UPDATES ON DIAGNOSIS AND MANAGEMENT OF GIST Dr















































- Slides: 84

UPDATES ON DIAGNOSIS AND MANAGEMENT OF GIST Dr Praveen Royal Mokkapati Moderator: Professor NK Shukla

OVERVIEW • INTRODUCTION • DIAGNOSIS • GUIDELINES ON MANAGEMENT • LOCALISED • ADVANCED • METASTATIC • IMATINIB RESISTANCE

INTRODUCTION • 1 in 1, 000 • 60 to 65 years age group • Equal gender distribution • Pediatric GIST

CLINICAL PRESENTATION • Asymptomatic • Lump • Bleeding • Rupture

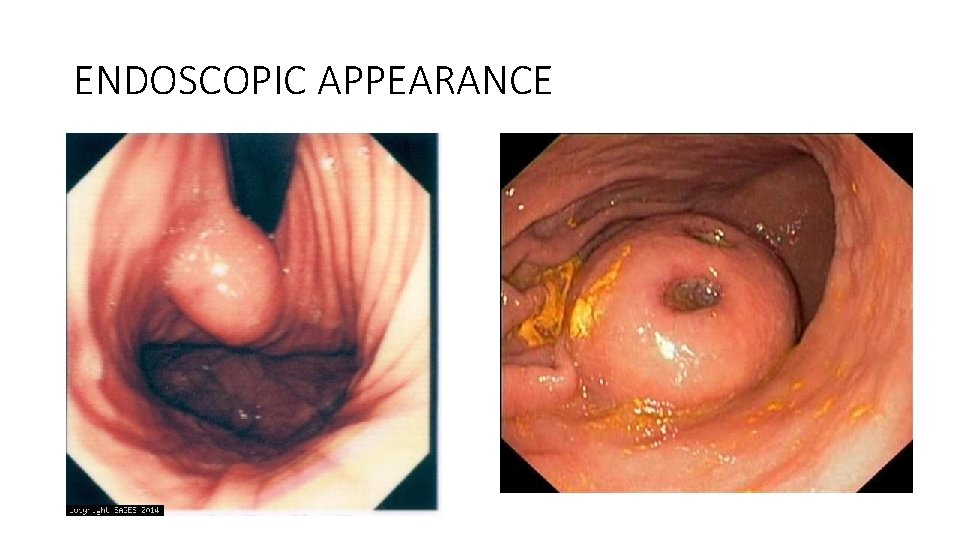
ENDOSCOPIC APPEARANCE

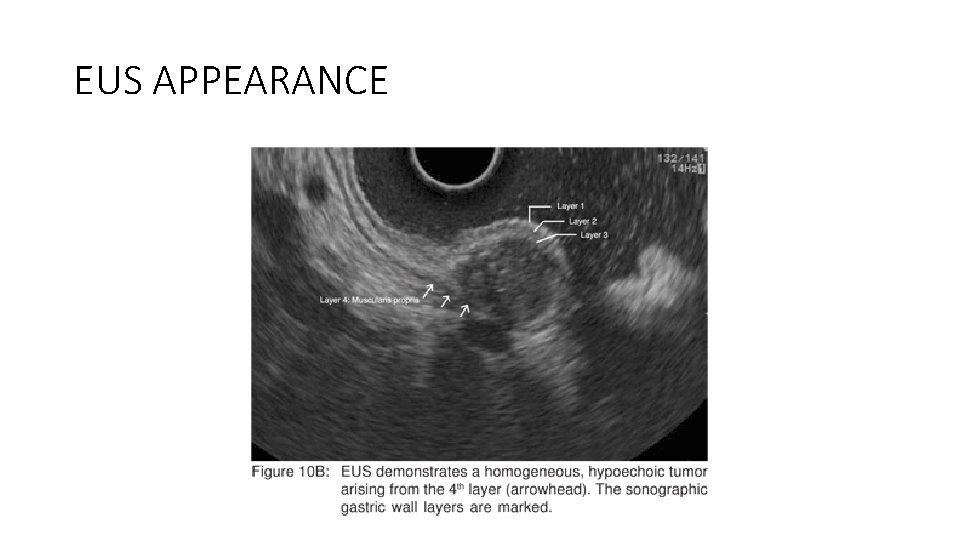
EUS APPEARANCE

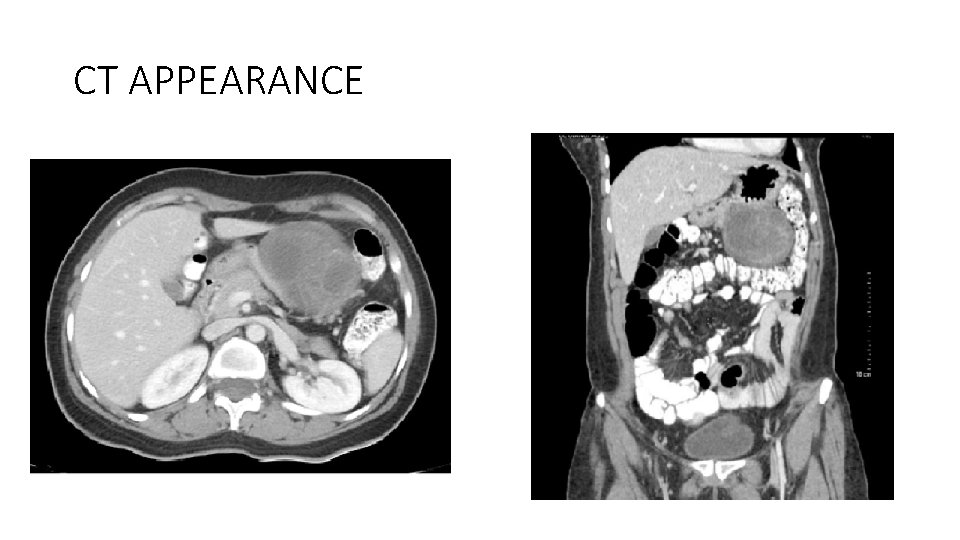
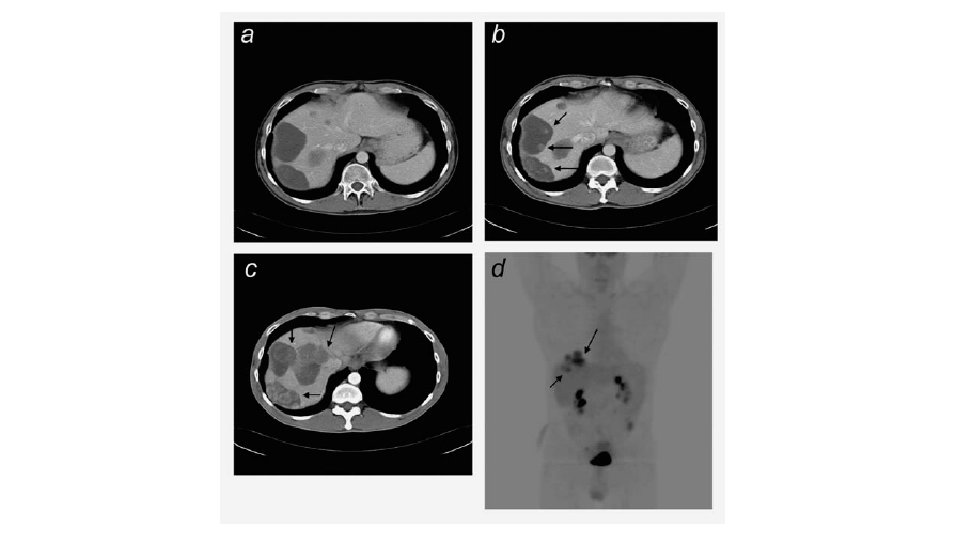
CT APPEARANCE


