Thermochemistry Dr Ron Rusay Energy Joules J calorie



























![Hrxn° = [1 Hf (c) + 2 Hf (d)] [1 Hf (a) + Hrxn° = [1 Hf (c) + 2 Hf (d)] [1 Hf (a) +](https://slidetodoc.com/presentation_image_h/1ebbdb5c0be7468e66f64e88e7e2cb3f/image-38.jpg)

















- Slides: 64

Thermochemistry Dr. Ron Rusay

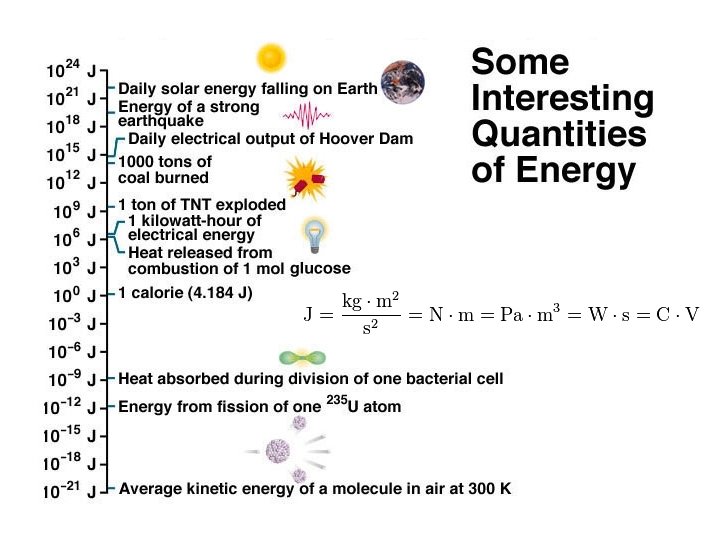
Energy Joules (J) / calorie (cal) : (4. 184 J = 1 cal) ð Can be defined as the capacity to do work. ð Chemical energy is defined as heat. ð Name five other types of Energy.

Two Types of Energy ð Potential: due to an object’s position or material’s composition - which can be converted to work ð Kinetic: due to motion of an object ð KE = 1/2 mv 2 ð (m = mass, v = velocity)


Law of Conservation of Energy ð Different forms of energy can be inter -converted but can neither be created nor destroyed. ð (Euniverse ð Describe is constant) three inter-conversions of energy.

Temperature v. Energy ð Temperature reflects random motions of particles; i. e. the kinetic energy of a system. ð Heat involves a transfer of energy between 2 objects due to different energies and temperature differences. Always: HOT cold

Heat (Energy) Loss

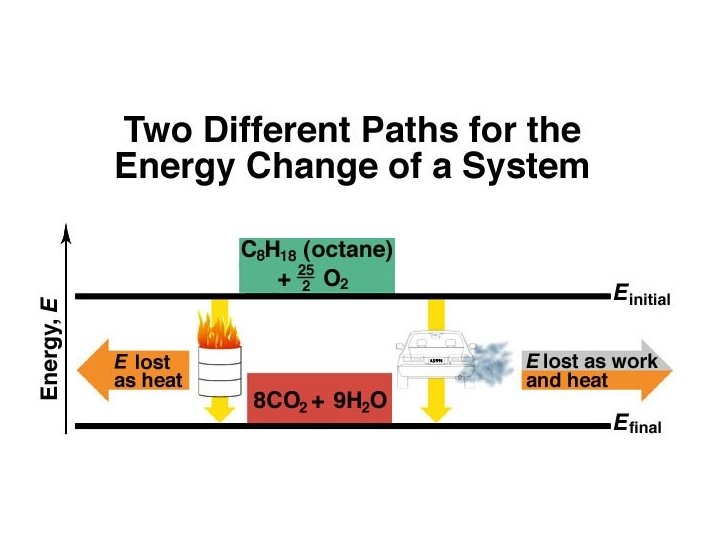
Energy: A State Function ð ð Depends only on the state of the system - not the path of how it arrived at that state. It is independent of pathway.

