Introduction to Chemistry Mr Hrouda Cascade High School























































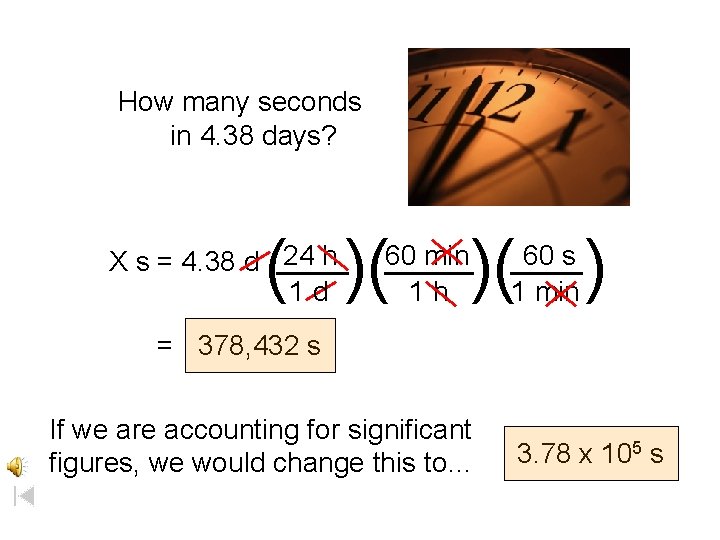















- Slides: 97

Introduction to Chemistry Mr. Hrouda Cascade High School

Chemistry is the study of the composition of matter, its chemical and physical changes, and the changes that accompany these changes.

…Matter is anything that occupies space. Chemistry Purpose with a

Areas of Chemistry • Organic The study of most carbon-containing compounds • Inorganic The study of all substances not classified as organic, mainly those compounds that do not contain carbon • Analytical The identification of the components and composition of materials • Physical The study of the properties, changes, and relationships between energy and matter • Biochemistry The study of substances and processes occurring in living things

Job Skills for the Future • • • Evaluate and Analyze Think Critically Solve Math Problems Organize and Use References Synthesize Ideas Apply Ideas to New Areas Be Creative Make Decisions with Incomplete Information Communicate in Many Modes Chemistry will develop ALL of these skills in YOU!

Careers in Chemistry research (new products) production (quality control) development (scale up manufacturing processes) chemical sales software engineering Pharmacist teaching The skills you will develop by an earnest study of chemistry will help you in any career field.

Research Basic Research – Carried out for the sake of increasing knowledge – Driven by curiosity or a desire to know – Roy Plunkett ‘discovers’ Teflon is a nonstick material Applied Research – Carried out to solve a specific problem – Safer refrigerant that does not harm ozone layer Technological Development – Production and use of products that improve our quality of life – Computer chips, biodegradable materials, catalytic converters for automobiles

Safety

Basic Safety Rules Use common sense. No unauthorized experiments. No horseplay. Handle chemicals/glassware with respect.

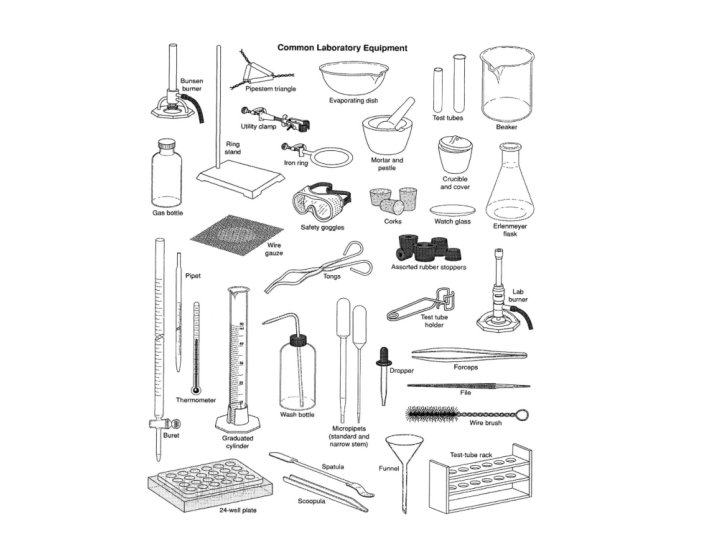
Safety Features of the Lab safety shower fire blanket fire extinguisher eye wash fume hood circuit breaker switch

Government Regulation of Chemicals The government regulates chemicals to reduce the risk to the… • Consumer FDA, USDA, Consumer Product Safety Commission • Worker OSHA • Environment EPA Chemical Stewardship

Thalidomide • Prescription drug for morning sickness • Drug can be made in two ways – Put together same material in more than one way. • A = “good” drug (stops morning sickness) • B = “bad” drug (birth defects) • Side-effect from “bad” drug – Stopped development in fetus • Short arms; “flipper-babies”

Safety Symbols Eye Protection Required Heat Protection Clothing Protection Required Glassware Safety Hand Protection Required Laboratory Hygiene Chemical Safety Sharp Object Hazard Caustic Substance Waste Disposal

Safety Equipment Safety Goggles Fire Extinguisher Type A Type B Type C Safety Shower

Chemical Burns Flammable Health Reactive Special Chemical burns on feet. Skin burned by chemicals

SAFETY in the Science Classroom Obey the safety contract – Use common sense – No unauthorized experiments – Wear safety glasses – Safety is an attitude! – Don’t take anything out of lab – Read and follow all instructions


20 ? 15 ? 1 m. L 1. 50 15. 0 xm. L 10

Material Safety Data Sheet (MSDS) • Gives information about a chemical. • Lists “Dos” and “Don’ts. ”

Knowledge = Safety • Material Safety Data Sheet (MSDS) – Lists hazards, special handling instructions, and risks associated with a material. Supplied by manufacturer. • Acute Exposure – Single episode can cause great damage • Chronic Exposure – Many episodes over a period of time cause damage • • Carcinogen – causes cancer Mutagen – causes mutations (genetic defects) Tetragen – causes birth defects Neurotoxin – severely poisonous and toxic

Scientific Law vs. Scientific Theory A law states what happens. Law of Gravity A theory tries to explain why or how something happens. Theory of Gravity Atomic Theory Collision Theory of Reactions

Experiments • Law – A verbal or mathematical description of a phenomenon that allows for general predictions – Describes what happens and not why – Unlikely to change greatly over time unless a major experimental error is discovered • Theory Attempts to explain why nature behaves as it does – Is incomplete and imperfect, evolving with time to explain new facts as they are discovered – Copyright 2007 Pearson Benjamin Cummings. All rights reserved.

