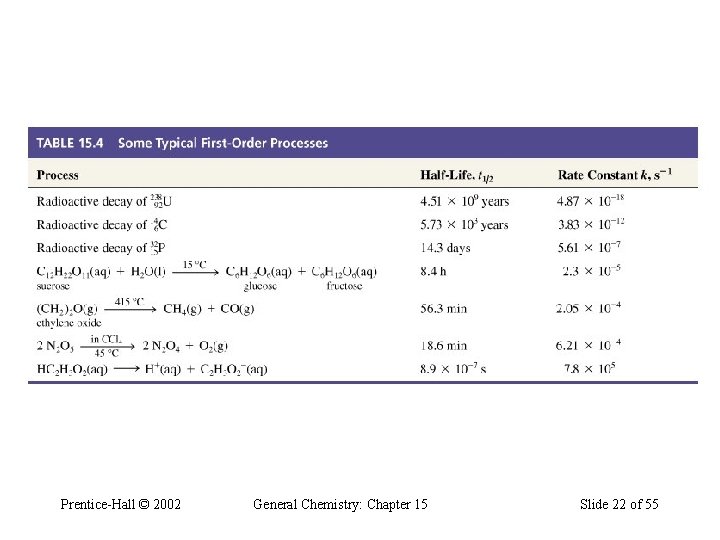
Some Typical First-Order Processes Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 22 of 55

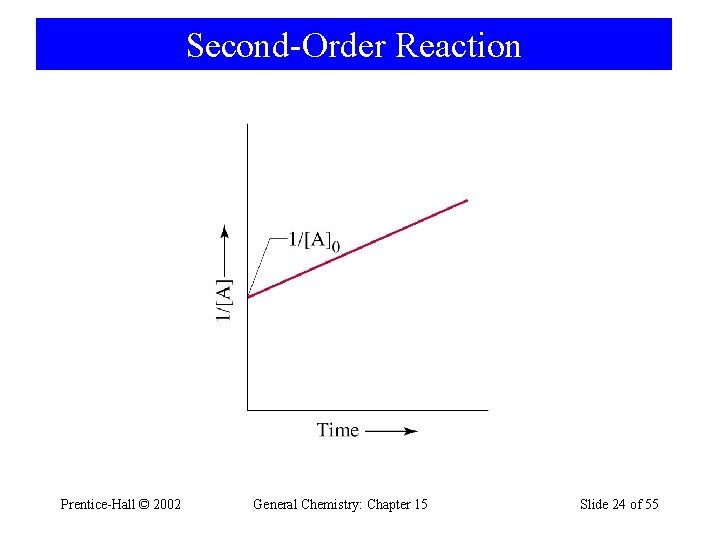
15 -6 Second-Order Reactions • Rate law where sum of exponents m + n +… = 2. A → products d[A] dt = -k[A]2 [A]t d[A] [A]2 [A]0 [k] = M-1 s-1 = L mol-1 s-1 t = - k dt 0 1 1 = kt + [A]t [A]0 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 23 of 55

Second-Order Reaction Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 24 of 55

Pseudo First-Order Reactions • Simplify the kinetics of complex reactions • Rate laws become easier to work with. CH 3 CO 2 C 2 H 5 + H 2 O → CH 3 CO 2 H + C 2 H 5 OH • If the concentration of water does not change appreciably during the reaction. – Rate law appears to be first order. • Typically hold one or more reactants constant by using high concentrations and low concentrations of the reactants under study. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 25 of 55
![Testing for a Rate Law Plot A vs t Plot lnA vs t Plot Testing for a Rate Law Plot [A] vs t. Plot ln[A] vs t. Plot](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-26.jpg)
Testing for a Rate Law Plot [A] vs t. Plot ln[A] vs t. Plot 1/[A] vs t. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 26 of 55

15 -7 Reaction Kinetics: A Summary • Calculate the rate of a reaction from a known rate law using: Rate of reaction = k [A]m[B]n …. • Determine the instantaneous rate of the reaction by: Finding the slope of the tangent line of [A] vs t or, Evaluate –Δ[A]/Δt, with a short Δt interval. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 27 of 55