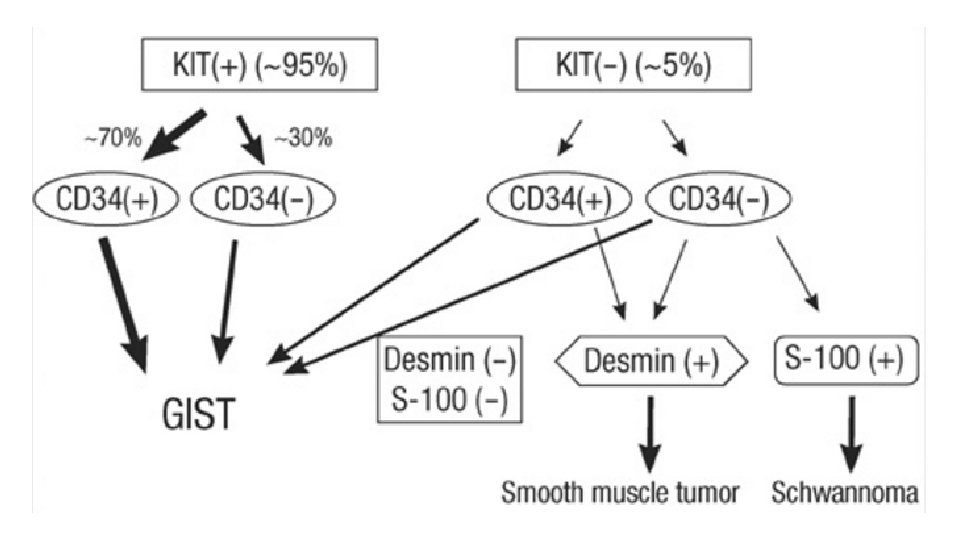
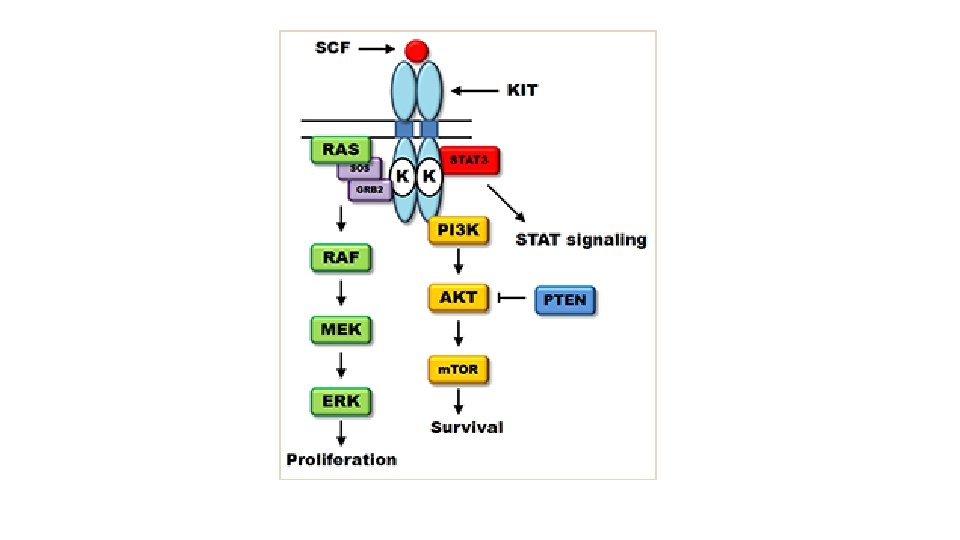
MOLECULAR BIOLOGY • c. KIT • PDGFRA • Wild type



DOG 1, DOG 1. 1, K 9

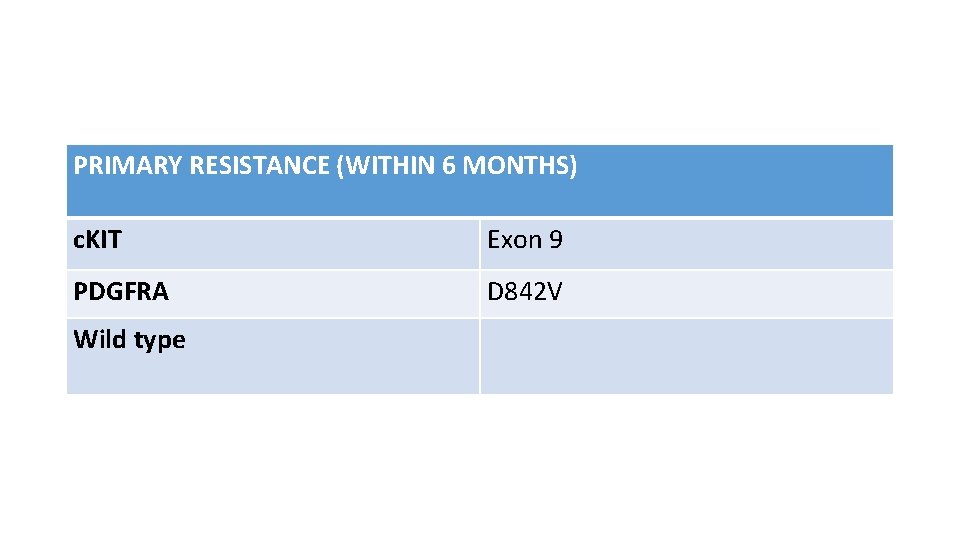
PRIMARY RESISTANCE (WITHIN 6 MONTHS) c. KIT Exon 9 PDGFRA D 842 V Wild type

SECONDARY RESISTANCE ATP binding site Exon 13, 14 Activation loop Exon 17, 18

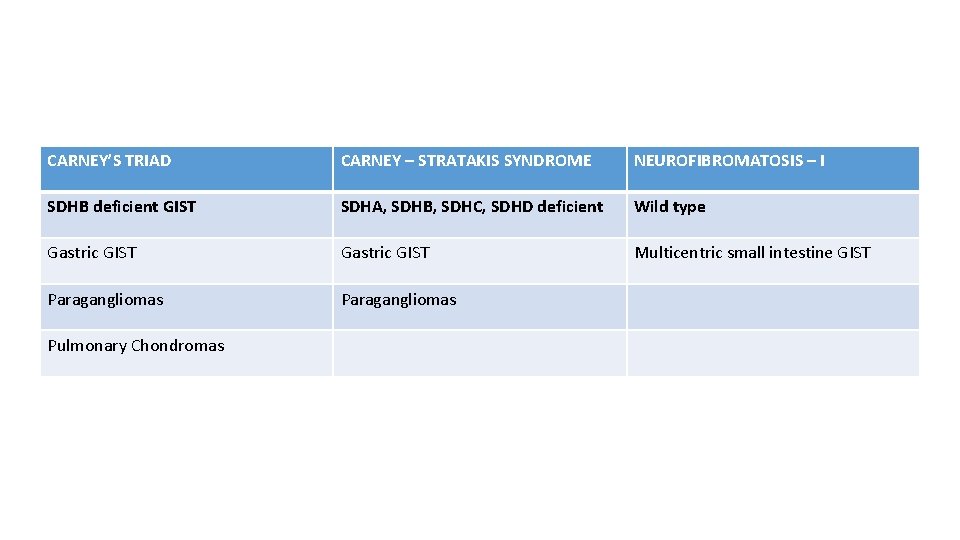
CARNEY’S TRIAD CARNEY – STRATAKIS SYNDROME NEUROFIBROMATOSIS – I SDHB deficient GIST SDHA, SDHB, SDHC, SDHD deficient Wild type Gastric GIST Multicentric small intestine GIST Paragangliomas Pulmonary Chondromas

