NUCLEAR CHEMISTRY Introduction to Nuclear Chemistry Nuclear chemistry















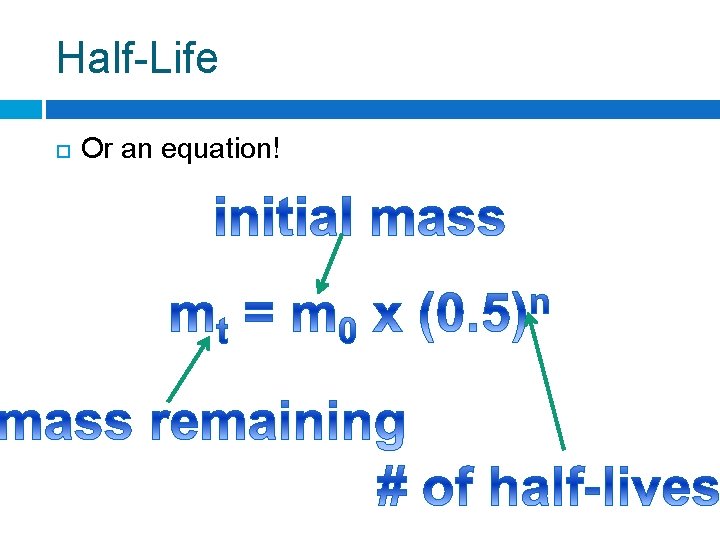











- Slides: 46

NUCLEAR CHEMISTRY

Introduction to Nuclear Chemistry Nuclear chemistry is the study of the structure ofatomic nuclei and changes they undergo.