System and Surroundings ð System: That on which we focus attention ð Surroundings: Everything else in the universe ð Universe = System + Surroundings


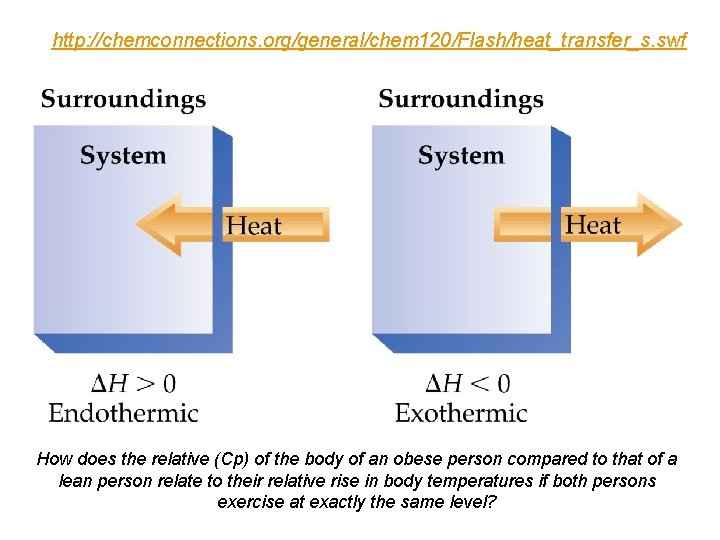
Defining Energy Change Exo- and Endo- thermic (Exergonic and Endergonic) ð Two types of energy change : ð Exothermic: Heat flows out of the system (to the surroundings). …negative sign ð Endothermic: Heat flows into the system (from the surroundings). …positive sign

First of Three Laws of Thermodynamics First Law of Thermodynamics: The energy of the universe is constant or “energy is conserved”. ð

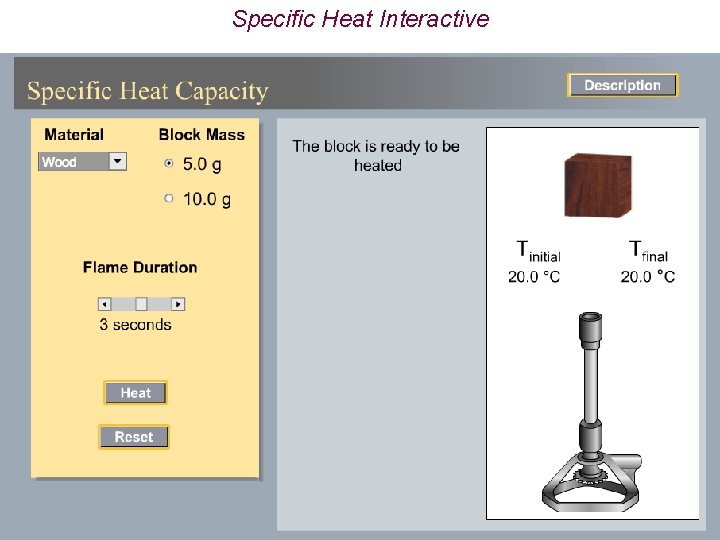
Heat Capacity (Specific Heat) P O http: //chemconnections. org/general/chem 120/Flash/specific_heat_s. swf

Terminology ð Specific heat capacity per gram = J/°C g or J/K g ð Molar heat capacity per mole = J/°C mol or J/K mol

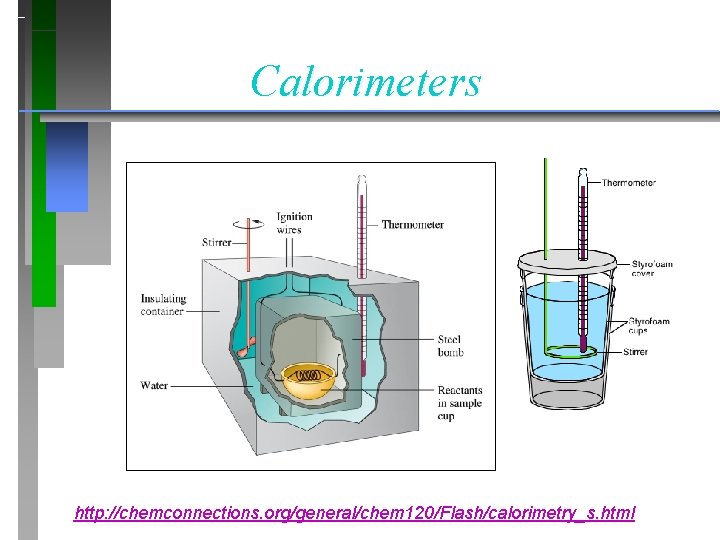
Calorimeters http: //chemconnections. org/general/chem 120/Flash/calorimetry_s. html