Summary of Kinetics • Determine the order of reaction by: Using the method of initial rates. Find the graph that yields a straight line. Test for the half-life to find first order reactions. Substitute data into integrated rate laws to find the rate law that gives a consistent value of k. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 28 of 55

Summary of Kinetics • Find the rate constant k by: Determining the slope of a straight line graph. Evaluating k with the integrated rate law. Measuring the half life of first-order reactions. • Find reactant concentrations or times for certain conditions using the integrated rate law after determining k. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 29 of 55

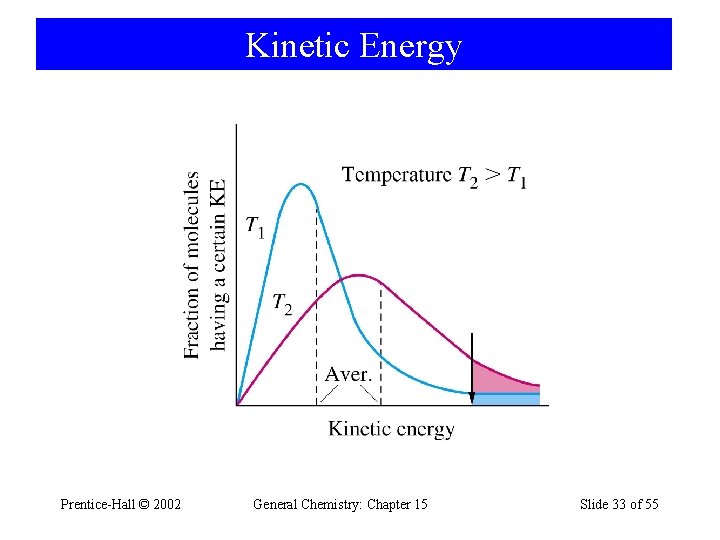
15 -8 Theoretical Models for Chemical Kinetics Collision Theory • Kinetic-Molecular theory can be used to calculate the collision frequency. – In gases 1030 collisions per second. – If each collision produced a reaction, the rate would be about 106 M s-1. – Actual rates are on the order of 104 M s-1. • Still a very rapid rate. – Only a fraction of collisions yield a reaction. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 30 of 55

Activation Energy • For a reaction to occur there must be a redistribution of energy sufficient to break certain bonds in the reacting molecule(s). • Activation Energy is: – The minimum energy above the average kinetic energy that molecules must bring to their collisions for a chemical reaction to occur. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 31 of 55

Activation Energy Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 32 of 55

Kinetic Energy Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 33 of 55

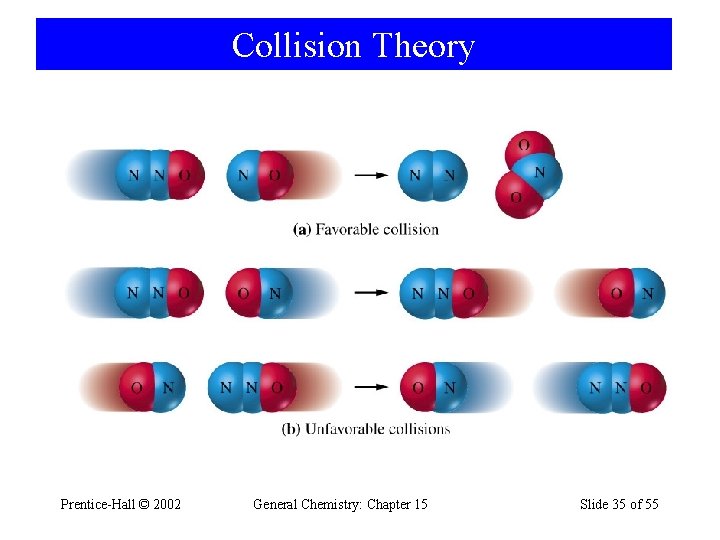
Collision Theory • If activation barrier is high, only a few molecules have sufficient kinetic energy and the reaction is slower. • As temperature increases, reaction rate increases. • Orientation of molecules may be important. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 34 of 55

