Bioinorganic chemistry Introduction Bioinorganic chemistry as a highly

Bioinorganic chemistry Introduction
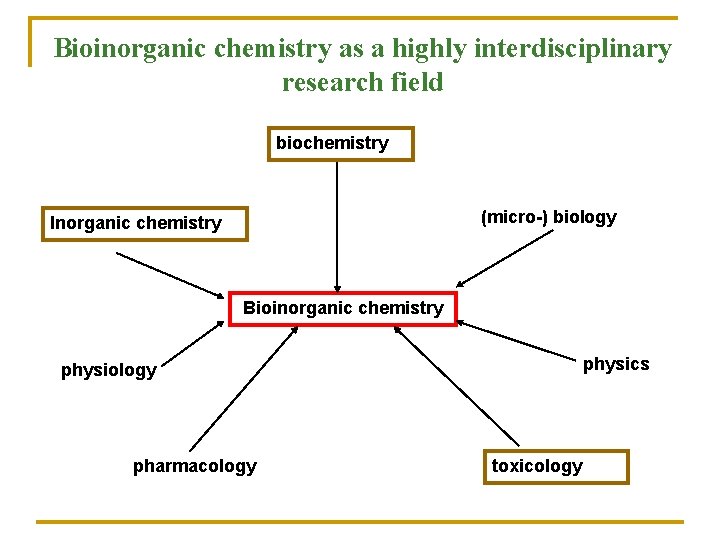
Bioinorganic chemistry as a highly interdisciplinary research field biochemistry (micro-) biology Inorganic chemistry Bioinorganic chemistry physics physiology pharmacology toxicology

Evolution of life essential elements Earth solidified ~ 4 billion years ago 81 stabile elements Elements of the living organism: 1. 2. 3. Elements in large scale: 11 elements H, C, N, O, Na, Mg, P, S, Cl, K, Ca Elements in small scale: 7 elements Mn, Fe, Co, Cu, Zn, I, Mo Elements of a few species: 7 elements B, F, Si, V, Cr, Se, Sn
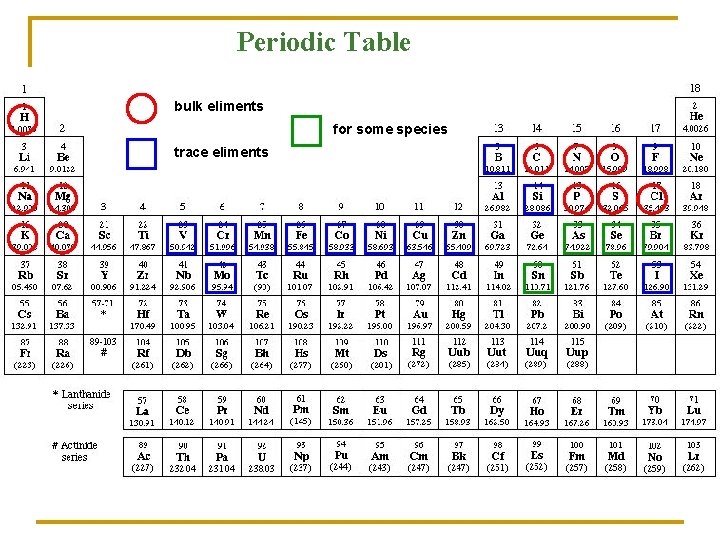
Periodic Table bulk eliments for some species trace eliments
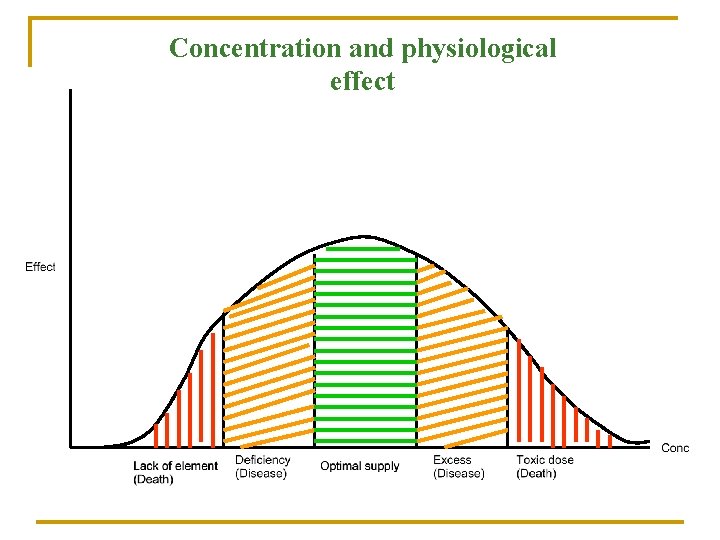
Concentration and physiological effect

Metals

Metals essential for life: n The role for most is uncertain n Na, K, Mg, Ca n V, Cr, Mn, Fe Co, Ni, Cu, Zn n Mo, W