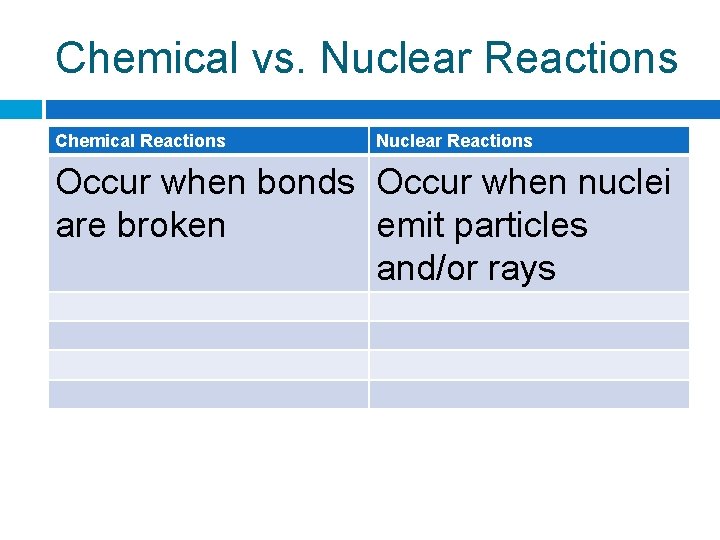
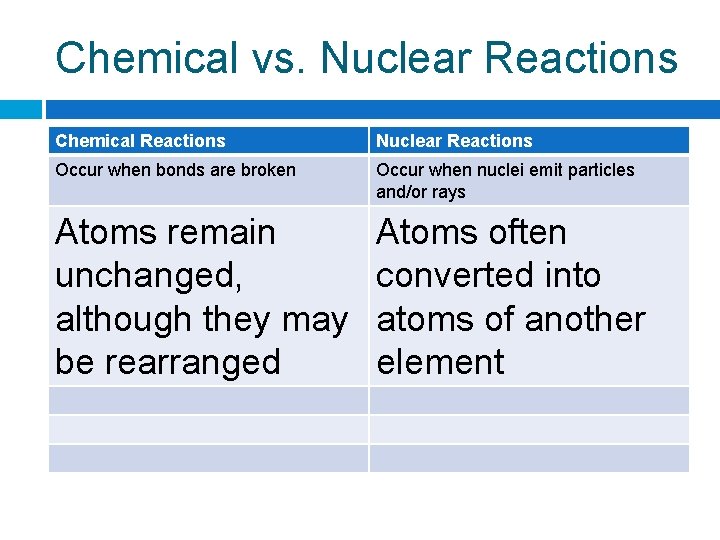
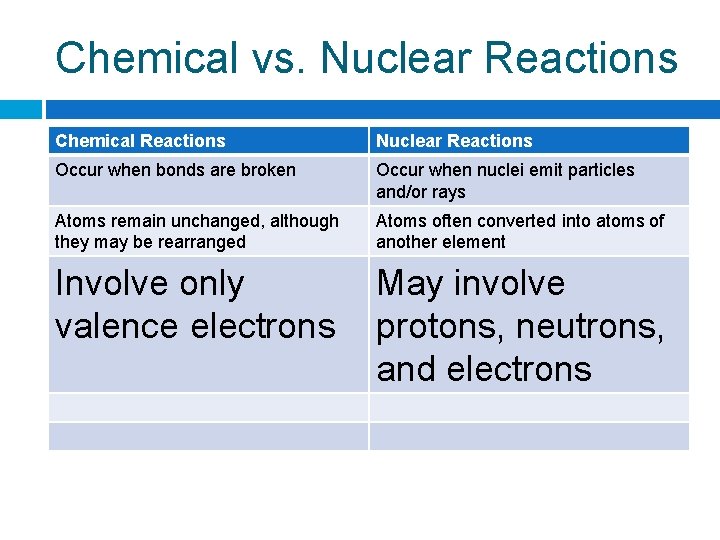
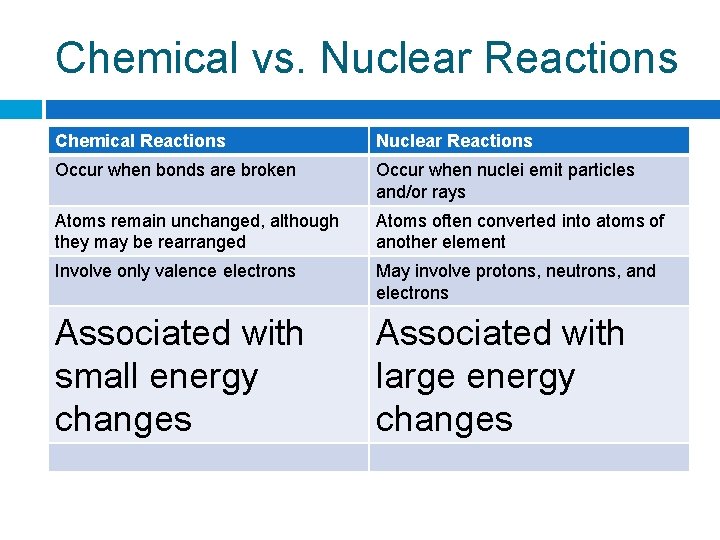
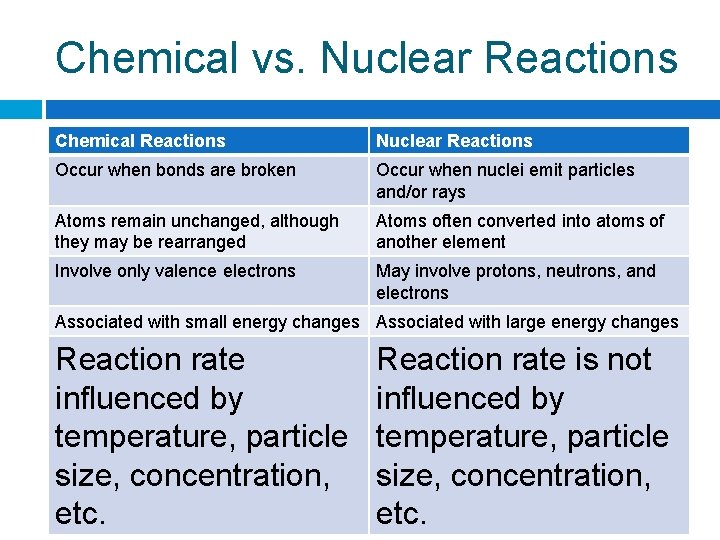
Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds Occur when nuclei are broken emit particles and/or rays

Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element

Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons

Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons Associated with small energy changes Associated with large energy changes

Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons Associated with small energy changes Associated with large energy changes Reaction rate influenced by temperature, particle size, concentration, etc. Reaction rate is not influenced by temperature, particle size, concentration, etc.