Collision Theory Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 35 of 55

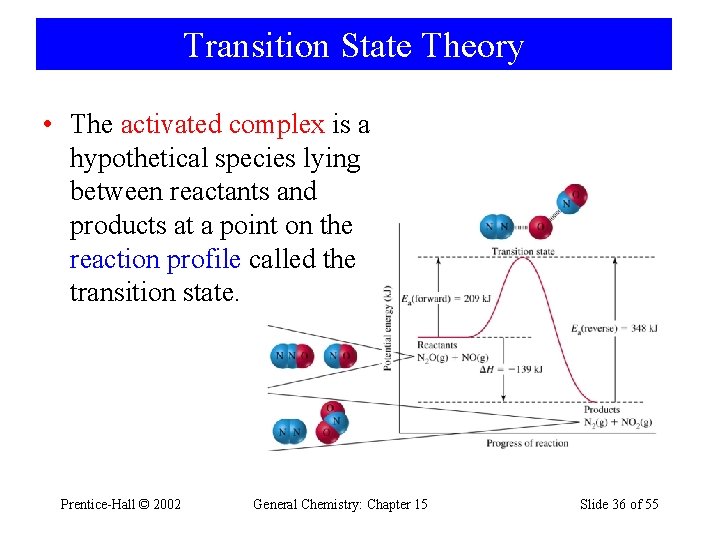
Transition State Theory • The activated complex is a hypothetical species lying between reactants and products at a point on the reaction profile called the transition state. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 36 of 55

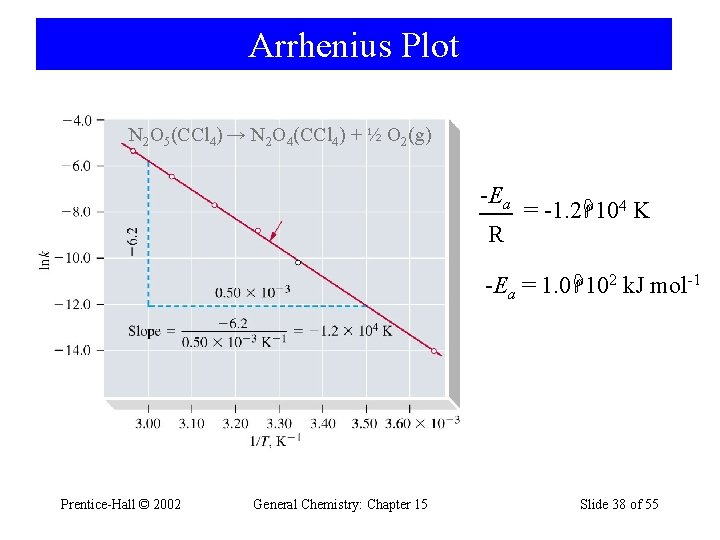
15 -9 Effect of Temperature on Reaction Rates • Svante Arrhenius demonstrated that many rate constants vary with temperature according to the equation: k = Ae-Ea/RT ln k = Prentice-Hall © 2002 -Ea 1 R T + ln A General Chemistry: Chapter 15 Slide 37 of 55

Arrhenius Plot N 2 O 5(CCl 4) → N 2 O 4(CCl 4) + ½ O 2(g) -Ea R = -1. 2 104 K -Ea = 1. 0 102 k. J mol-1 Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 38 of 55

Arrhenius Equation k= ln k 2– ln k 1 = ln Prentice-Hall © 2002 -Ea 1 ln k = Ae-Ea/RT k 1 k 2 R T + ln A -Ea 1 R = 1 -E a + ln A - ln A T 2 R T 1 -Ea 1 R T 2 - General Chemistry: Chapter 15 1 T 1 Slide 39 of 55