The Way Science Works… • Science involves critical thinking, or applying logic and reason to observations and conclusions.

How does scientific knowledge advance? 1. curiosity 2. good observations 3. determination 4. persistence

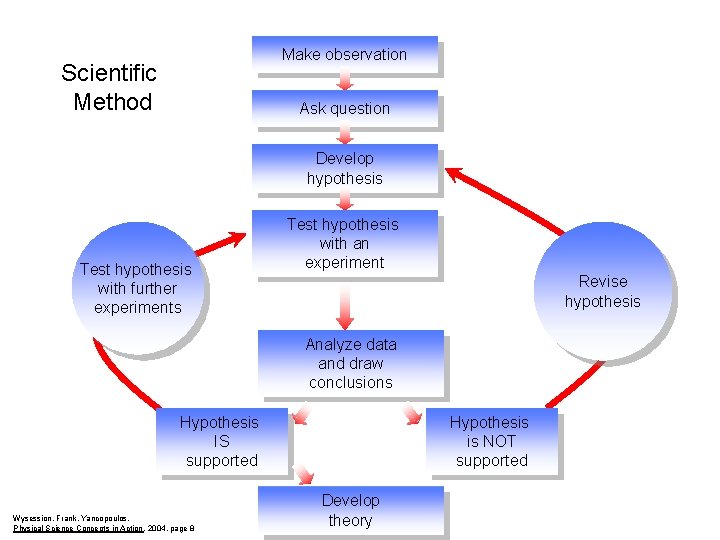
The Scientific Method A series of logical steps to follow in order to solve problems.

Parts of the Scientific Method • OBSERVE • FORMULATE A QUESTION • FORM A HYPOTHESIS • DESIGN AND CONDUCT AN EXPERIMENT • MAKE OBSERVATIONS • RECORD ANALYZE DATA • DRAW CONCLUSIONS • FORMULATE NEW QUESTIONS and CONTINUE CYCLE

Make observation Scientific Method Ask question Develop hypothesis Test hypothesis with further experiments Test hypothesis with an experiment Revise hypothesis Analyze data and draw conclusions Hypothesis IS supported Wysession, Frank, Yancopoulos, Physical Science Concepts in Action , 2004, page 8 Hypothesis is NOT supported Develop theory

Using the scientific method requires that one be a good observer. observation uses the five senses inference involves a judgment or assumption

“My mother the eye doctor” Observation or Inference? The One Names The piece players adult mother of the player areis ofteams holding isof written the holding in paper holding a the catcher. umpire. isbatter picture on the iscalled aaathe piece note bat. isuniforms. are the is an of from talking. on optometrist “Reds. ” paper. opposite the mother or teams. opthalmologist of the batter. (an eye doctor). wearing abat catcher’s mask.

Data Observations are also called data. There are two types of data. qualitative data quantitative data descriptions; no numbers measurements; must have numbers and UNITS

Variables and Controls • A variable is anything that can change in an experiment. – Independent variable: The variable being changed or controlled by the scientist. – Dependent variable: The variable being measured or observed by the scientist. • A controlled experiment tests only one variable at a time.

Variables • Independent causes a change in Dependent D – dependent M – manipulative R – responsive I – independent Y – y-axis X – x-axis What is being tested measured What is changed Result

Control Group Purpose: To have something to compare the results to at the end of the experiment IS NOT exposed to the independent variable!

A Scientific Experiment procedure the order of events in an experiment; the “recipe” variable any factor that could influence the result Experiments must be controlled; they must have two set-ups that must differ by only one variable. The conclusion must be based on the data.

A Controlled Experiment?

Chemistry and Manipulating Numerical Data

Measurements v science is based on measurements Numbers v mathematics is based on numbers v exact numbers are v all measurements have: obtained by: - counting - magnitude - definition - uncertainty - units

Making Measurements • Measurements are made in this class using SI units. • LENGTH (m): distance between 2 points • VOLUME (L): space occupied. • MASS (kg): the amount of matter in an object. • WEIGHT (N): the force with which gravity pulls on a quantity of matter.

No Cussing! The following 4 -Letter words are forbidden here: Inch Foot Yard Mile Pint Acre And we never swear the BIG F (useo. C) Please keep it clean and Metric

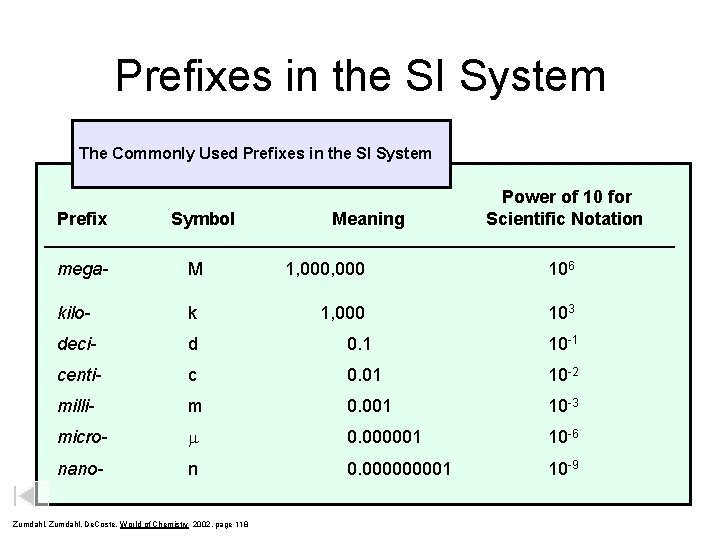
Prefixes in the SI System The Commonly Used Prefixes in the SI System Prefix Symbol Meaning Power of 10 for Scientific Notation ____________________________________ mega- M kilo- k deci- d 0. 1 10 -1 centi- c 0. 01 10 -2 milli- m 0. 001 10 -3 micro- m 0. 000001 10 -6 nano- n 0. 00001 10 -9 Zumdahl, De. Coste, World of Chemistry 2002, page 118 1, 000 106 1, 000 103