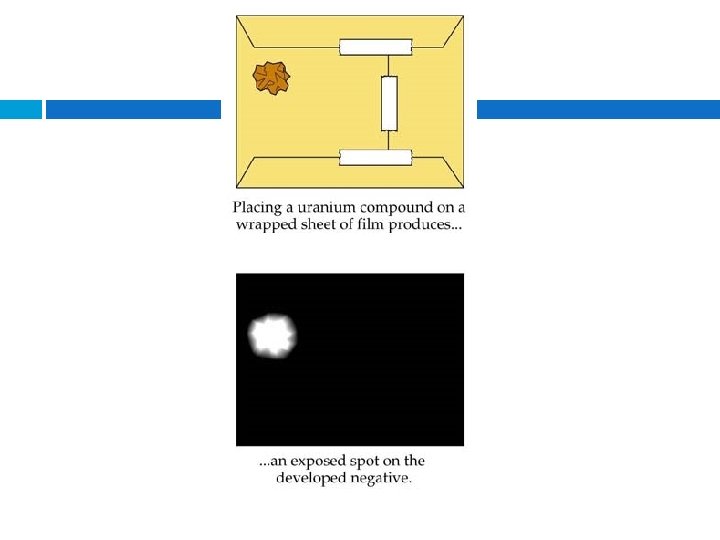
The Discovery of Radioactivity (1895 – 1898): found that invisible rays were emitted when electrons bombarded the surface of certain materials. Becquerel accidently discovered that phosphorescent salts produced spontaneous emissions that darkened photographic plates

Antoine Henri Becquierel


Radioactive decay Discovered by Antoine Henri Becquerel in 1896 He saw that photographic plates developed bright spots when exposed to uranium metals

Radioactive Decay – nucleus decays spontaneously giving off an energetic particle

The Discovery of Radioactivity (1895 – 1898): isolated the components ( atoms) emitting the rays – process by which particles give off – the penetrating rays and particles by a radioactive source

The Discovery of Radioactivity (1895 – 1898): identified 2 new elements, polonium and on the basis of their radium radioactivity contradicted These findings Dalton’s theory of indivisible atoms.

Marie Sklodowska Curie with her daughter, Irene.

The Discovery of Radioactivity (1895 – 1898): – atoms of the element with different numbers of – isotopes of atoms with nuclei (too / neutrons) – when unstable nuclei energy by emitting to attain more atomic configurations ( process)

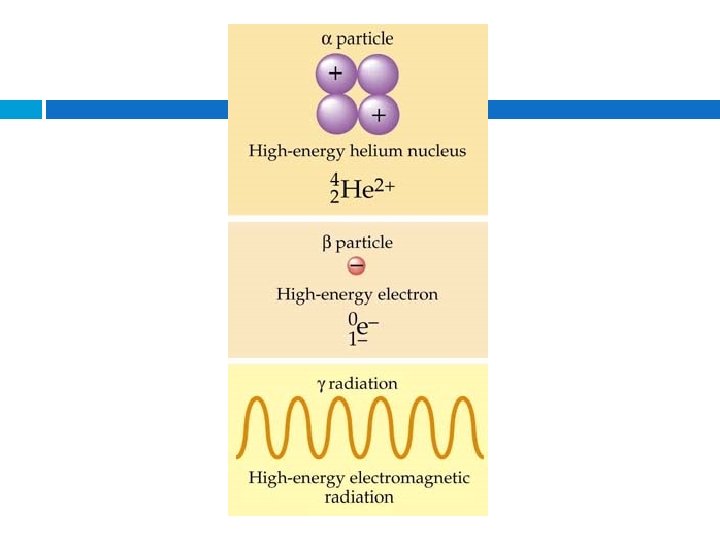
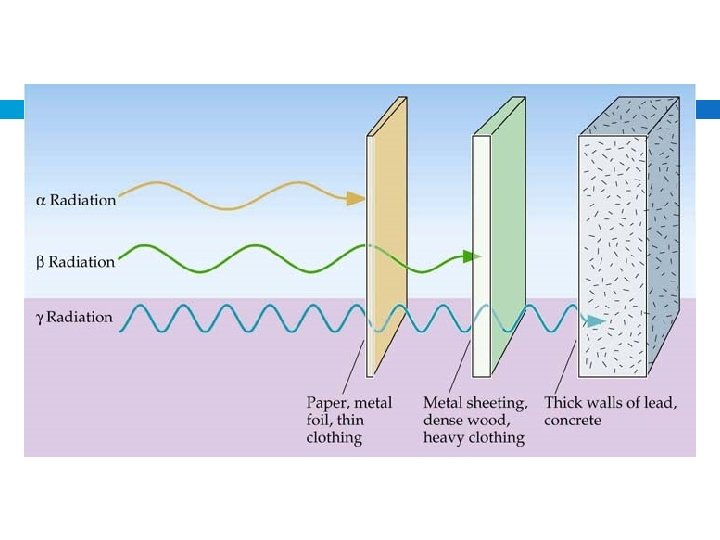
Alpha radiation Composition – Alpha particles, same as helium 4 nuclei 2 Symbol – Helium nuclei, He, α Charge – 2+ Mass (amu) – 4 Approximate energy – 5 Me. V Penetrating power – low (0. 05 mm body tissue) Shielding – paper, clothing