15 -10 Reaction Mechanisms • A step-by-step description of a chemical reaction. • Each step is called an elementary process. – Any molecular event that significantly alters a molecules energy of geometry or produces a new molecule. • Reaction mechanism must be consistent with: – Stoichiometry for the overall reaction. – The experimentally determined rate law. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 40 of 55

Elementary Processes • Unimolecular or bimolecular. • Exponents for concentration terms are the same as the stoichiometric factors for the elementary process. • Elementary processes are reversible. • Intermediates are produced in one elementary process and consumed in another. • One elementary step is usually slower than all the others and is known as the rate determining step. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 41 of 55

A Rate Determining Step Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 42 of 55

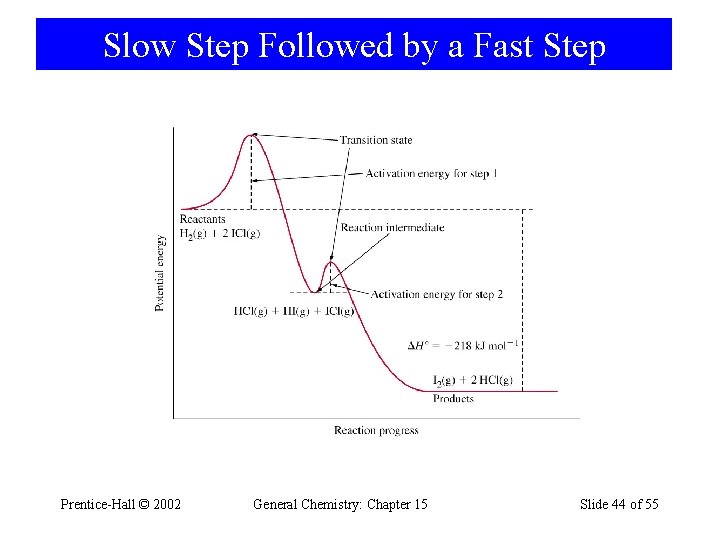
Slow Step Followed by a Fast Step H 2(g) + 2 ICl(g) → I 2(g) + 2 HCl(g) d[P] dt = k[H 2][ICl] Postulate a mechanism: H 2(g) + ICl(g) slow HI(g) + HCl(g) HI(g) + ICl(g) fast I 2(g) + HCl(g) H 2(g) + 2 ICl(g) → I 2(g) + 2 HCl(g) Prentice-Hall © 2002 General Chemistry: Chapter 15 d[HI] dt d[I 2] dt d[P] dt = k[H 2][ICl] = k[HI][ICl] = k[H 2][ICl] Slide 43 of 55

Slow Step Followed by a Fast Step Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 44 of 55

Fast Reversible Step Followed by a Slow Step 2 NO(g) + O 2(g) → 2 NO 2(g) d[P] dt = -kobs[NO 2]2[O 2] Postulate a mechanism: fast 2 NO(g) k 1 k -1 K= slow k 1 k-1 = N 2 O 2(g) [N 2 O 2] = k-1 [NO]2 = K [NO]2 [N 2 O 2] [NO] N 2 O 2(g) + O 2(g) k 2 2 NO 2(g) 2 NO(g) + O 2(g) → 2 NO 2(g) Prentice-Hall © 2002 k 1 General Chemistry: Chapter 15 d[NO 2] dt d[I 2] dt = k 2[N 2 O 2][O 2] = k 2 k 1 k-1 [NO]2[O 2] Slide 45 of 55