GUIDELINES • ESMO • NCCN • CANADA, SWITZERLAND, JAPAN

• LOCALISED DISEASE • ADVANCED DISEASE • METASTATIC DISEASE

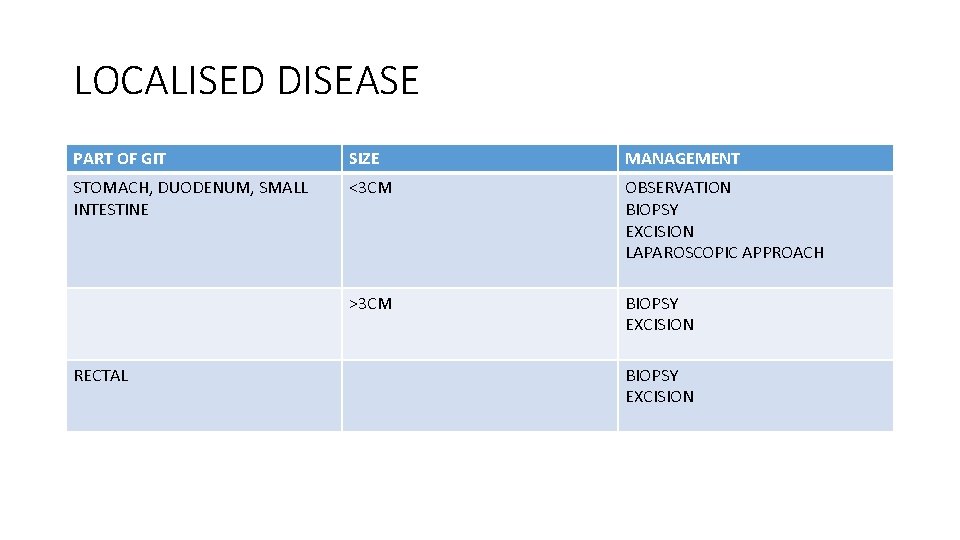
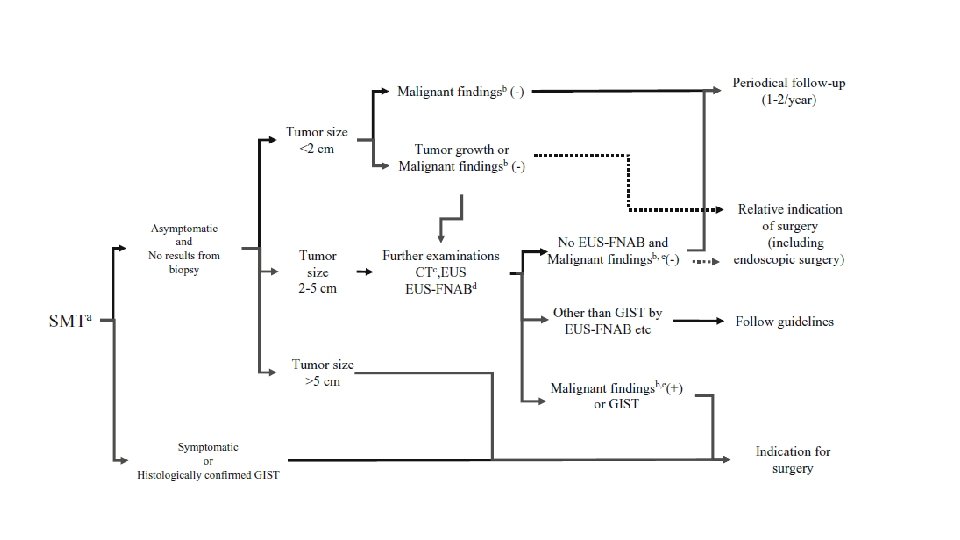
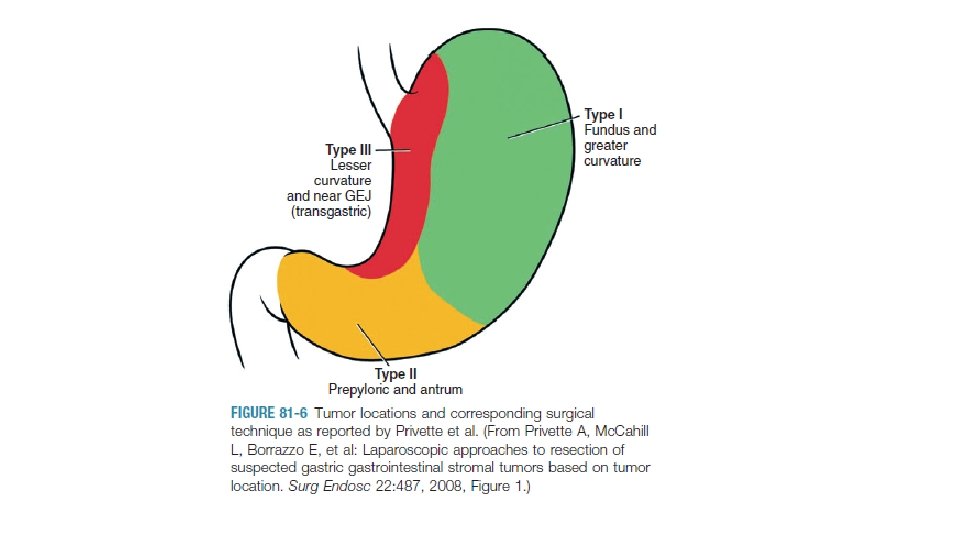
LOCALISED DISEASE PART OF GIT SIZE MANAGEMENT STOMACH, DUODENUM, SMALL INTESTINE <3 CM OBSERVATION BIOPSY EXCISION LAPAROSCOPIC APPROACH >3 CM BIOPSY EXCISION RECTAL BIOPSY EXCISION


LOCALISED DISEASE PART OF GIT SIZE MANAGEMENT STOMACH, DUODENUM, SMALL INTESTINE <3 CM OBSERVATION BIOPSY EXCISION LAPAROSCOPIC APPROACH >3 CM BIOPSY EXCISION RECTAL BIOPSY EXCISION


• R 1 RESECTION • EXACT R 1 SITE KNOWN? • INCREASED MORBIDITY? • ADJUVANT TREATMENT • INDICATIONS? • DURATION? • PFS vs OS