A physical quantity must include: Number + Unit

Scientific Notation Scientist use special notation to express or very small numbers. very large Example I: 300, 000 g can be written as… 3 x 108 g Ex II: 1, 007, 000 sec can be written as… 1. 007 x 109 sec Ex III: 0. 000 000 004 76 m. L can be written as… 4. 76 x 10 -12 m. L

Method to express really big or small numbers. Format is Mantissa x Decimal part of original number 6. 02 x Base Power Decimal you moved 23 10 We just move the decimal point around. 60200000000000

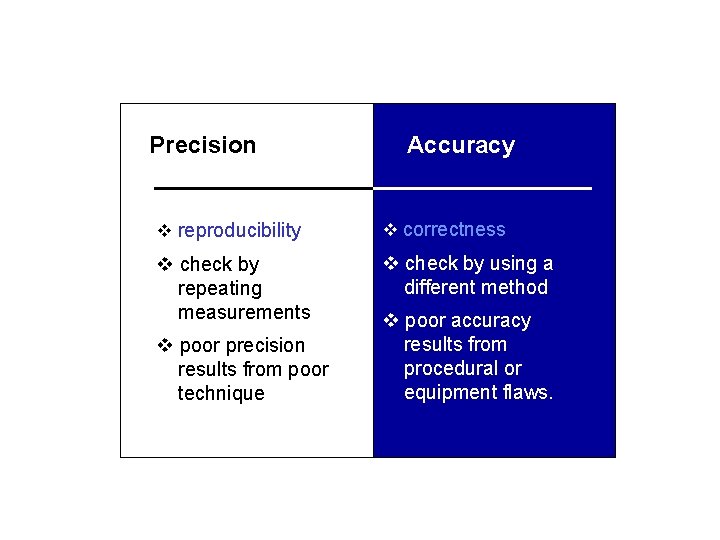
Precision v reproducibility Accuracy v correctness v check by using a v check by different method repeating measurements v poor accuracy v poor precision results from poor technique results from procedural or equipment flaws.

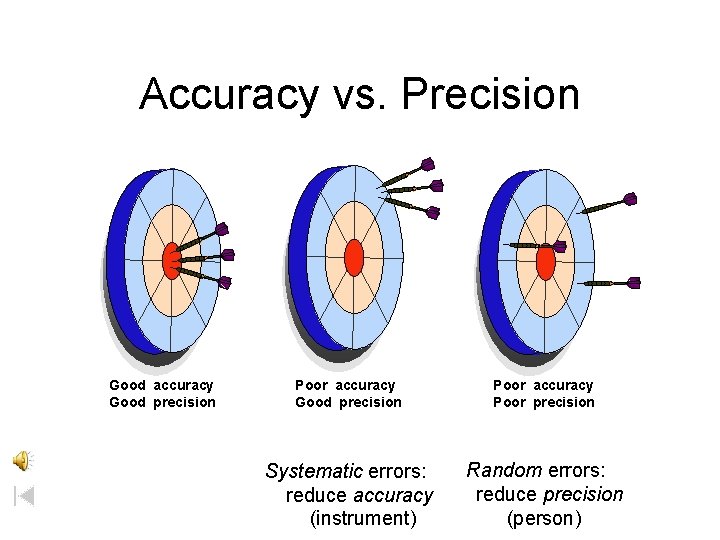
Accuracy vs. Precision Good accuracy Good precision Poor accuracy Poor precision Systematic errors: reduce accuracy (instrument) Random errors: reduce precision (person)

Density = mass/volume • Example: • – What is the mass of 10 m. L of a • liquid that has a density of 3. 76 • g/m. L? • d = m / v • m = dv • m = (3. 76 g/m. L)(10 m. L) • m = 37. 6 g • m = 40 g (correct # of sig. figs. )

Temperature Conversions • Temperature is a measure of the average kinetic energy in a system. • K = Kelvin • °F = degree Fahrenheit • °C = degree Celsius

Temperature Conversions K = °C + 273 °F = (1. 8 x °C) + 32 °C = (°F – 32) / 1. 8

Percentage Error • Calculate this value in labs where the accepted value is given. • %error = Accepted value - measured value x 100 Accepted value Accepted = actual Measured = theoretical

Reviewing Concepts Measurement • Why do scientists use scientific notation? • What system of units do scientists use for measurements? • How does the precision of measurements affect the precision of scientific calculations? • List the SI units for mass, length, and temperature.

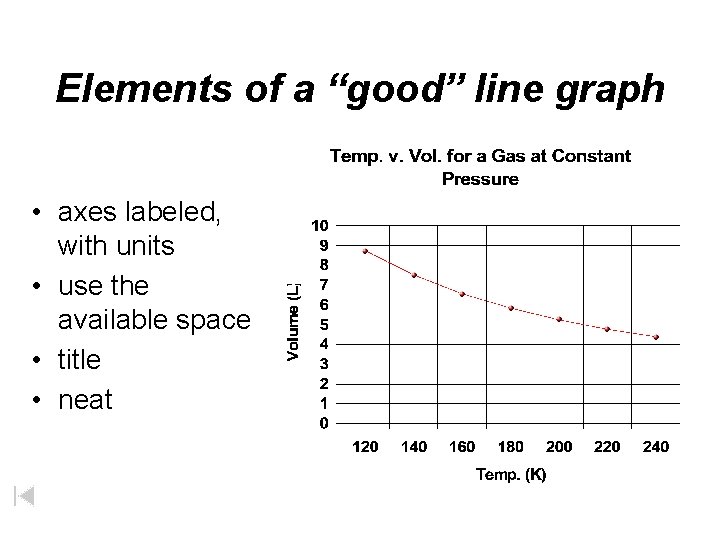
Organizing Data is organized and presented in tables, charts, and graphs Graph – visual representation of data 1)title 2) x and y axis labeled 3) units for both the x and y axis 4) scale is evenly and correctly spaced for data 5) legend when appropriate