The Steady State Approximation 2 NO(g) k 1 k-1 N 2 O 2(g) + O 2(g) 2 NO(g) N 2 O 2(g) k 3 2 NO 2(g) d[NO 2] dt d[N 2 O 2] dt Prentice-Hall © 2002 N 2 O 2(g) k 1 k 2 N 2 O 2(g) + O 2(g) N 2 O 2(g) 2 NO(g) k 3 2 NO 2(g) = k 3[N 2 O 2][O 2] = k 1[NO]2 – k 2[N 2 O 2] – k 3[N 2 O 2][O 2] = 0 General Chemistry: Chapter 15 Slide 46 of 55
![The Steady State Approximation dN 2 O 2 dt k 1NO2 k The Steady State Approximation d[N 2 O 2] dt = k 1[NO]2 – k](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-47.jpg)
The Steady State Approximation d[N 2 O 2] dt = k 1[NO]2 – k 2[N 2 O 2] – k 3[N 2 O 2][O 2] = 0 k 1[NO]2 = [N 2 O 2](k 2 + k 3[O 2]) [N 2 O 2] = d[NO 2] dt Prentice-Hall © 2002 k 1[NO]2 (k 2 + k 3[O 2]) = k 3[N 2 O 2][O 2] = k 1 k 3[NO]2[O 2] General Chemistry: Chapter 15 (k 2 + k 3[O 2]) Slide 47 of 55
![Kinetic Consequences of Assumptions 2 NOg dNO 2 dt k 1 k 3NO2O Kinetic Consequences of Assumptions 2 NO(g) d[NO 2] dt = k 1 k 3[NO]2[O](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-48.jpg)
Kinetic Consequences of Assumptions 2 NO(g) d[NO 2] dt = k 1 k 3[NO]2[O 2] Let k 2 << k 3 (k 2 + k 3[O 2]) d[NO 2] dt Let k 2 >> k 3 Prentice-Hall © 2002 dt N 2 O 2(g) k 2 N 2 O 2(g) + O 2(g) = Or d[NO 2] k 1 = k 1 k 3[NO]2[O 2] ( k 3[O 2]) k 1 k 3[NO]2[O 2] ( k 2) General Chemistry: Chapter 15 2 NO(g) k 3 2 NO 2(g) = k 1[NO]2 = k 1 k 3 k 2 [NO]2[O 2] Slide 48 of 55

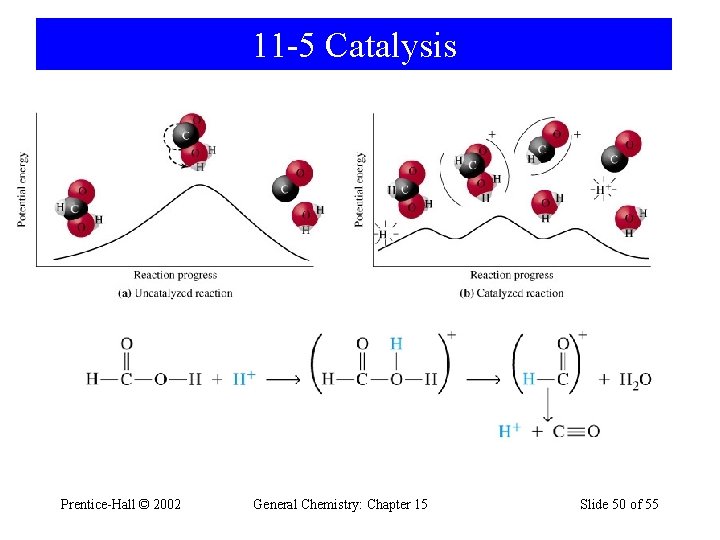
11 -5 Catalysis • Alternative reaction pathway of lower energy. • Homogeneous catalysis. – All species in the reaction are in solution. • Heterogeneous catalysis. – The catalyst is in the solid state. – Reactants from gas or solution phase are adsorbed. – Active sites on the catalytic surface are important. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 49 of 55