QUESTION

Heat Capacities

QUESTION

Why can you burn the top of your mouth with hot pizza and not the bottom? (The top & bottom are at the same temperature!) http: //www. dailymotion. com/video/x 3 hfwx_the-science-of-pizza_people

Why can you burn the top of your mouth with hot pizza and not the bottom? (The top & bottom are at the same temperature. ) (Cp) on body fat. In obese mice (fat content 52. 76% body wt) the heat capacity was 2. 65 k. J kg-1 K-1 and in lean mice (fat content 7. 55% body wt) the heat capacity was 3. 66 k. J kg-1 K-1.

Specific Heat Interactive

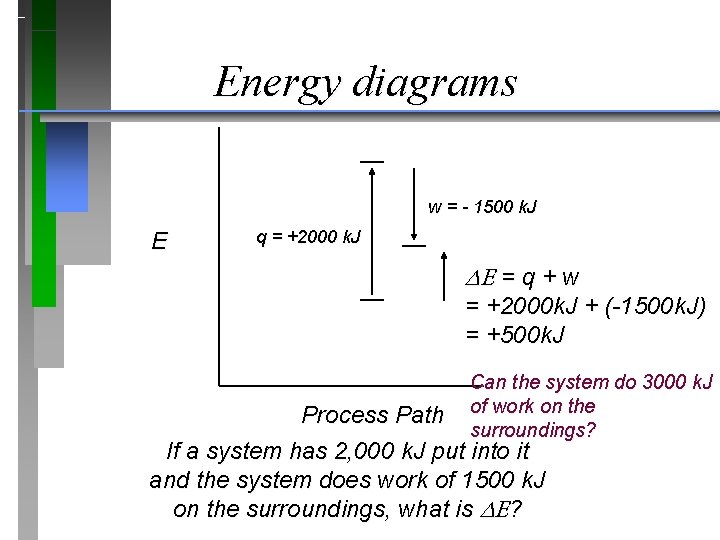
Energy diagrams w = - 1500 k. J E q = +2000 k. J = q + w = +2000 k. J + (-1500 k. J) = +500 k. J Can the system do 3000 k. J of work on the surroundings? Process Path If a system has 2, 000 k. J put into it and the system does work of 1500 k. J on the surroundings, what is ?

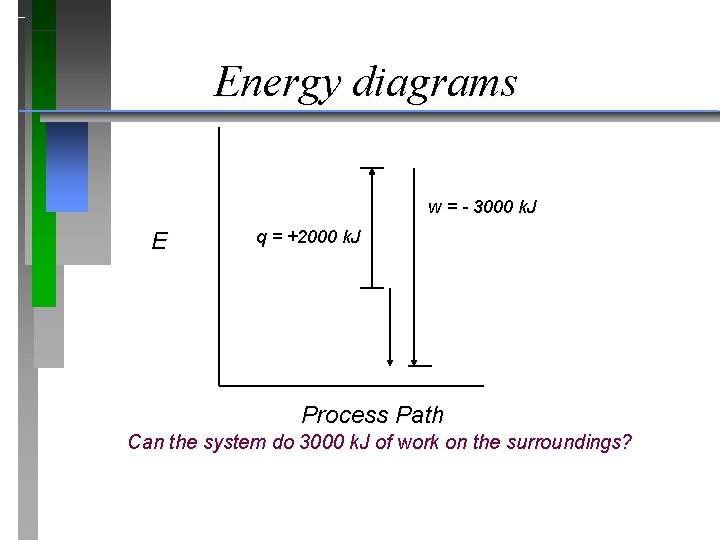
Energy diagrams w = - 3000 k. J E q = +2000 k. J Process Path Can the system do 3000 k. J of work on the surroundings?