Beta radiation Composition – Beta particles, same as an electron Symbol – e-, β Charge – 1 Mass (amu) – 1/1837 (practically 0) Approximate energy – 0. 05 – 1 Me. V Penetrating power – moderate (4 mm body tissue) Shielding – metal foil

Gamma radiation Composition – High-energy electromagnetic radiation Symbol – γ Charge – 0 Mass (amu) – 0 Approximate energy – 1 Me. V Penetrating power – high (penetrates body easily) Shielding – lead, concrete



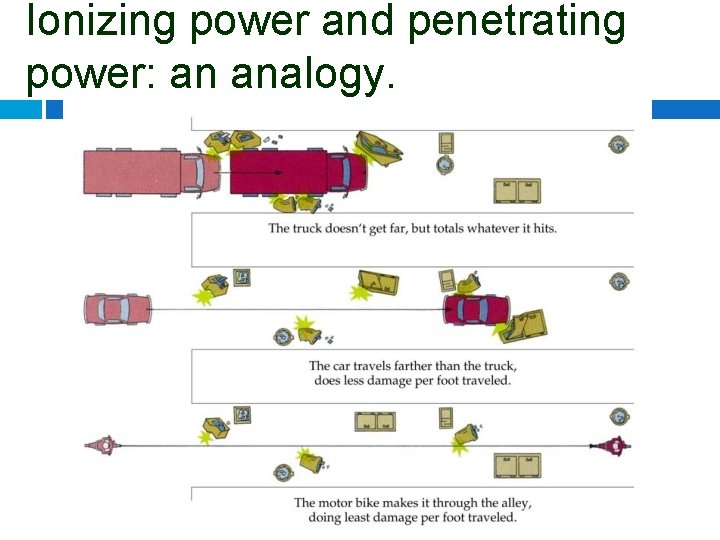
Ionizing power and penetrating power: an analogy.

Types of radioactive decay alpha particle emission beta emission positron emission electron capture gamma emission