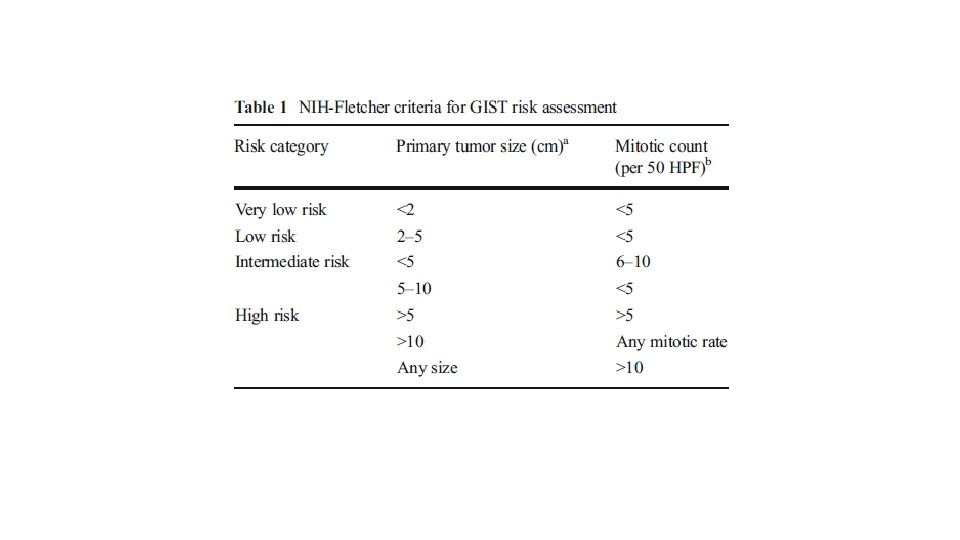
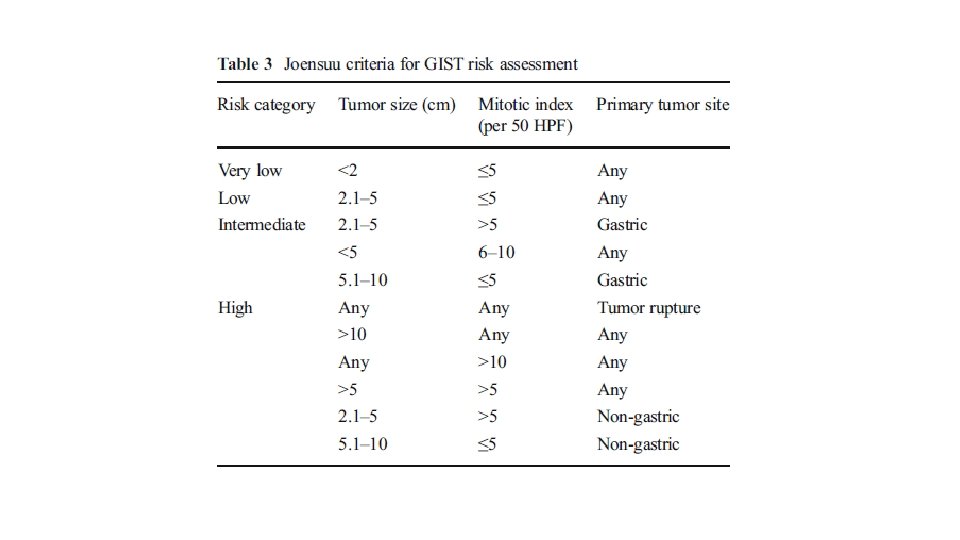
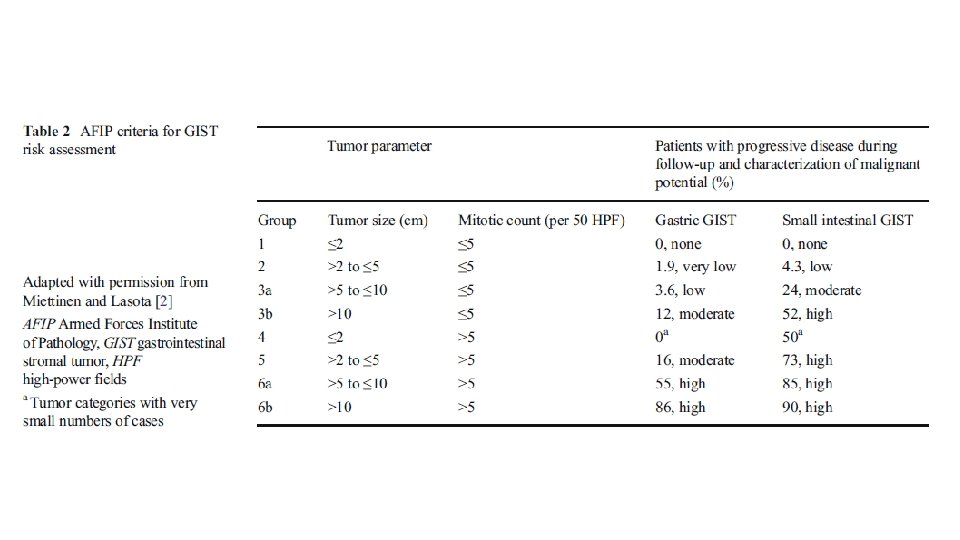
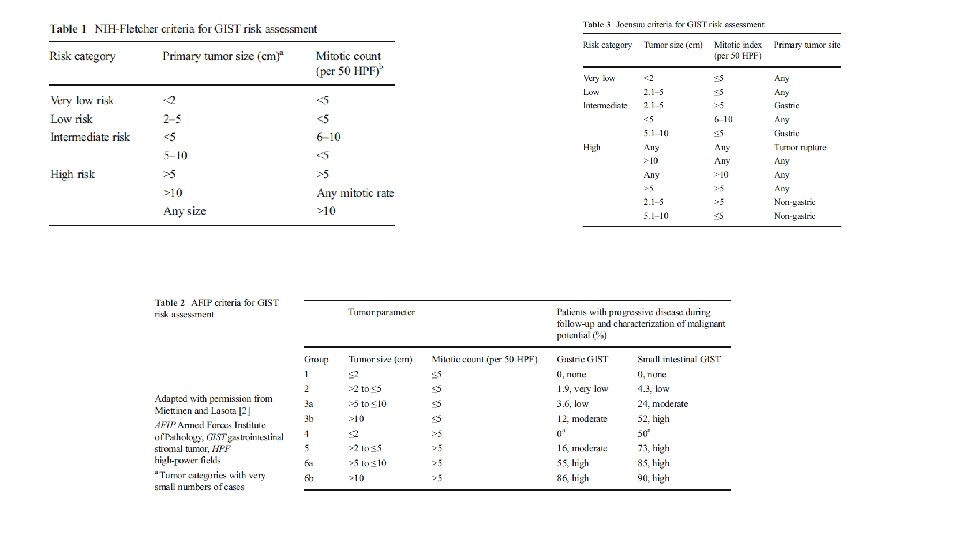
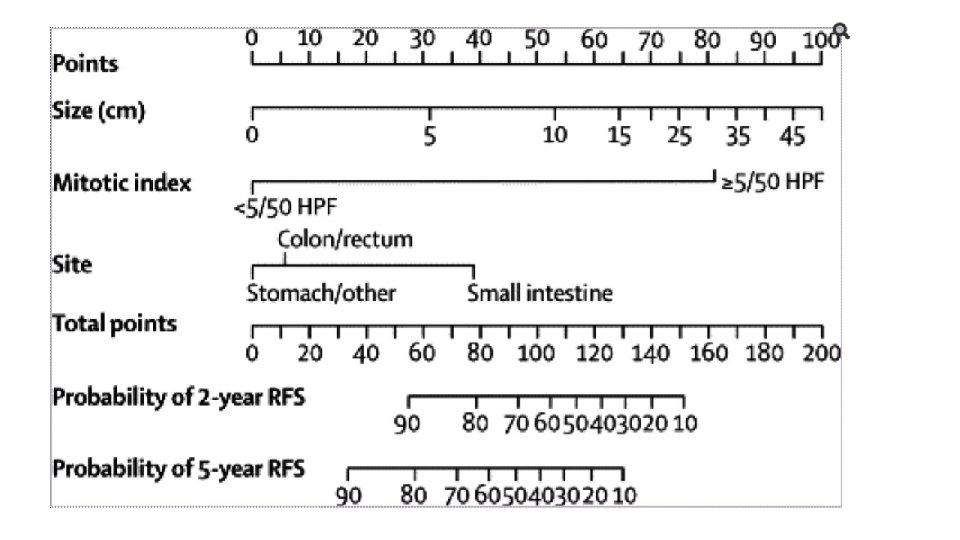
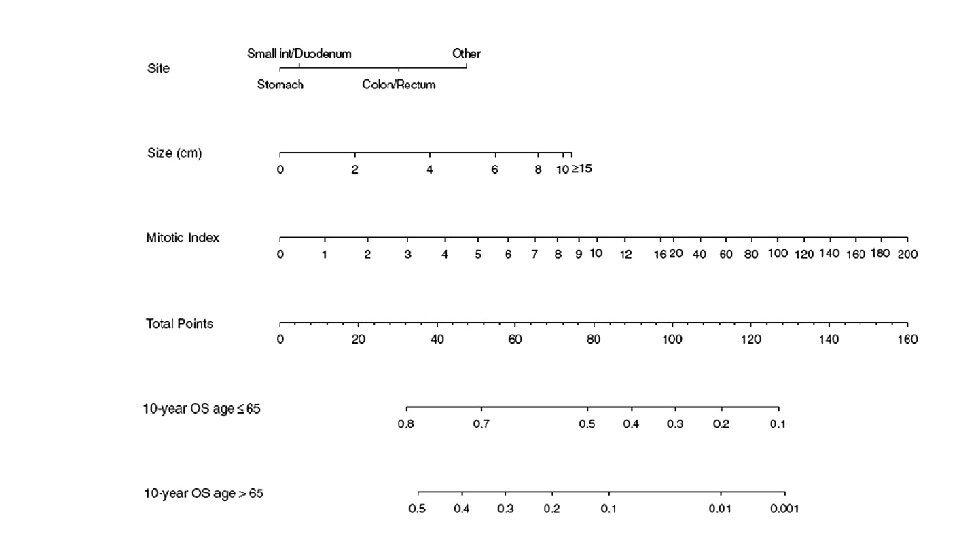
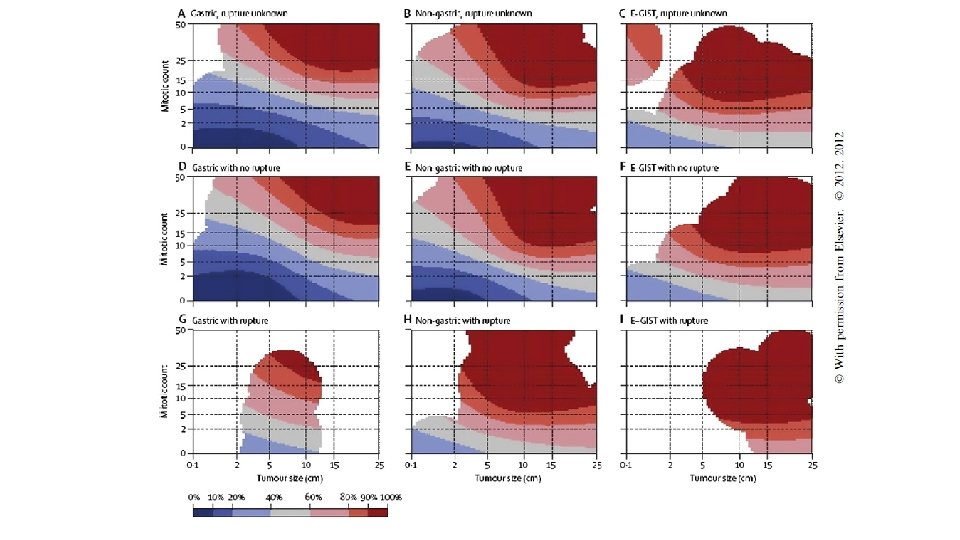
RISK STRATIFICATION • NIH Consensus criteria • Modified NIH Consensus criteria or modified consensus criteria • AFIP criteria • Nomograms MSKCC, Italian • Non linear model