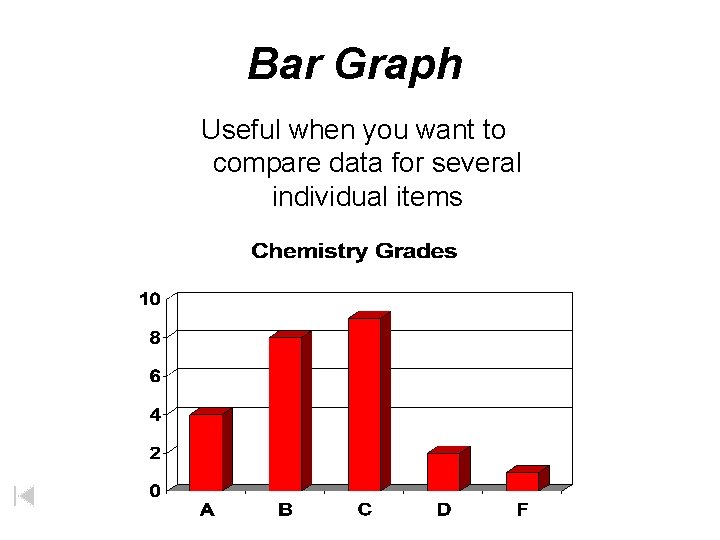
Bar Graph Useful when you want to compare data for several individual items

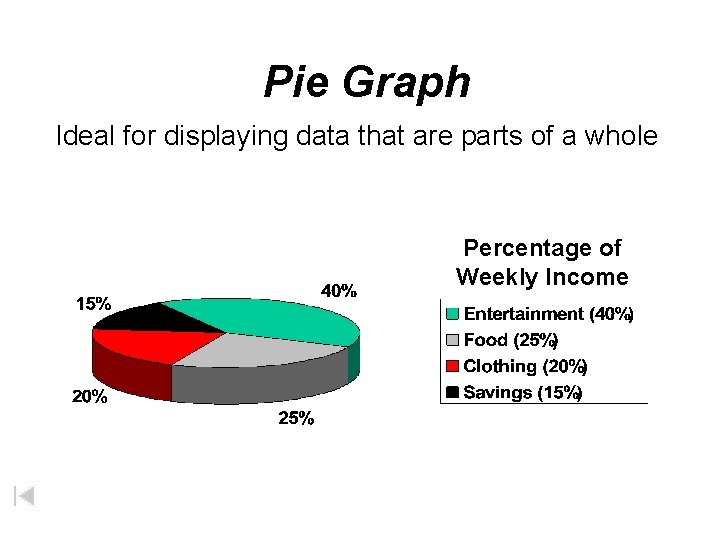
Pie Graph Ideal for displaying data that are parts of a whole Percentage of Weekly Income

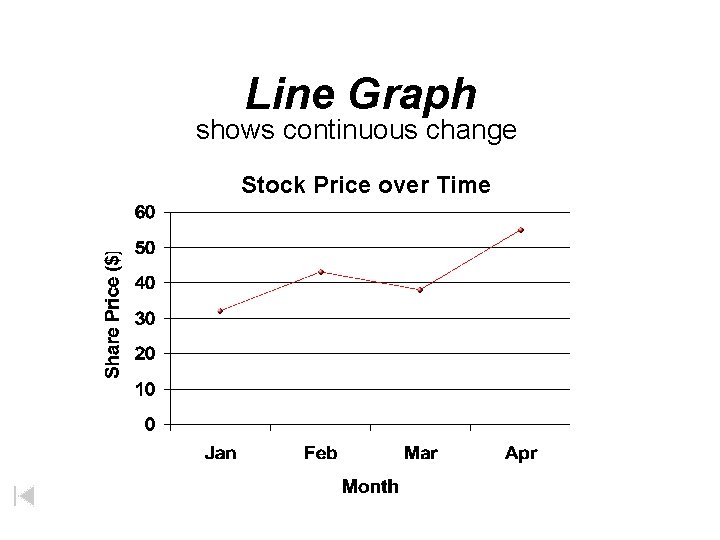
Line Graph shows continuous change Stock Price over Time

Elements of a “good” line graph • axes labeled, with units • use the available space • title • neat

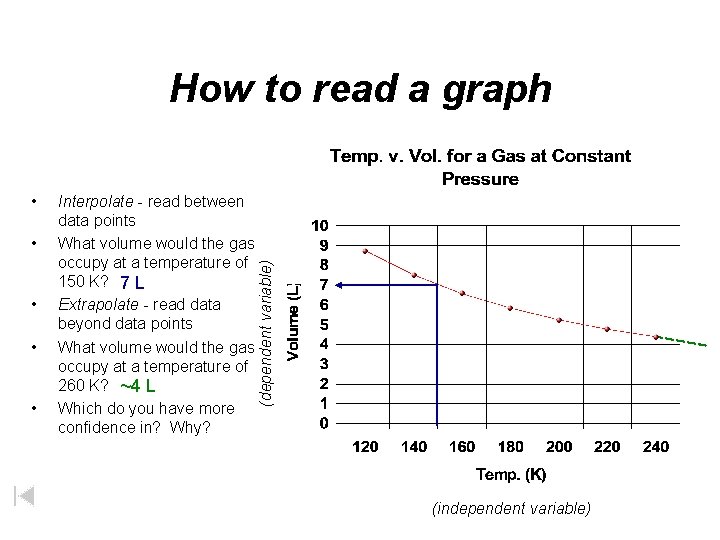
• • • Interpolate - read between data points What volume would the gas occupy at a temperature of 150 K? 7 L Extrapolate - read data beyond data points What volume would the gas occupy at a temperature of 260 K? ~4 L Which do you have more confidence in? Why? (dependent variable) How to read a graph (independent variable)

Graphs • Line Graph – Used to show trends or continuous change • Bar Graph – Used to display information collected by counting • Pie Graph – Used to show some fixed quantity is broken down into parts

Reviewing Concepts Presenting Scientific Data • How do scientists organize data? • How can scientists communicate experimental results? • What does a given point represent on a line graph?