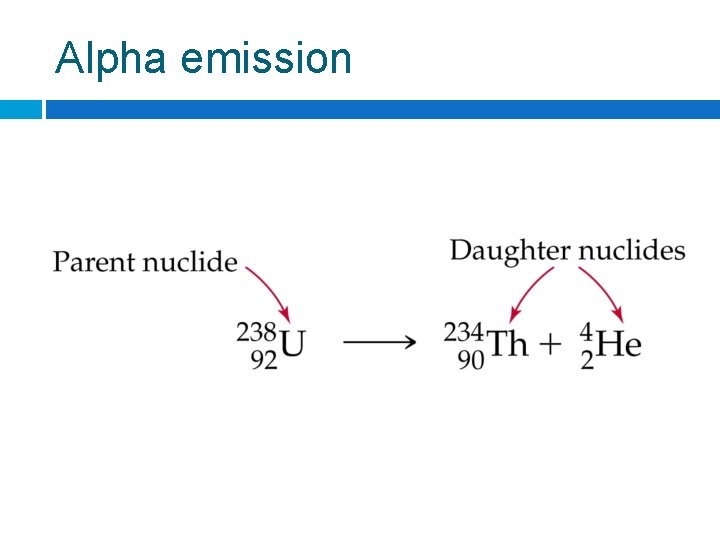
Alpha emission

Beta Particle emisson

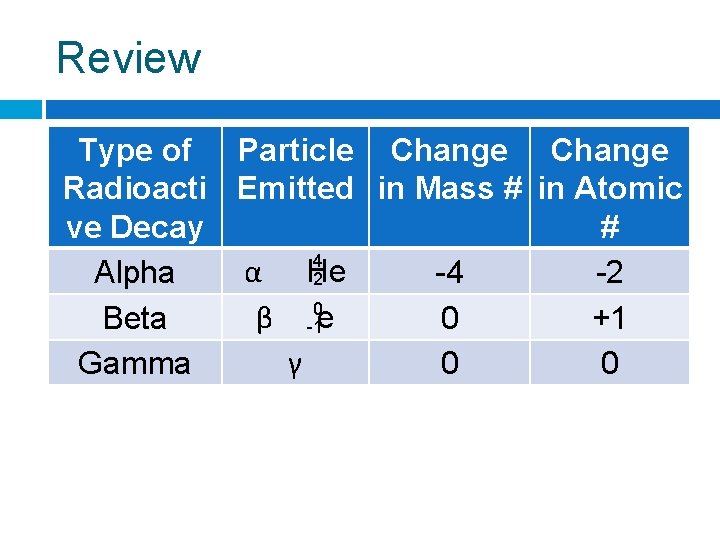
Review Type of Particle Change Radioacti Emitted in Mass # in Atomic ve Decay # 4 α He Alpha -4 -2 2 0 β Beta 0 +1 -1 e γ Gamma 0 0

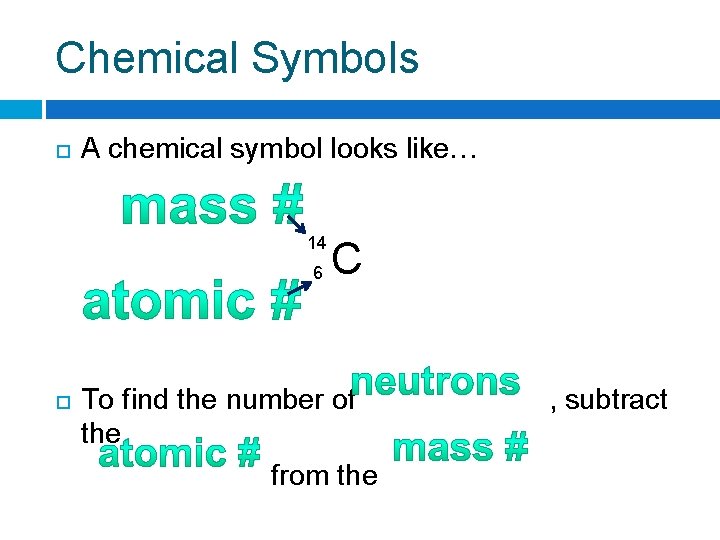
Chemical Symbols A chemical symbol looks like… 14 6 C To find the number of the from the , subtract