PRE IMATINIB • 5 YEAR SURVIVAL RATE 35% OVERALL • Among 127 patients with completely resected primary GIST • PFS ON CHEMO RECURRENCE FREE SURVIVAL AT PERCENTAGE 1 YEAR 83% 2 YEARS 75% 5 YEARS 63%

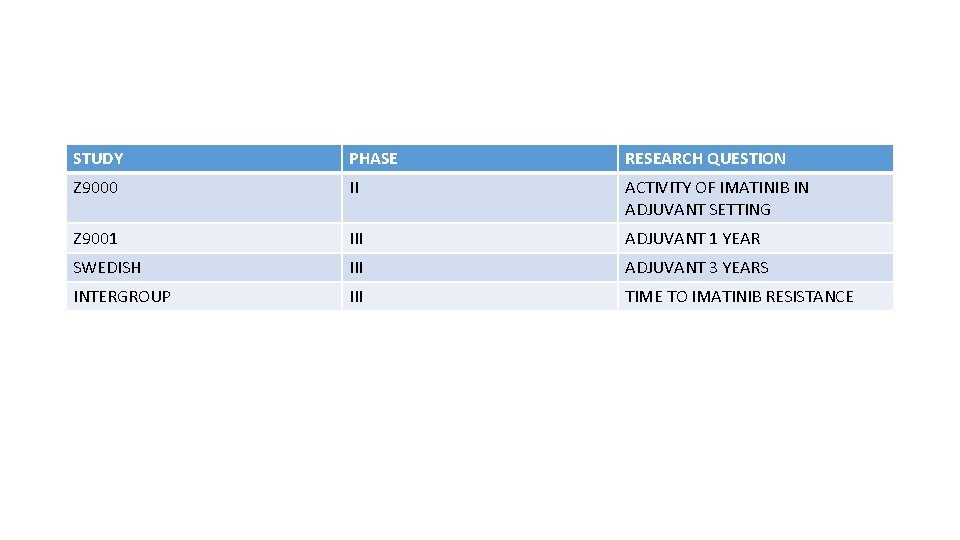
STUDY PHASE RESEARCH QUESTION Z 9000 II ACTIVITY OF IMATINIB IN ADJUVANT SETTING Z 9001 III ADJUVANT 1 YEAR SWEDISH III ADJUVANT 3 YEARS INTERGROUP III TIME TO IMATINIB RESISTANCE

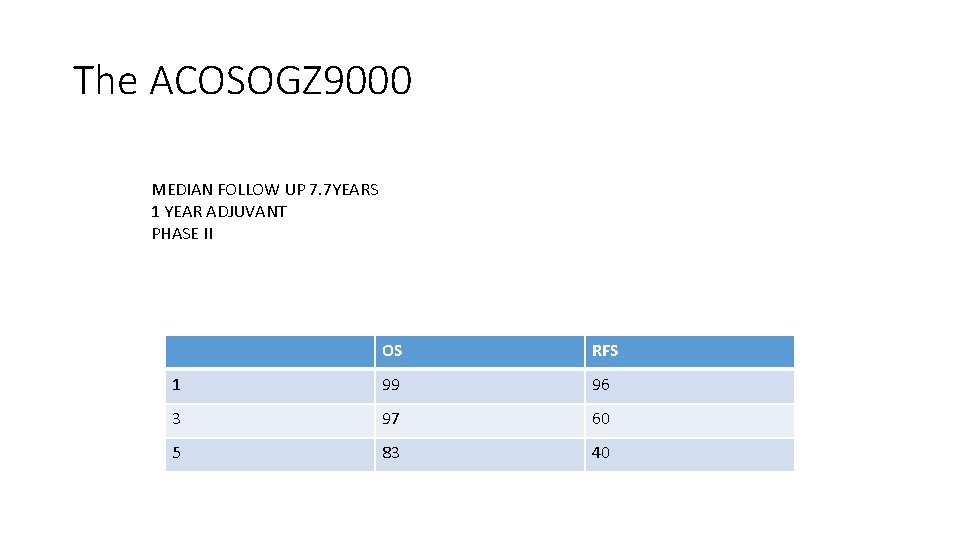
The ACOSOGZ 9000 MEDIAN FOLLOW UP 7. 7 YEARS 1 YEAR ADJUVANT PHASE II OS RFS 1 99 96 3 97 60 5 83 40

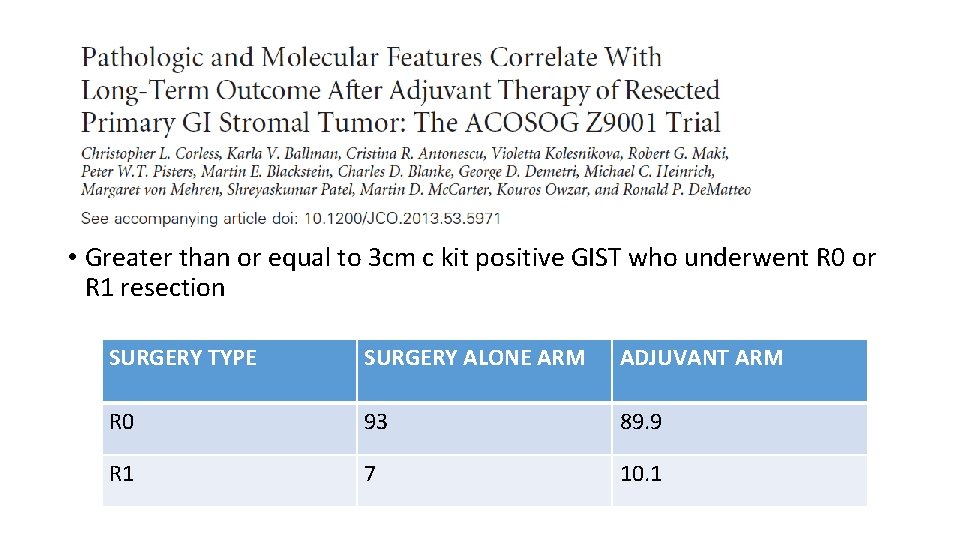
• Greater than or equal to 3 cm c kit positive GIST who underwent R 0 or R 1 resection SURGERY TYPE SURGERY ALONE ARM ADJUVANT ARM R 0 93 89. 9 R 1 7 10. 1


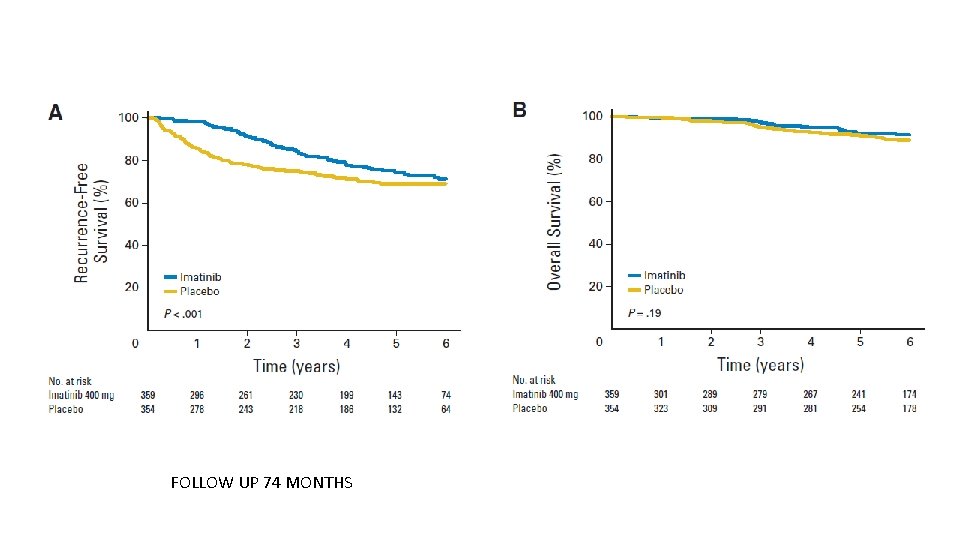
FOLLOW UP 74 MONTHS

• • Greater than 10 cm size Greater than 10 mitosis/50 hpf Greater than 5 cm and greater than 5 mitosis/50 hpf Tumor rupture