Performing these conversions is a lot like going on a road trip.


Converting Metric Measurements To convert, move the decimal place the number of stairs you step on in the direction you are traveling. kilo- 103 hecto- 102 deca- 101 Example: 3. 75 km = ? mm ? = 3, 750, 000 mm basic unit 1 deci- 10 -1 centi- 10 -2 milli- 10 -3

Steps to Dimensional Analysis Determine your target What do you want to know? Determine what you already know Set up the problem Units New Units! Units Solve the Problem Check if your answer is reasonable… if not start over Sig Fig your final answer Same # of sig figs that you started with

Use dimensional analysis when converting between other units. • Ex: How many inches are in 1500 meters?

Calculation Corner: Unit Conversion 1 foot = 12 inches

Calculation Corner: Unit Conversion 1 foot 12 inches 1 foot “Conversion factors” ( 3 feet )( 12 inches 1 foot ) = 36 inches

How many cm are in 1. 32 meters? equality: 1 m = 100 cm (or 0. 01 m = 1 cm) applicable conversion factors: 1 m ______ 100 cm or ( 100 cm ______ 1 m ) ______ cm = 132 cm X cm = 1. 32 m 100 1 m We use the idea of unit cancellation to decide upon which one of the two conversion factors we choose.

How many meters is 8. 72 cm? equality: 1 m = 100 cm applicable conversion factors: 1 m ______ 100 cm or ( 100 cm ______ 1 m ) 1 m X m = 8. 72 cm ______ = 0. 0872 m 100 cm Again, the units must cancel.

How many feet is 39. 37 inches? equality: 1 ft = 12 in applicable conversion factors: ______ 1 ft 12 in X ft = 39. 37 in or ______ 12 in 1 ft ( ) ____ 1 ft = 3. 28 ft 12 in Again, the units must cancel.

How many kilometers is 15, 000 decimeters? ( )( 1 m X km = 15, 000 dm ____ 10 dm ) 1 km ______ = 1. 5 km 1, 000 m
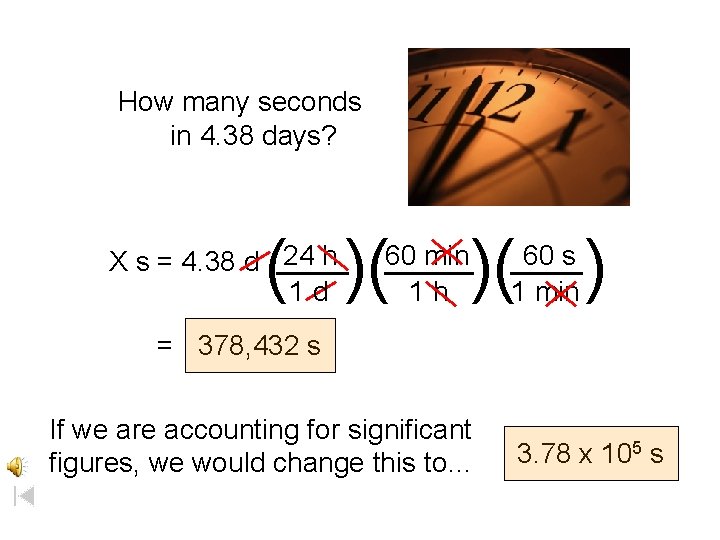
How many seconds in 4. 38 days? ( )( 24 h X s = 4. 38 d ____ 1 d )( ) 60 min _____ 1 h 60 s ____ 1 min = 378, 432 s If we are accounting for significant figures, we would change this to… 3. 78 x 105 s

Significant Figures What is the point? ! Simply: They tell us how to round The real point: To make sure that the answer you get isn’t more precise than the numbers you started with

Rules for Counting Significant Figures 1. Nonzero integers always count as significant figures. 2. Zeros: There are three classes of zeroes. a. Leading zeroes precede all the nonzero digits and DO NOT count as 2 significant figures. Example: 0. 0025 has ____ b. Captive zeroes are zeroes between nonzero numbers. These always 4 significant figures. count as significant figures. Example: 1. 008 has ____ c. Trailing zeroes are zeroes at the right end of the number. Trailing zeroes are only significant if the number contains a decimal point. 3 significant figures. Example: 1. 00 x 102 has ____ Trailing zeroes are not significant if the number does not contain a decimal 1 significant figure. point. Example: 100 has ____ 3. Exact numbers, which can arise from counting or definitions such as 1 in = 2. 54 cm, never limit the number of significant figures in a calculation. Ohn-Sabatello, Morlan, Knoespel, Fast Track to a 5 Preparing for the AP Chemistry Examination 2006, page 53

Significant figures: Rules for zeros Leading zeros are not significant. Leading zero 0. 421 – three significant figures Captive zeros are significant. Captive zero 4012 – four significant figures Trailing zeros are significant. Trailing zero 114. 20 – five significant figures

Significant Figures Atlantic - Pacific Rule: Decimal Present: Count from the Pacific side Decimal Absent: Count from the Atlantic side Start counting at the first non-zero number and count until you reach the end of the number Ex. I: 3. 00700 Decimal Present… Pacific (left)… 6 sig. figs. Ex. II: 300, 700 Decimal Absent… Atlantic (right)… 4 sig. figs.