QUESTION


QUESTION


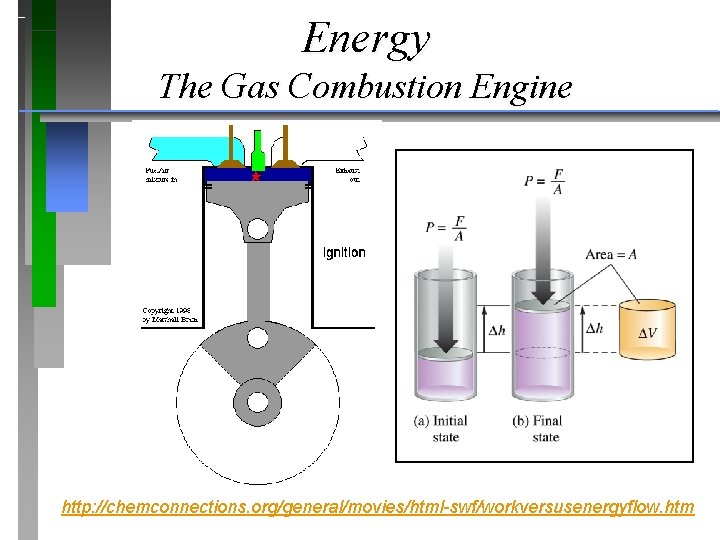
Energy The Gas Combustion Engine http: //chemconnections. org/general/movies/html-swf/workversusenergyflow. htm


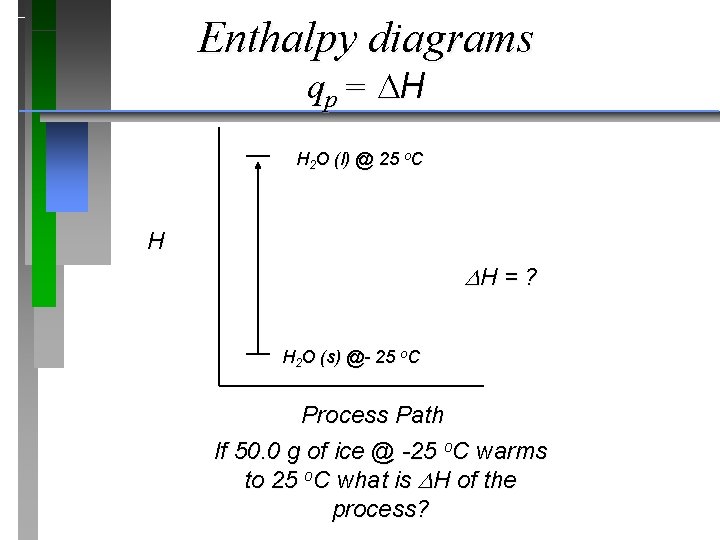
Enthalpy diagrams qp = H H 2 O (l) @ 25 o. C H H = ? H 2 O (s) @- 25 o. C Process Path If 50. 0 g of ice @ -25 o. C warms to 25 o. C what is H of the process?

http: //chemconnections. org/general/movies/Heating. Curves. swf

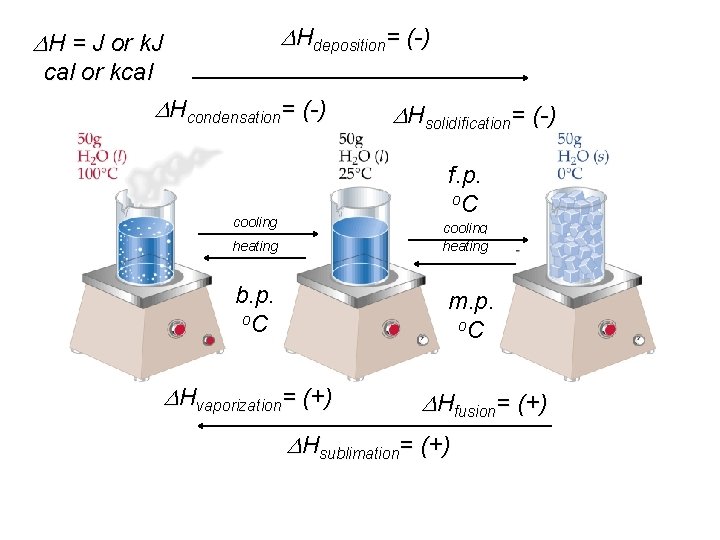
Hdeposition= (-) H = J or k. J cal or kcal Hcondensation= (-) Hsolidification= (-) f. p. o. C cooling heating b. p. o. C m. p. o. C Hvaporization= (+) Hfusion= (+) Hsublimation= (+)

Enthalpies H? s l g H 2 O (l) @ 25 o. C H = Cp liq x mass x T H H 2 O (l) @ 0 o. C H 2 O (s) @- 25 o. C Hfusion = 6. 009 k. J/mol H = Cp ice x mass x T Process Path H = Hice + Hfusion + Hliq If 50. 0 g of ice @ -25 o. C warms H = ? to 25 o. C what is H of the process?