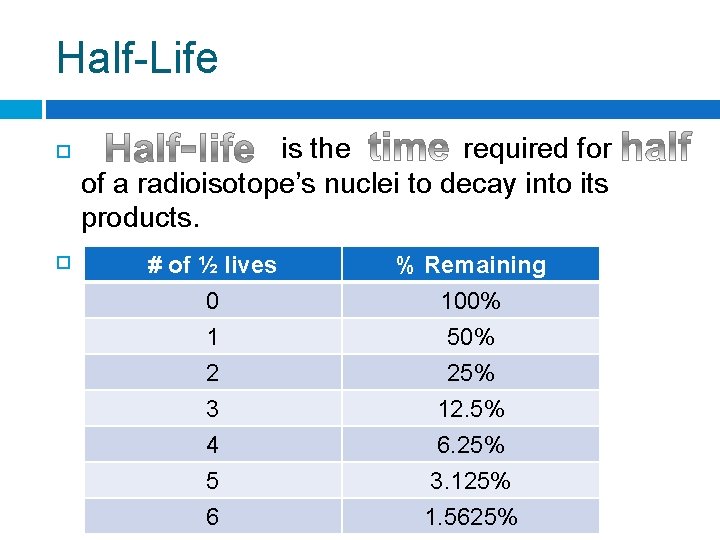
Half-Life is the required for of a radioisotope’s nuclei to decay into its products. For any # ofradioisotope, ½ lives % Remaining 0 1 2 100% 50% 25% 3 4 5 6 12. 5% 6. 25% 3. 125% 1. 5625%

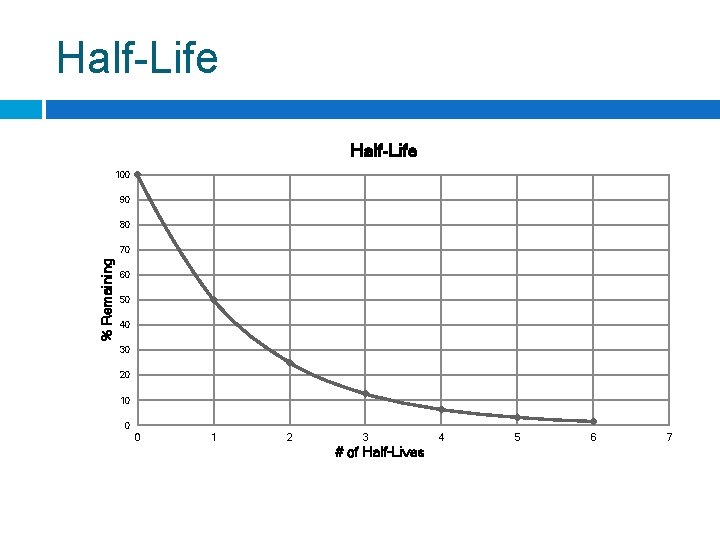
Half-Life 100 90 80 % Remaining 70 60 50 40 30 20 10 0 0 1 2 3 # of Half-Lives 4 5 6 7

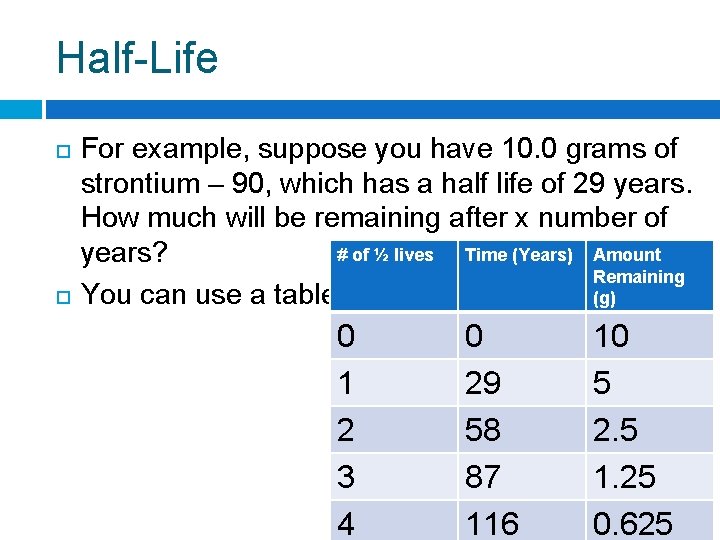
Half-Life For example, suppose you have 10. 0 grams of strontium – 90, which has a half life of 29 years. How much will be remaining after x number of # of ½ lives Time (Years) Amount years? Remaining You can use a table: (g) 0 1 2 3 4 0 29 58 87 116 10 5 2. 5 1. 25 0. 625
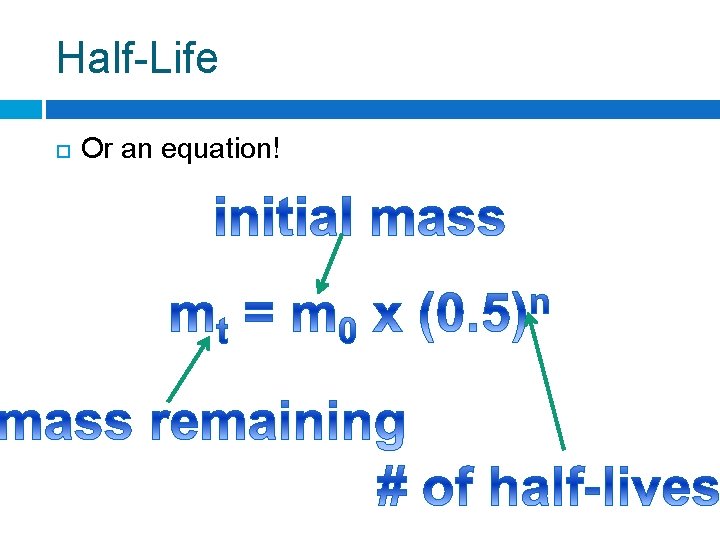
Half-Life Or an equation!