Significant Figures Counting Sig Fig Examples 1. 23. 50 4 sig figs 2. 402 3 sig figs 3. 5, 280 3 sig figs 4. 0. 080 2 sig figs Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

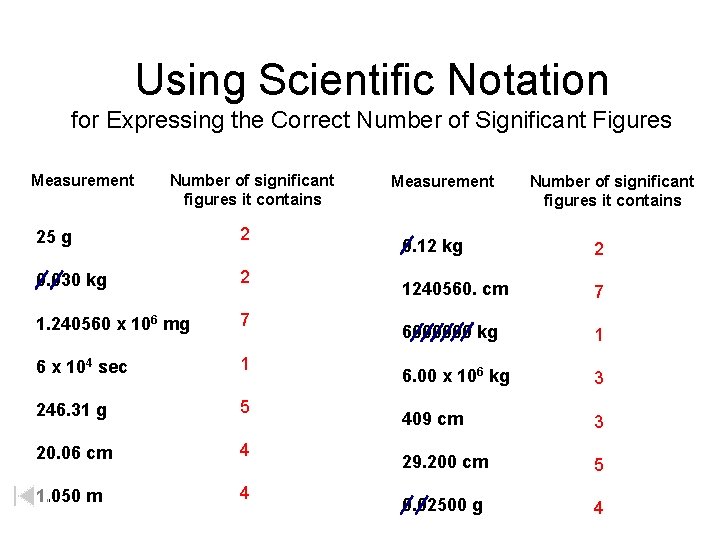
Using Scientific Notation for Expressing the Correct Number of Significant Figures Measurement Number of significant figures it contains 25 g 2 0. 030 kg 2 1. 240560 x 106 mg 7 6 x 104 sec 1 246. 31 g 5 20. 06 cm 4 1. 050 m 4 Measurement Number of significant figures it contains 0. 12 kg 2 1240560. cm 7 6000000 kg 1 6. 00 x 106 kg 3 409 cm 3 29. 200 cm 5 0. 02500 g 4

Before we take this trip, let’s review significant figures… Determine how many significant figures are in each of the following measurements. 1) 2) 3) 4) 5) 6) 5 0. 0034050 L ______ 5 33. 600 m ______ 7500. 0 g ______ 5 3 47, 900 mm ______ 10 7, 000, 001 miles ______ 8. 07 Hz ______ 3

More practice… Round the following measurements off so that they each contain 3 significant figures. 7) 366. 2 L ______ 366 L 9, 050, 000 mg 8) 9, 047, 022 mg ______ 9) 12. 76 g ______ 12. 8 g 1. 00 x 103 J 10) 999. 9 J ______ Notice this one must be in scientific notation to have 3 sig. figs.

Significant Figures in Calculations When multiplying and dividing, limit and round to the least number of significant figures in any of the factors. Example: 23. 0 x 432 x 19 = 188, 784 = 190, 000 The answer is expressed as 190, 000 or 1. 9 x 105 since 19 has only two sig. figs.

Significant Figures in Calculations When adding and subtracting, limit and round your answer to the least number of decimal places in any of the numbers involved in the calculation. Example: 123. 25 + 46. 0 +86. 257 = 255. 507 = 255. 5 The answer is expressed as 255. 5 since 46. 0 has only one decimal place.

Before we take this trip, let’s review significant figures… Perform the prescribed operations. Round your answers to the proper # of sig. figs. 11) 36. 57 m / 3. 21 s = ______ 11. 4 m/s 97. 6 g 12) 41. 376 g + 13. 3 g + 42. 9 g=______ 76. 2 m 2 13) 5. 67 m x 13. 44 m ______ 14) (5. 83 m/ 2. 67 s) /2. 1 s ______ 1. 0 m/s 2 60 V 15) 9. 374 V x 6 ______ From now on, we will round all our answers to the correct # of significant figures.

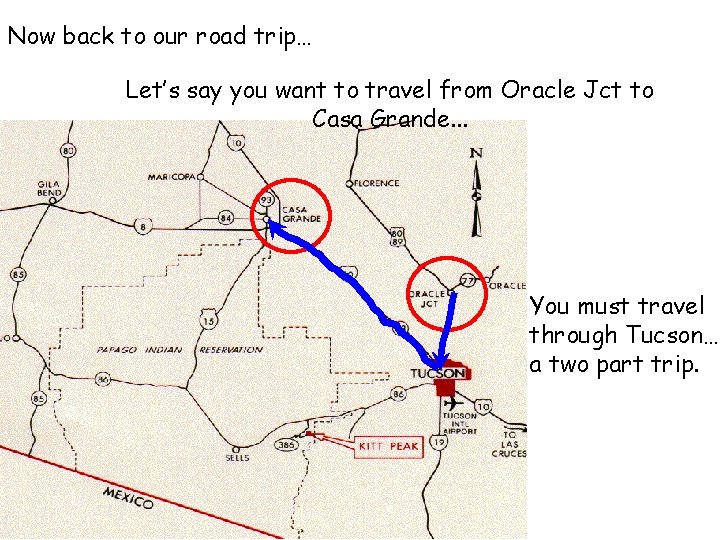
Now back to our road trip… Let’s say you want to travel from Oracle Jct to Casa Grande. . . You must travel through Tucson… a two part trip.

Rule: You must stay on the roads

X

Rule: You need a map. Sometimes the map can be in your head. But, not at first.

OK…Lets talk about making maps and solving conversion problems. The method we use is called the “Factor-Label” method, a. k. a. dimensional analysis.

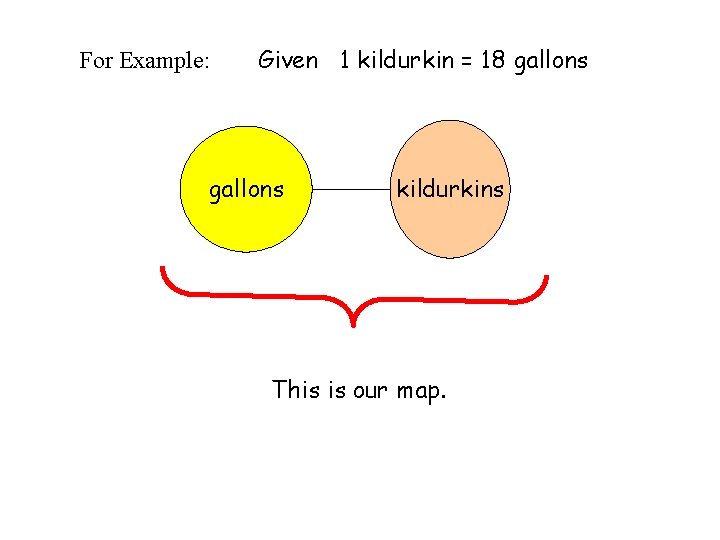
For Example: Given 1 kildurkin = 18 gallons kildurkins This is our map.