QUESTION

“Heat of Reaction” http: //chemconnections. org/general/movies/hesslaw. mov Change in Enthalpy ð The heat of any reaction can be calculated from enthalpies of formation of reactants and products. (“Hess’s Law”) ð Hrxn° = np Hf (products) nr Hf (reactants)

QUESTION A) 4675 k. J B) -1545 k. J D) -1720 k. J C) -290 k. J

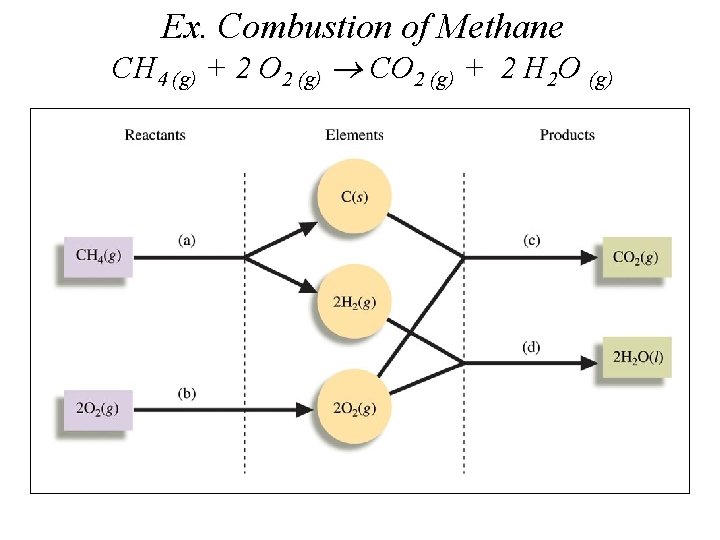
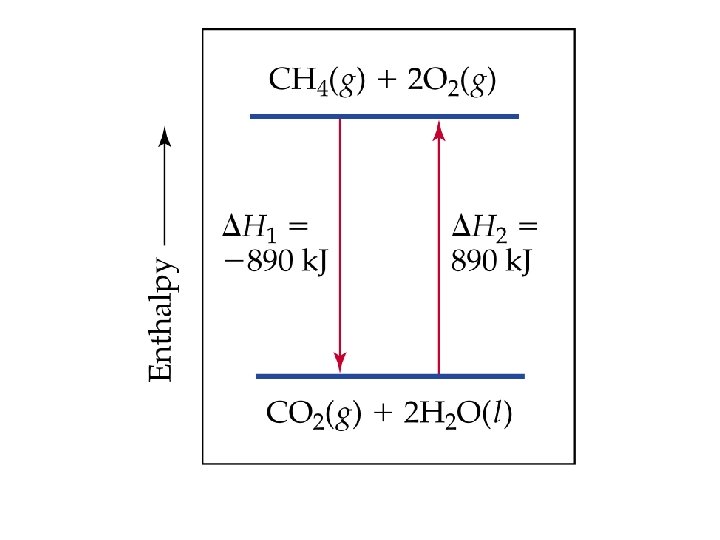
Ex. Combustion of Methane CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g)
![Hrxn 1 Hf c 2 Hf d 1 Hf a Hrxn° = [1 Hf (c) + 2 Hf (d)] [1 Hf (a) +](https://slidetodoc.com/presentation_image_h/1ebbdb5c0be7468e66f64e88e7e2cb3f/image-38.jpg)
Hrxn° = [1 Hf (c) + 2 Hf (d)] [1 Hf (a) + 2 Hf (b)] - Hrxn°= [-394 k. J+(-572 k. J)]-[-75 k. J+0 k. J]= -891 k. J

QUESTION

Exo- and Endo- thermic (Exergonic and Endergonic) ð Heat exchange accompanies chemical reactions. ð Exothermic: Heat flows out of the system (to the surroundings). …negative sign ð Endothermic: Heat flows into the system (from the surroundings). …positive sign

QUESTION

Remember from earlier slides: http: //chemconnections. org/general/chem 120/Flash/specific_heat_s. swf (Cp) of a body: In obese mice (fat content 52. 76% body wt) the heat capacity was 2. 65 k. J kg -1 K-1 and in lean mice (fat content 7. 55% body wt) the heat capacity was 3. 66 k. J kg-1 K-1.

http: //chemconnections. org/general/chem 120/Flash/heat_transfer_s. swf How does the relative (Cp) of the body of an obese person compared to that of a lean person relate to their relative rise in body temperatures if both persons exercise at exactly the same level?