11 -5 Catalysis Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 50 of 55

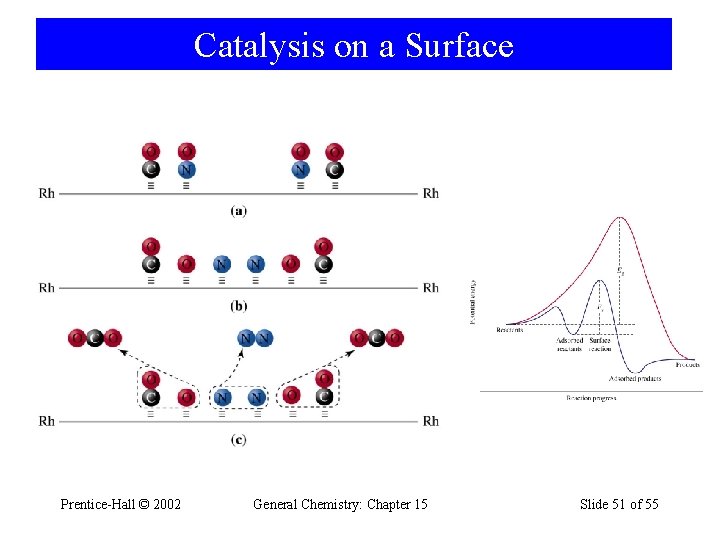
Catalysis on a Surface Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 51 of 55

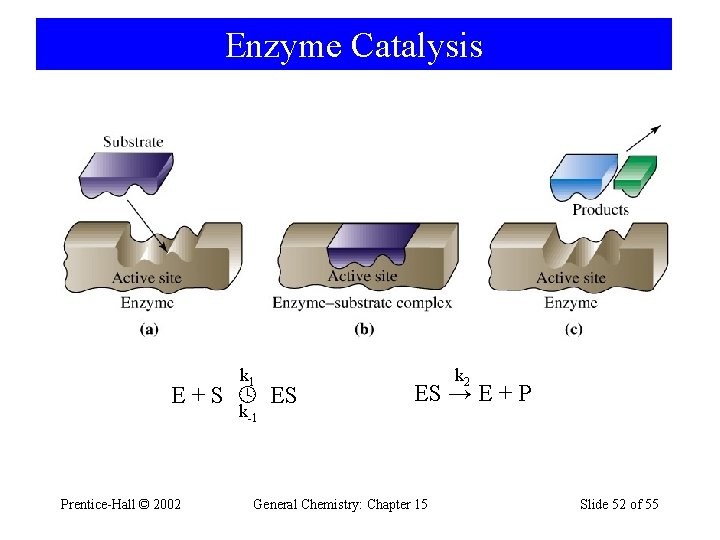
Enzyme Catalysis k 1 E + S ES k-1 Prentice-Hall © 2002 k 2 ES → E + P General Chemistry: Chapter 15 Slide 52 of 55
![Saturation Kinetics k 1 dP k 2 E S ES E Saturation Kinetics k 1 d[P] k 2 E + S ES → E +](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-53.jpg)
Saturation Kinetics k 1 d[P] k 2 E + S ES → E + P dt k-1 d[P] dt = k 2[ES] = k 1[E][S] – k-1[ES] – k 2[ES]= 0 k 1[E][S] = (k-1+k 2 )[ES] [E] = [E]0 – [ES] k 1[S]([E]0 –[ES]) = (k-1+k 2 )[ES] = Prentice-Hall © 2002 General Chemistry: Chapter 15 k 1[E]0 [S] (k-1+k 2 ) + k 1[S] Slide 53 of 55
![MichaelisMenten dP dt dP k 2E0 dt dP dt k 1 Michaelis-Menten d[P] dt d[P] = k 2[E]0 dt d[P] dt = = k 1](https://slidetodoc.com/presentation_image_h2/9b526c430bbf12d737eb1fc05ea1f355/image-54.jpg)
Michaelis-Menten d[P] dt d[P] = k 2[E]0 dt d[P] dt = = k 1 k 2[E]0 [S] (k-1+k 2 ) + k 1[S] k 2[E]0 [S] (k-1+k 2 ) + [S] k 1 d[P] dt = Prentice-Hall © 2002 k 2 [E]0 [S] KM General Chemistry: Chapter 15 d[P] dt = k 2[E]0 [S] KM + [S] Slide 54 of 55

Chapter 15 Questions Develop problem solving skills and base your strategy not on solutions to specific problems but on understanding. Choose a variety of problems from the text as examples. Practice good techniques and get coaching from people who have been here before. Prentice-Hall © 2002 General Chemistry: Chapter 15 Slide 55 of 55