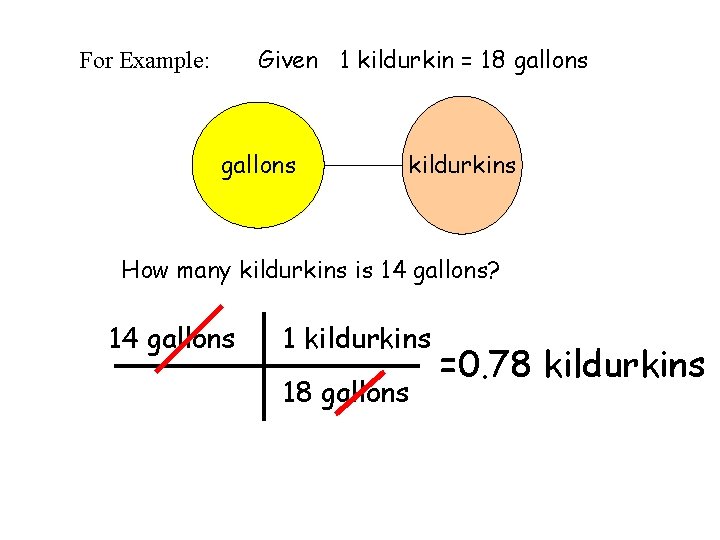
Given 1 kildurkin = 18 gallons For Example: gallons kildurkins How many kildurkins is 14 gallons? 14 gallons 1 kildurkins 18 gallons =0. 78 kildurkins

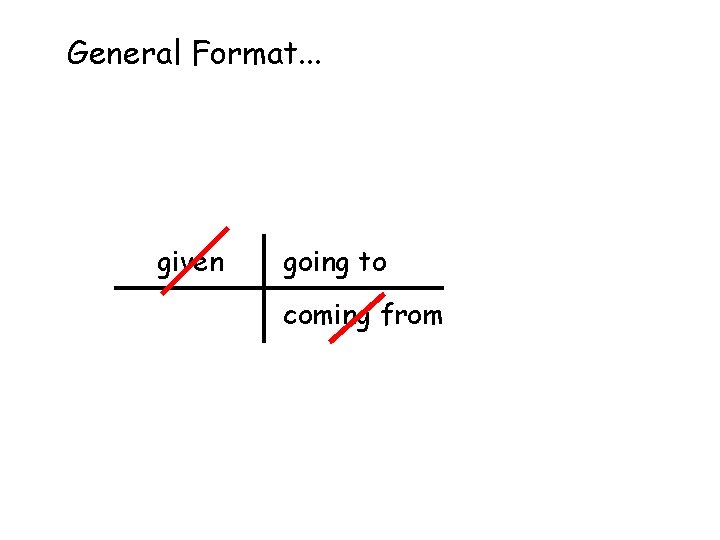
General Format. . . given going to coming from

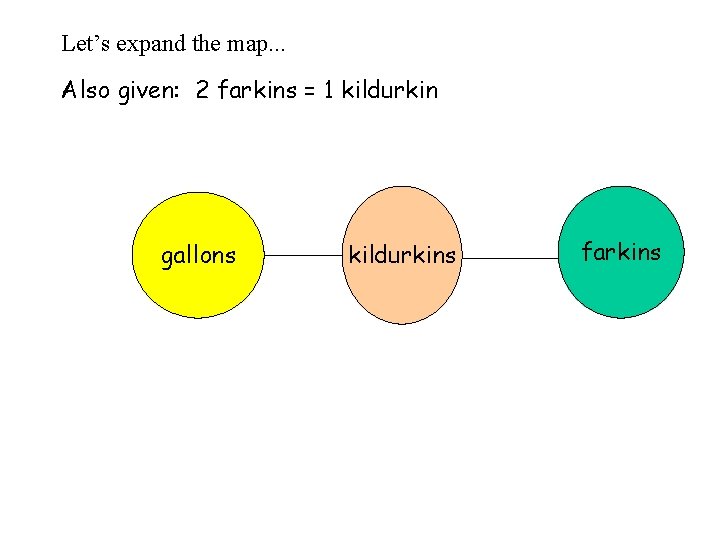
Let’s expand the map. . . Also given: 2 farkins = 1 kildurkin gallons kildurkins farkins

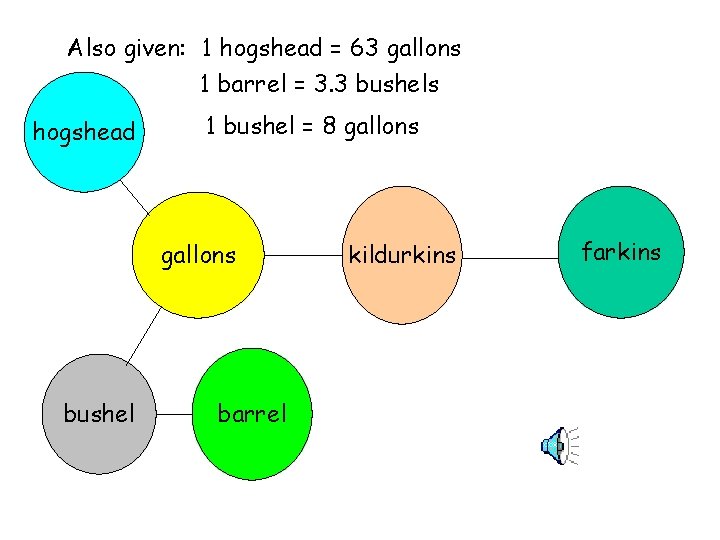
Also given: 1 hogshead = 63 gallons 1 barrel = 3. 3 bushels hogshead 1 bushel = 8 gallons bushel barrel kildurkins farkins