“Heat of Reaction” Change in Enthalpy ð The heat of any reaction can be calculated from enthalpies of formation of reactants and products. (“Hess’s Law”) Hrxn° = np Hf (products) nr Hf (reactants) http: //chemconnections. org/general/chem 120/Flash/hess_law_s. swf

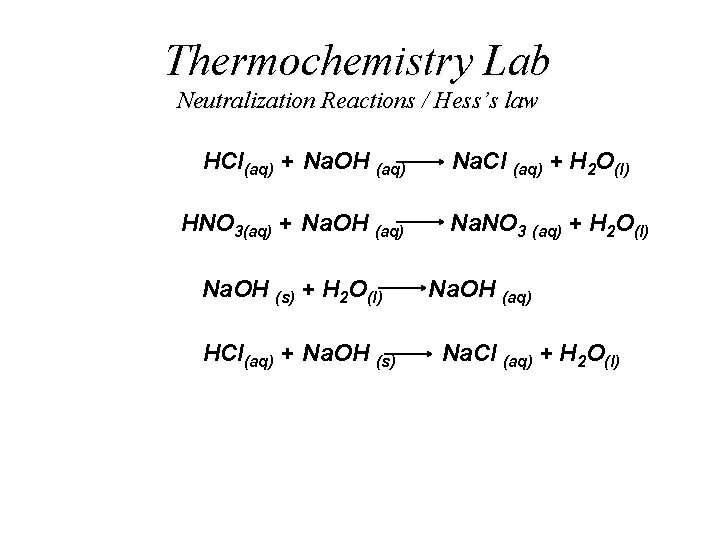
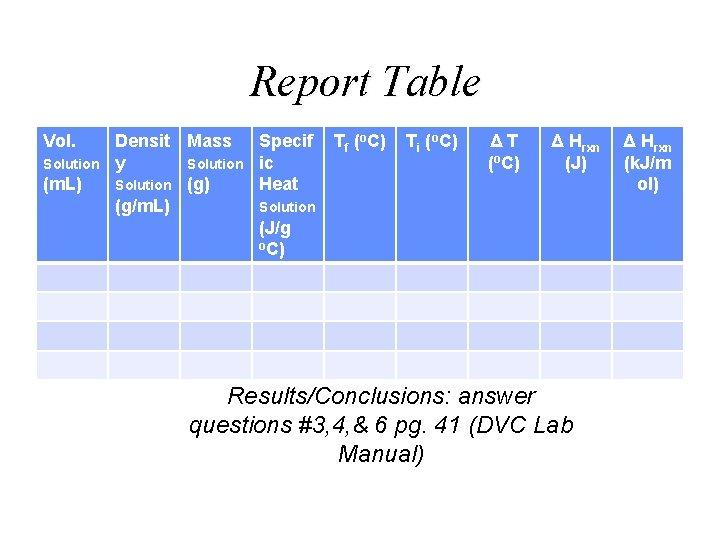
Thermochemistry Lab Neutralization Reactions / Hess’s law HCl(aq) + Na. OH (aq) HNO 3(aq) + Na. OH (aq) Na. OH (s) + H 2 O(l) HCl(aq) + Na. OH (s) Na. Cl (aq) + H 2 O(l) Na. NO 3 (aq) + H 2 O(l) Na. OH (aq) Na. Cl (aq) + H 2 O(l)