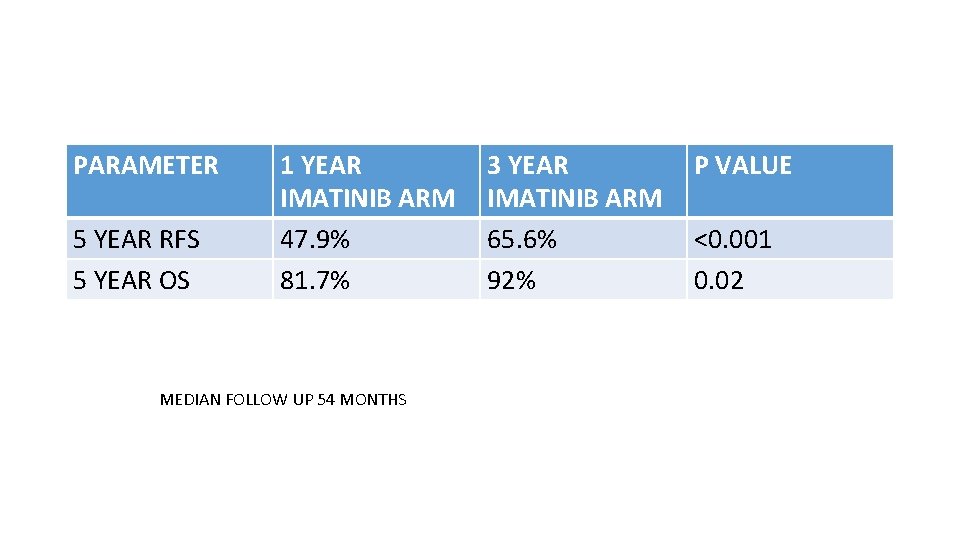
PARAMETER 5 YEAR RFS 5 YEAR OS 1 YEAR IMATINIB ARM 47. 9% 81. 7% MEDIAN FOLLOW UP 54 MONTHS 3 YEAR IMATINIB ARM 65. 6% 92% P VALUE <0. 001 0. 02


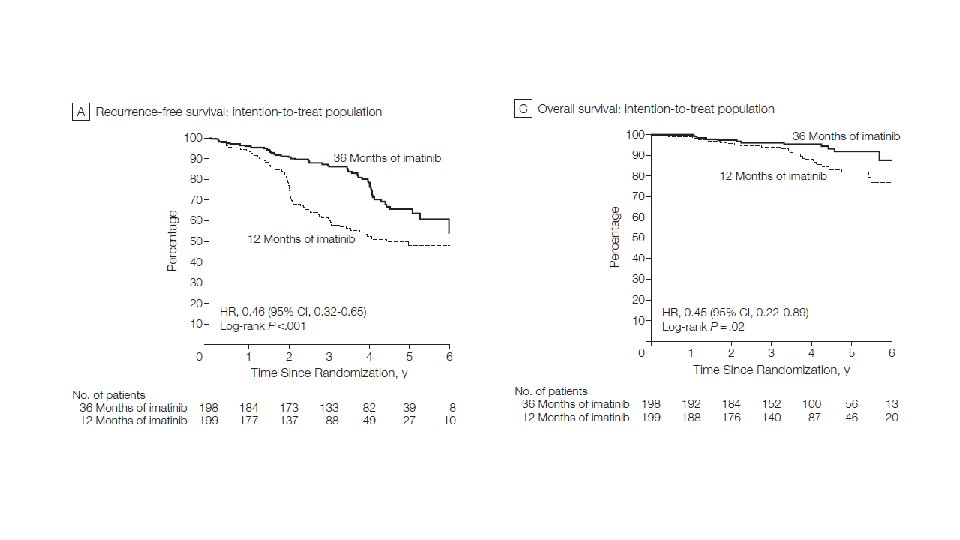
• INTERMEDIATE AND HIGH RISK GROUPS, > OR EQUAL TO 3 CM • 3 YEARS DURATION • DOESN’T BRING ABOUT FASTER RESISTANCE

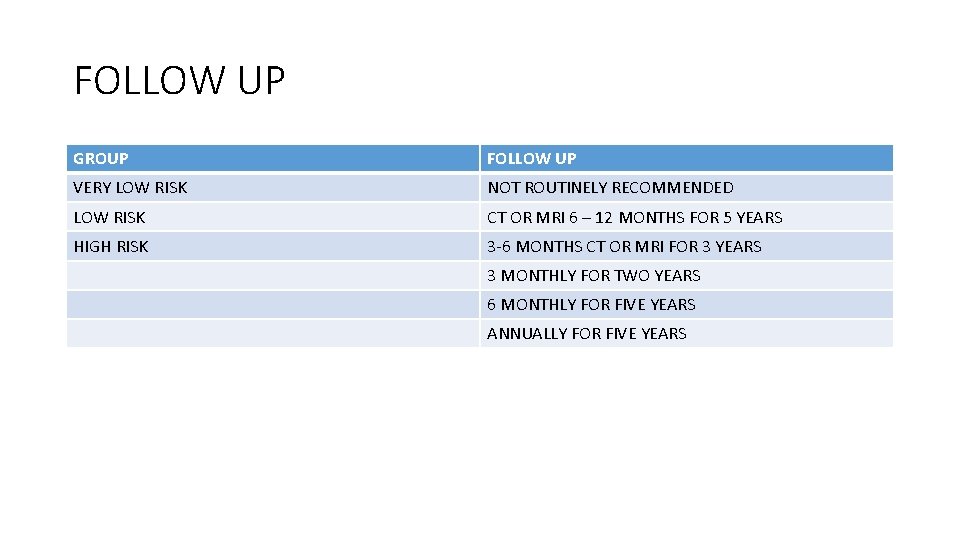
FOLLOW UP GROUP FOLLOW UP VERY LOW RISK NOT ROUTINELY RECOMMENDED LOW RISK CT OR MRI 6 – 12 MONTHS FOR 5 YEARS HIGH RISK 3 -6 MONTHS CT OR MRI FOR 3 YEARS 3 MONTHLY FOR TWO YEARS 6 MONTHLY FOR FIVE YEARS ANNUALLY FOR FIVE YEARS


ADVANCED DISEASE NEOADJUVANT THERAPY • INDICATION • INOPERABLE • MULTIVISCERAL RESECTION • FUNCTION SPARING RESECTION • R 1 • DURATION

ROLE OF MOLECULAR SUBTYPING • EXON 9 • D 842 V • WILD TYPE • IF NOT DONE ROUTINELY?

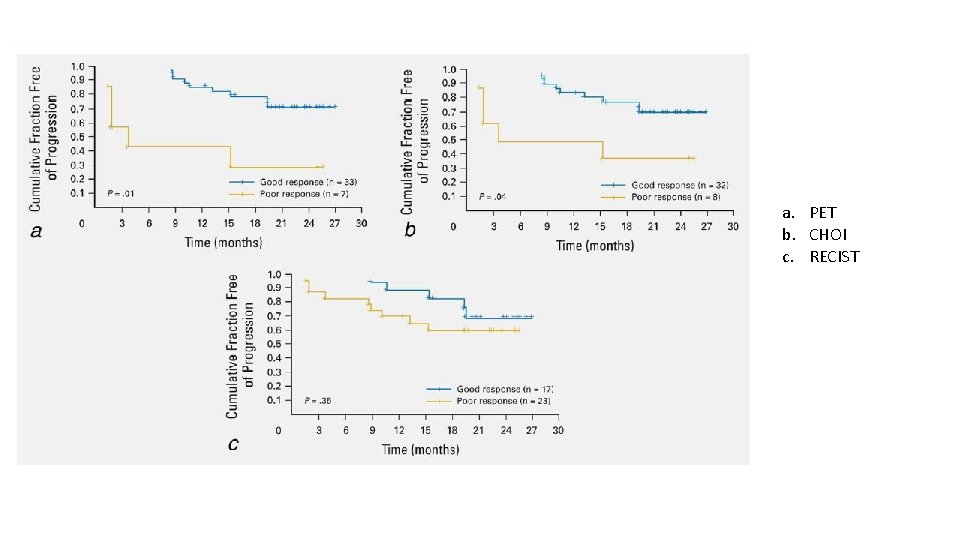
RESPONSE ASSESSMENT RECIST SWOG PET CHOI