Half-Life Example 1: If gallium – 68 has a half-life of 68. 3 minutes, how much of a 160. 0 mg sample is left after 1 half life? ____ 2 half lives? _____ 3 half lives? _____

Half-Life Example 2: Iron-59 is used in medicine to diagnose blood circulation disorders. The halflife of iron-59 is 44. 5 days. How much of a 2. 000 mg sample will remain after 133. 5 days? _______

Nuclear Fission of a nucleus - Very heavy nucleus is split into approximately fragments reaction releases several neutrons which more nuclei - If controlled, energy is released (like in ) Reaction control depends on reducing the of the neutrons (increases the reaction rate) and extra neutrons ( creases the reaction rate).

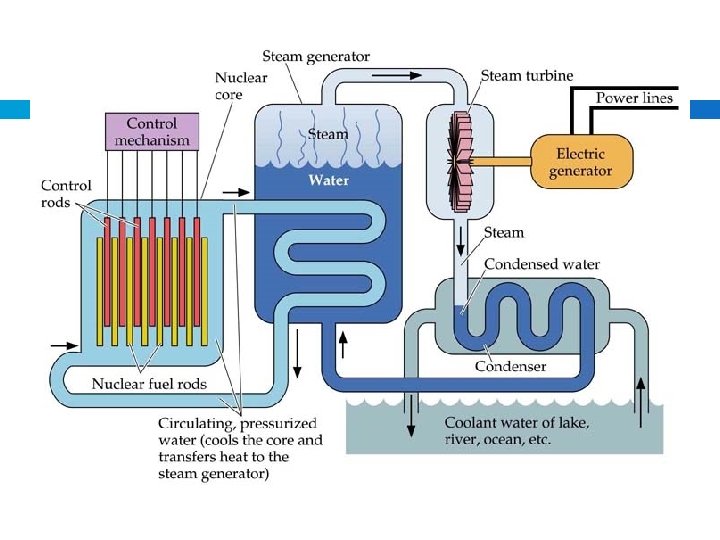
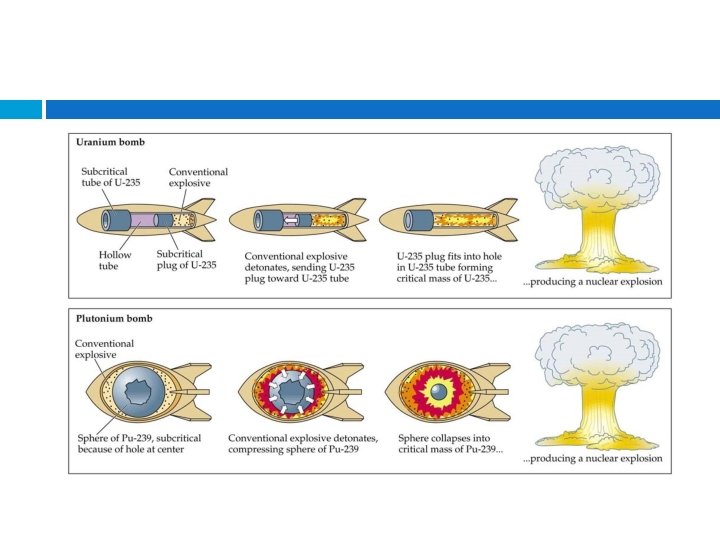
Nuclear Fission - 1 st controlled nuclear reaction in December 1942. 1 st uncontrolled nuclear explosion occurred July 1945. - Examples – atomic bomb, current nuclear power plants