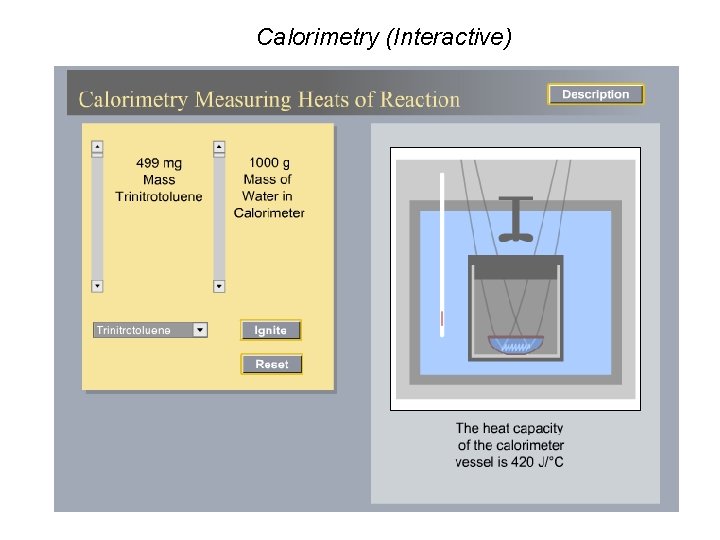
Calorimetry (Interactive)

Thermochemistry Lab Neutralization Reactions / Hess’s law Tfinal Tinitial

QUESTION

Report Table Vol. Solution (m. L) Densit Mass Specif y Solution ic Solution (g) Heat (g/m. L) Solution (J/g o. C) Tf (o. C) Ti (o. C) ΔT (o. C) Δ Hrxn (J) Results/Conclusions: answer questions #3, 4, & 6 pg. 41 (DVC Lab Manual) Δ Hrxn (k. J/m ol)

QUESTION

QUESTION


Exothermic Reaction http: //www. youtube. com/watch? v=rd. Csb. Zf 1_Ng


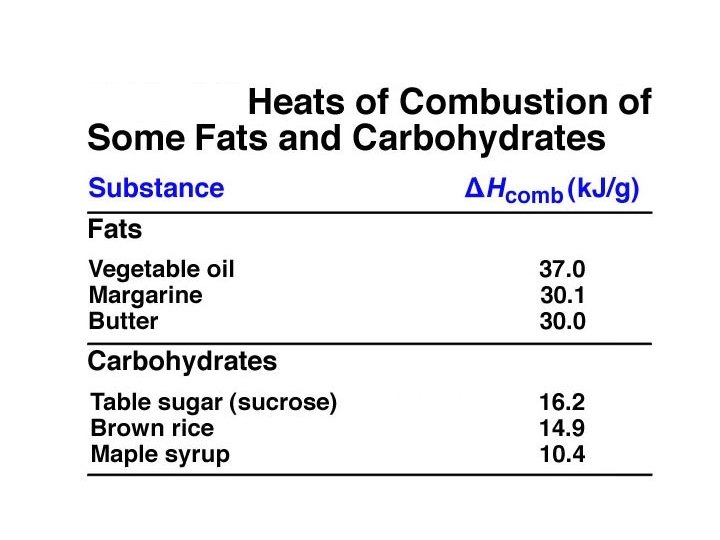
Heats of Combustion of octane releases 5, 470 k. J per mole of octane ( Hcomb = -5, 470 k. J/mol) ð How many gallons of water can be boiled by burning 1 gallon of gasoline? (Assume the water is at 25 o. C) ð How many grams of fat have the equivalent combustion energy as 1 gallon of gasoline? ð How many pounds of CO 2 are added to atmosphere from burning 1 gallon of gasoline? (This question relates to the Greenhouse Gas ð Workshop. )

Endothermic Reaction

QUESTION http: //chemconnections. org/general/chem 120/Flash/calorimetry_s. swf

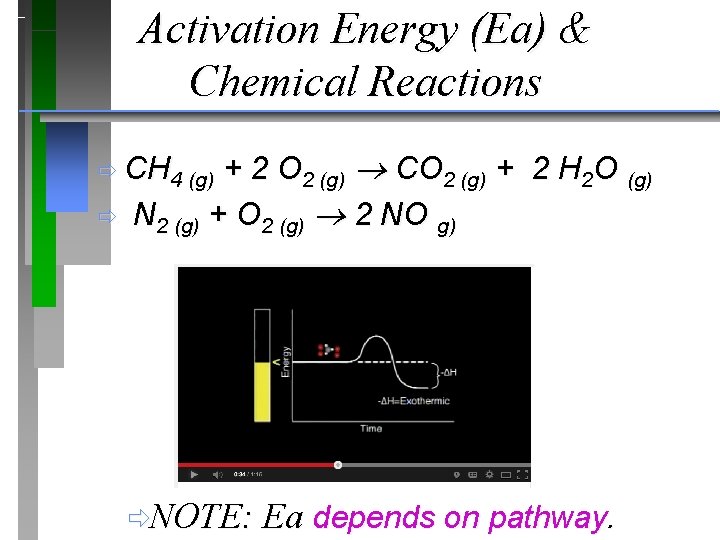
Activation Energy (Ea) & Chemical Reactions + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) N 2 (g) + O 2 (g) 2 NO g) ð CH 4 (g) ð ðNOTE: Ea depends on pathway.