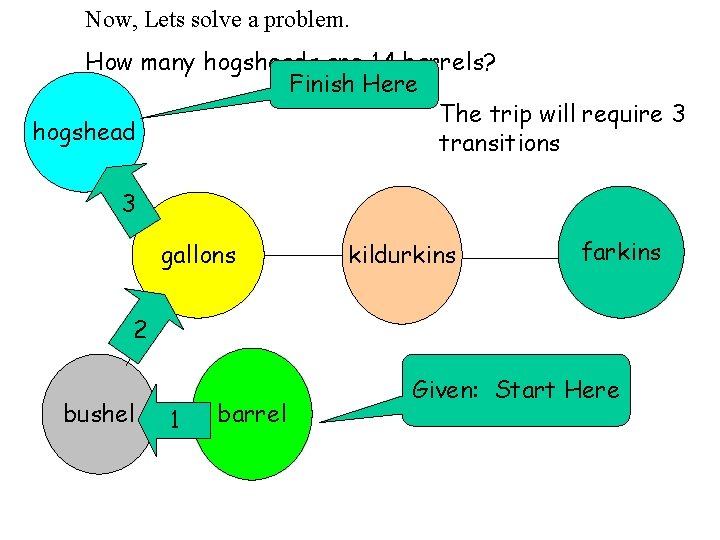
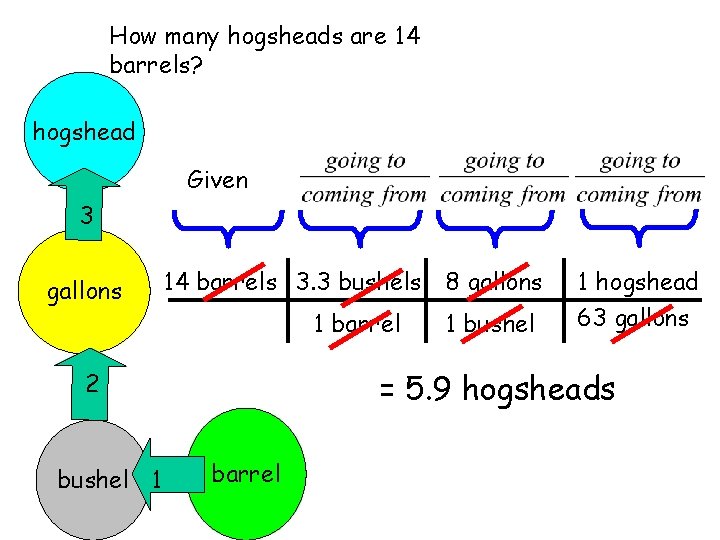
Now, Lets solve a problem. How many hogsheads are 14 barrels? Finish Here The trip will require 3 hogshead transitions 3 gallons kildurkins farkins 2 bushel 1 barrel Given: Start Here

How many hogsheads are 14 barrels? hogshead Given 3 gallons 14 barrels 3. 3 bushels 1 barrel 1 bushel 1 hogshead 63 gallons = 5. 9 hogsheads 2 bushel 1 8 gallons barrel

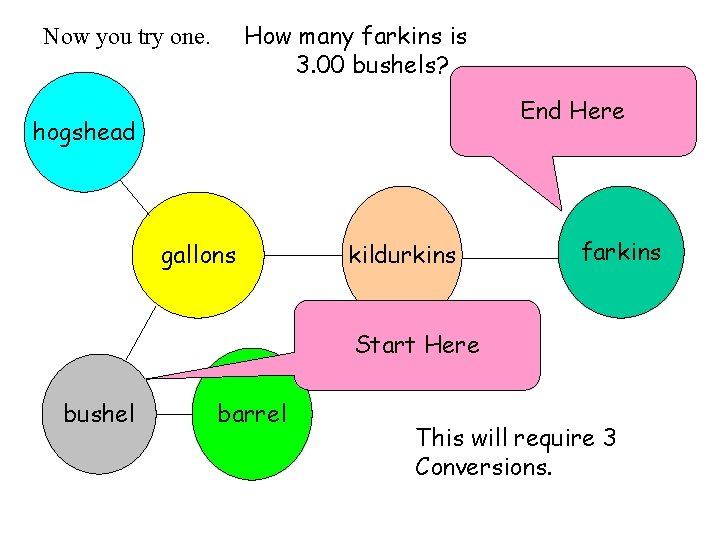
How many farkins is 3. 00 bushels? Now you try one. End Here hogshead gallons kildurkins farkins Start Here bushel barrel This will require 3 Conversions.

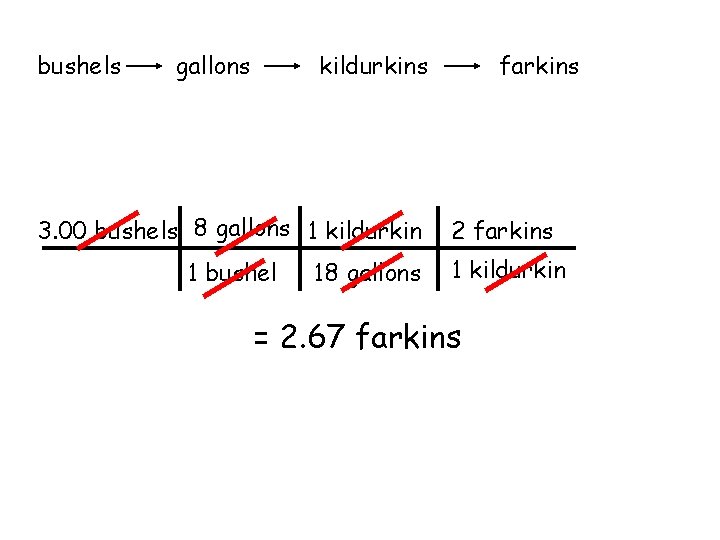
bushels gallons farkins kildurkins 3. 00 bushels 8 gallons 1 kildurkin 2 farkins 1 bushel 1 kildurkin 18 gallons = 2. 67 farkins

A General Procedure for Solving Problems • Read the problem carefully and make a list of the “knowns” and the ‘unknowns” • Look up all needed information – Your lecture notes will have much, if not all, of the needed information • Work out a plan and, following your plan, obtain an answer by carrying out the required math. • Check over your work – This is best done by estimating your answer – Ask yourself: “Does the answer seem reasonable? ”

How to Succeed in Chemistry • • • Learn the language Use the illustrations Review your notes frequently Work as many problems as possible Do NOT cram for exams.