B 2222 trial EORTC 62005 NORTH AMERICAN INTERGROUP S 0033

PSEUDO PROGRESSION • Increase in size • Liver metastasis May happen even in responsive tumors



a. PET b. CHOI c. RECIST

CHOI CRITERIA Minimum 10% decrease in tumor size Minimum 15% decrease in tumour density

EVIDENCE FOR NEOADJUVANT THERAPY • Based on only case reports and case series • No phase III studies • Only one prospective series

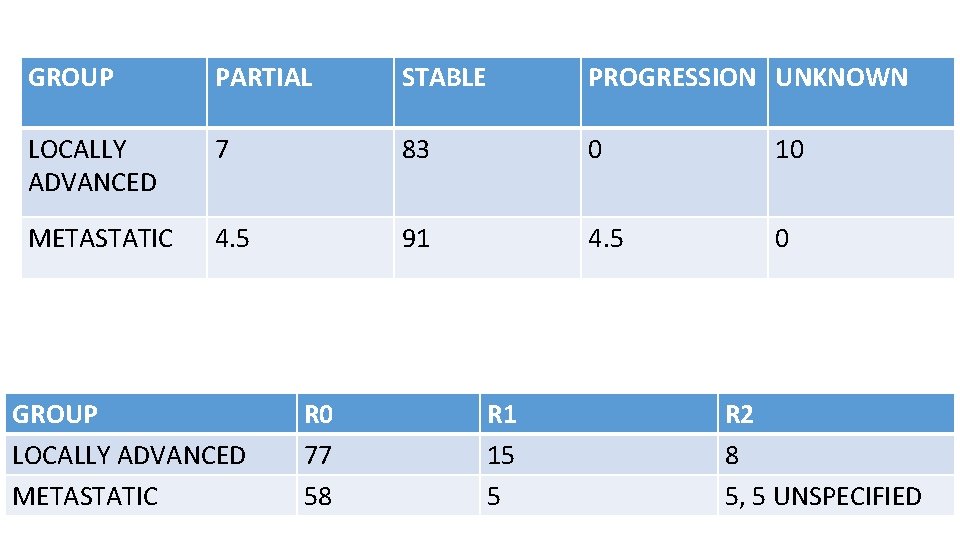
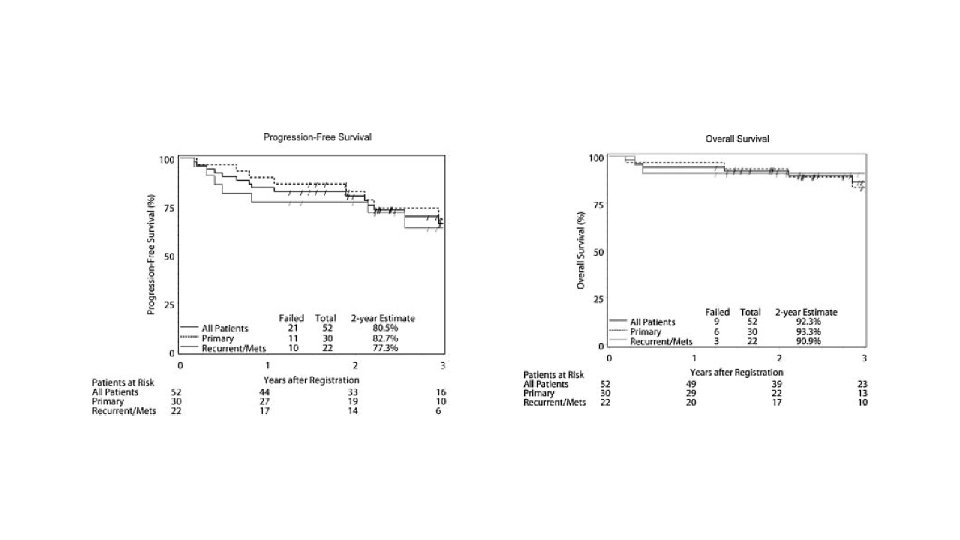
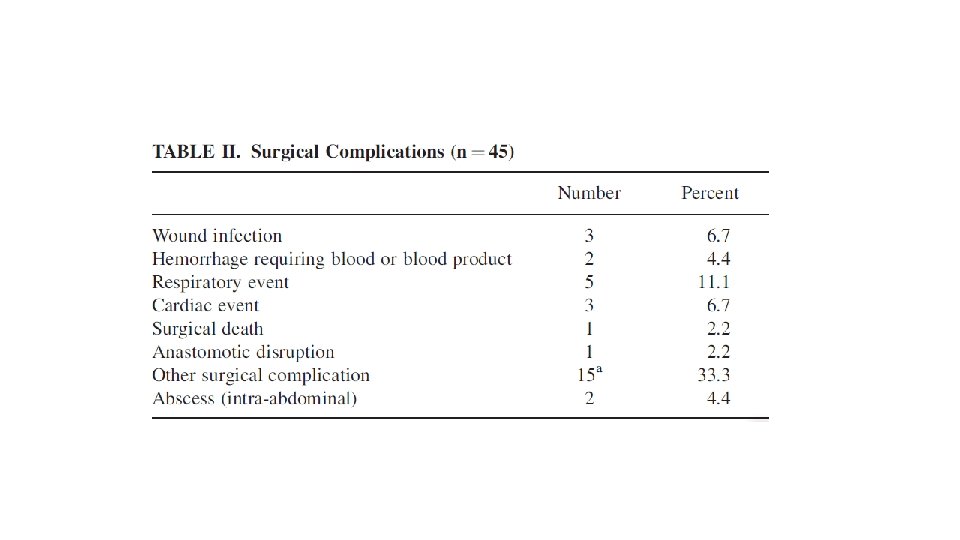
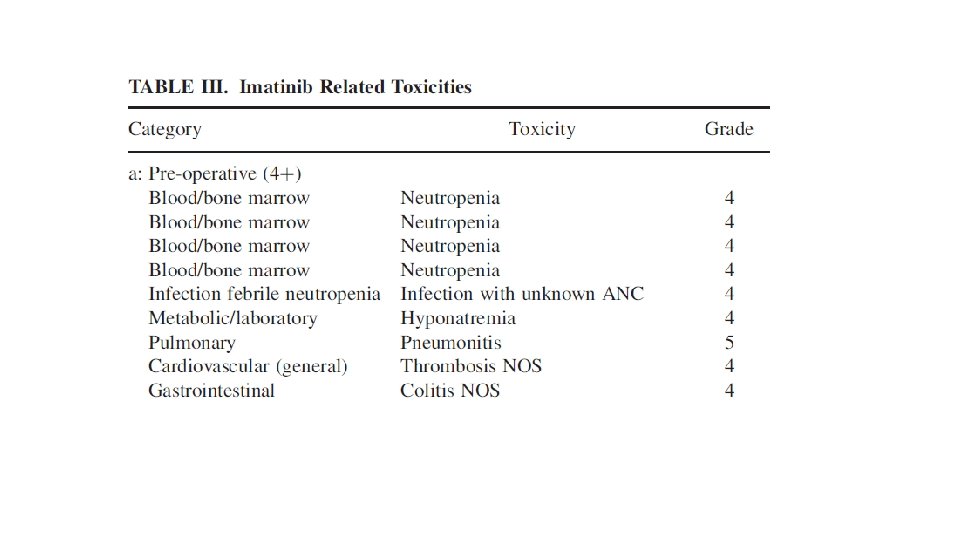
52 patients – 30 locally advanced(>5 CM) 22 metastatic Median follow up 3 years 8 – 12 weeks 600 mg imatinib neoadjuvant with 2 years adjuvant imatinib or till progression in metastatic cases Response assessment by RECIST using PET CT and CT

GROUP PARTIAL STABLE PROGRESSION UNKNOWN LOCALLY ADVANCED 7 83 0 10 METASTATIC 4. 5 91 4. 5 0 GROUP LOCALLY ADVANCED METASTATIC R 0 77 58 R 1 15 5 R 2 8 5, 5 UNSPECIFIED