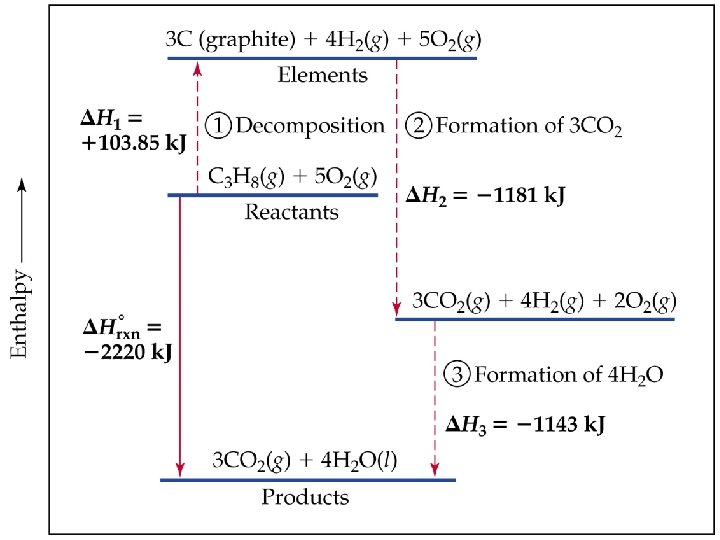
Hess’s Law Continued ð Reactants ð Products H = + (endothermic); H = - (exothermic) ð The change in enthalpy is the same whether the reaction takes place in one step or a series of steps.



Calculations via Hess’s Law ð 1. If a reaction is reversed, H is also reversed. N 2(g) + O 2(g) 2 NO(g) ð 2 NO(g) N 2(g) + O 2(g) ð ð H = 180 k. J 2. If the coefficients of a reaction are multiplied by an integer, H is multiplied by that same integer. ð 6 NO(g) 3 N 2(g) + 3 O 2(g) H = 540 k. J

QUESTION

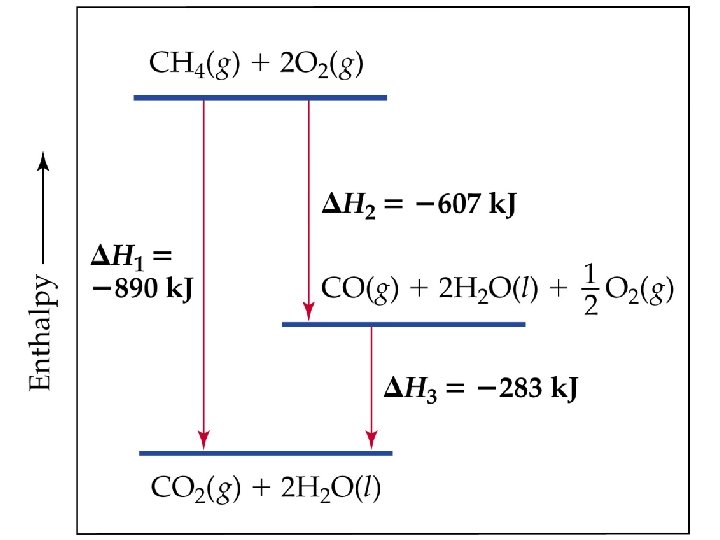
QUESTION Enthalpies of formation data are not always experimentally easy to obtain. However, enthalpies of combustion data are readily available. Calculate the enthalpy of formation of methane from the combustion data provided. C(graphite) + O 2(g) → CO 2(g) H 0 comb = − 393. 5 k. J H 2(g) + ½ O 2(g) → H 2 O(l) H 0 comb = − 285. 8 k. J CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(l) H 0 comb = − 890. 3 k. J A) − 19. 4 k. J/mol C) − 221. 9 k. J/mol B) − 74. 8 k. J/mol D) − 296. 0 k. J/mol