© 2003 John Wiley and Sons Publishers Courtesy David Bartruff/Corbis Images Cooling towers of a nuclear power plant.

Construction of a tunnel that will be used for burial of radioactive wastes deep within Yucca Mountain, Nevada.

Disposal of radioactive wastes by burial in a shallow pit.


Nuclear Fusion - Two light - combining of a nuclei combine to form a single heavier nucleus - Does not occur under standard conditions ( repels ) inexpensive - Advantages compared to fission noradioactivewaste , + + large start control - Disadvantages - requires amount of energy to , difficult to - Examples – energy output of stars, hydrogen

Applications Medicine Chemotherapy Power pacemakers Diagnostic tracers Agriculture Irradiate food Pesticide Energy Fission Fusion

X-ray examination of luggage at a security station.

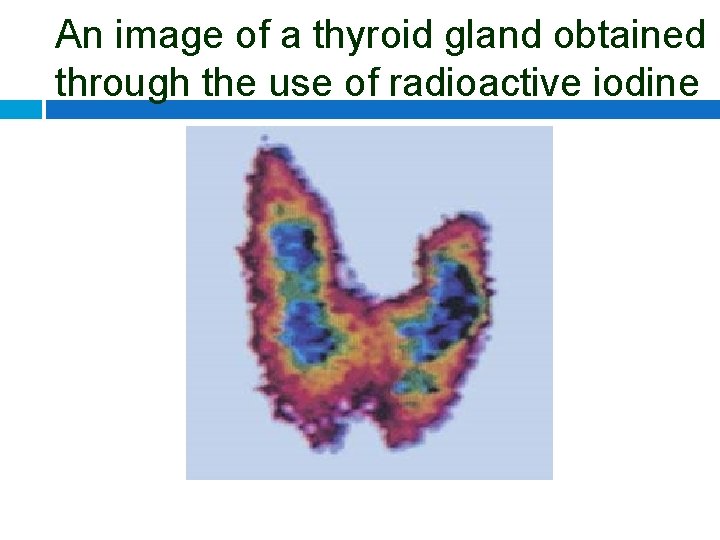
An image of a thyroid gland obtained through the use of radioactive iodine

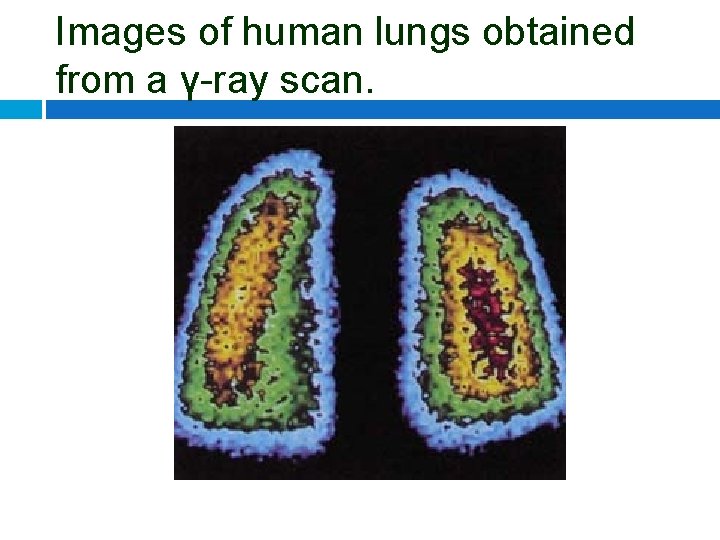
Images of human lungs obtained from a γ-ray scan.

A cancer patient receiving radiation therapy.