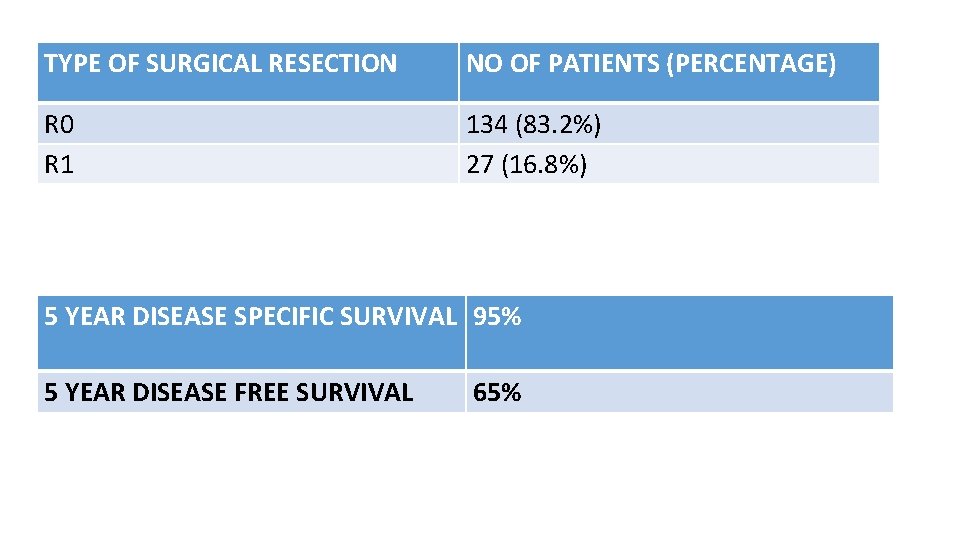
TYPE OF SURGICAL RESECTION NO OF PATIENTS (PERCENTAGE) R 0 R 1 134 (83. 2%) 27 (16. 8%) 5 YEAR DISEASE SPECIFIC SURVIVAL 95% 5 YEAR DISEASE FREE SURVIVAL 65%

RECTAL GIST

POSTOPERATIVE IMATINIB AFTER NEOADJUVANT

METASTATIC DISEASE • SURGERY? • TKI • DURATION • RESISTANCE

PHASE III • NCT 00956072 in Europe • Chi. CTR-TRC-00000244 in China

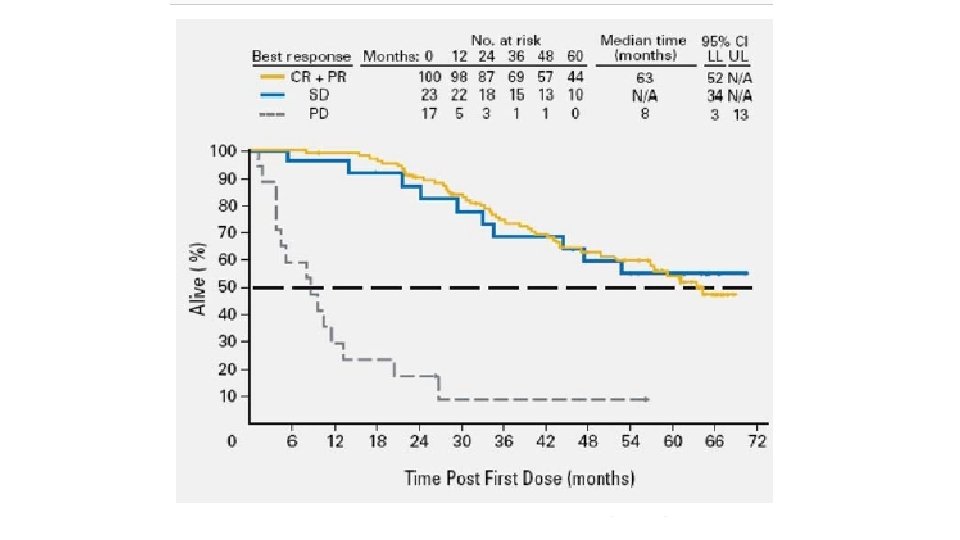
SURVIVAL 171 patients Median follow up 56. 6 months IMATINIB + SURGERY IMATINIB P VALUE 5 YR PFS 58% 40% 0. 4 5 YR OS 75. 4% 50% 0. 022 MEDIAN OS 87. 6 mo 59. 9 mo


PROGRESSION • Compliance • Pharmacokinetics • Secondary resistance • Limited progression vs Generalized progression

NEXT LINES • Imatinib 800 mg • Sunitinib • Regorafenib

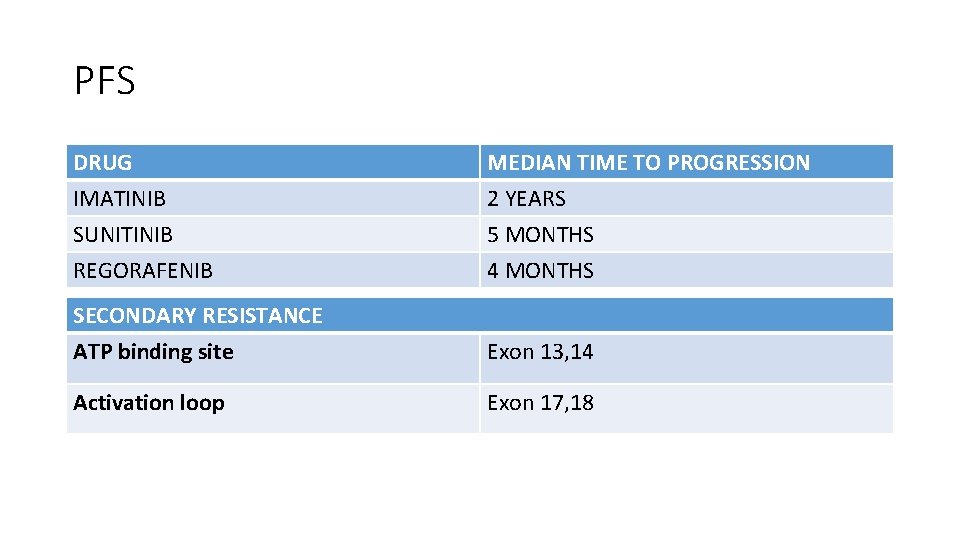
PFS DRUG IMATINIB SUNITINIB REGORAFENIB MEDIAN TIME TO PROGRESSION 2 YEARS 5 MONTHS 4 MONTHS SECONDARY RESISTANCE ATP binding site Exon 13, 14 Activation loop Exon 17, 18

• DRUGS ACTING AT RESISTANCE SITES • DRUGS ACTING DOWNSTREAM • DRUG COMBINATIONS

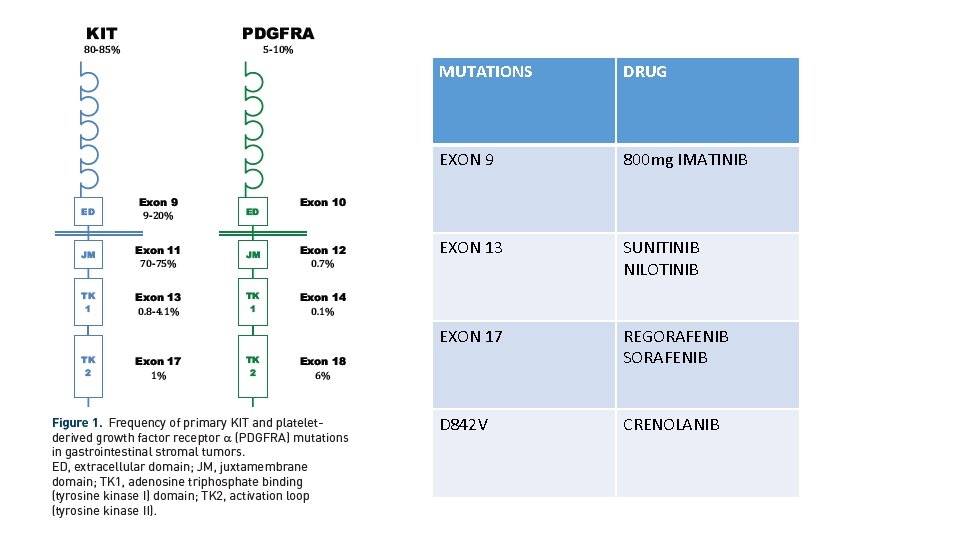
MUTATIONS DRUG EXON 9 800 mg IMATINIB EXON 13 SUNITINIB NILOTINIB EXON 17 REGORAFENIB SORAFENIB D 842 V CRENOLANIB



• DOVITINIB • PAZOPANIB • PONATINIB





1. 15 vs 7. 07 (p=0. 03) Colon cancer Renal cell cancers Melanoma

NOVARTIS VS UNION OF INDIA 1 st April 2013

THANK YOU