General roles of metal ions in biology Na, K: Charge carriers Osmotic and electrochemical gradients Nerve function Mg, Ca: Enzyme activators Structure promoters Lewis acids Mg 2+: chlorophyll, photosynthesis Ca 2+: insoluble phosphates
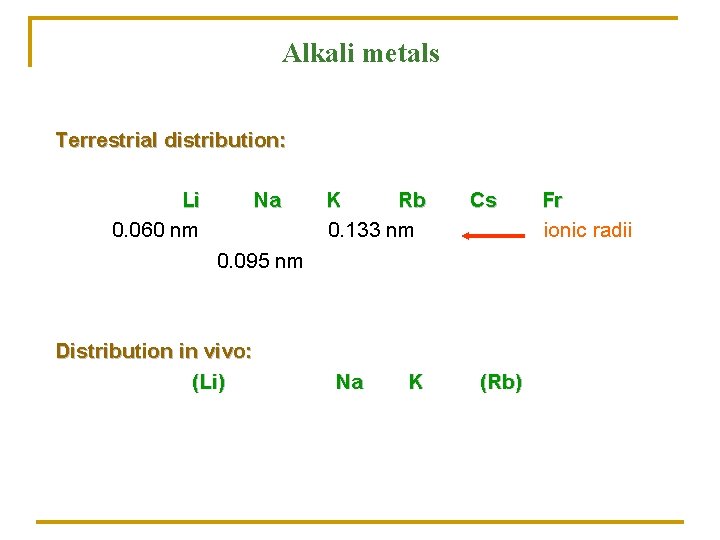
Alkali metals Terrestrial distribution: Li 0. 060 nm Na K Rb 0. 133 nm Cs 0. 095 nm Distribution in vivo: (Li) Na K (Rb) Fr ionic radii
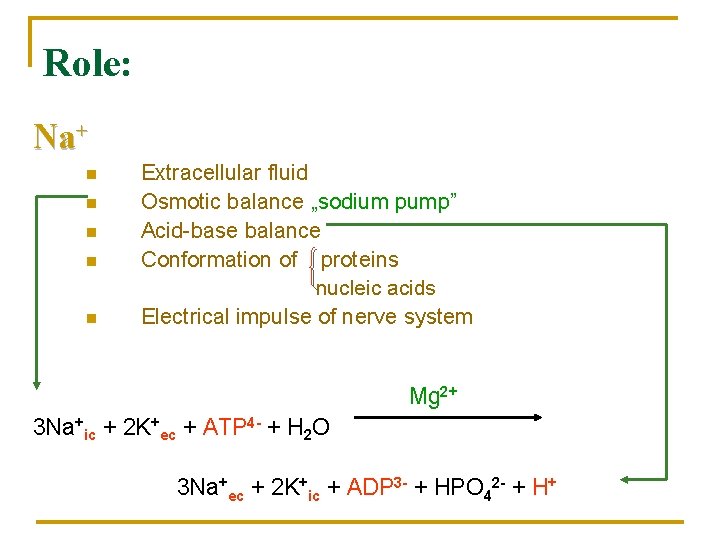
Role: Na+ n n n Extracellular fluid Osmotic balance „sodium pump” Acid-base balance Conformation of proteins nucleic acids Electrical impulse of nerve system Mg 2+ 3 Na+ic + 2 K+ec + ATP 4 - + H 2 O 3 Na+ec + 2 K+ic + ADP 3 - + HPO 42 - + H+
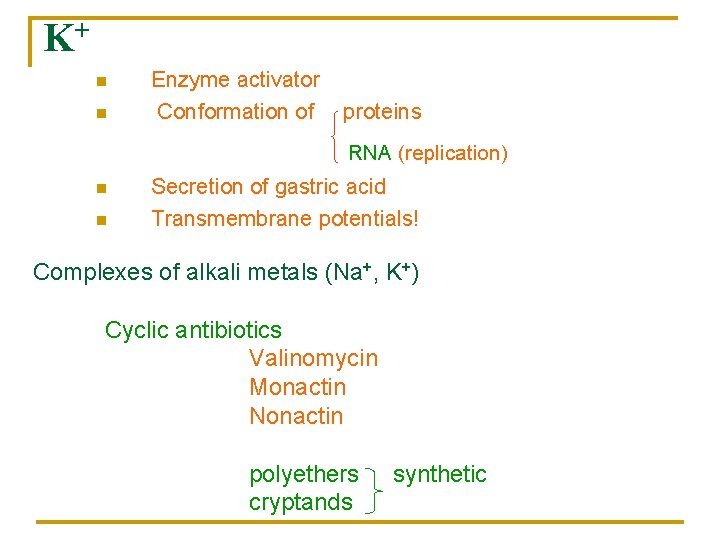
K+ n n Enzyme activator Conformation of proteins RNA (replication) n n Secretion of gastric acid Transmembrane potentials! Complexes of alkali metals (Na+, K+) Cyclic antibiotics Valinomycin Monactin Nonactin polyethers cryptands synthetic
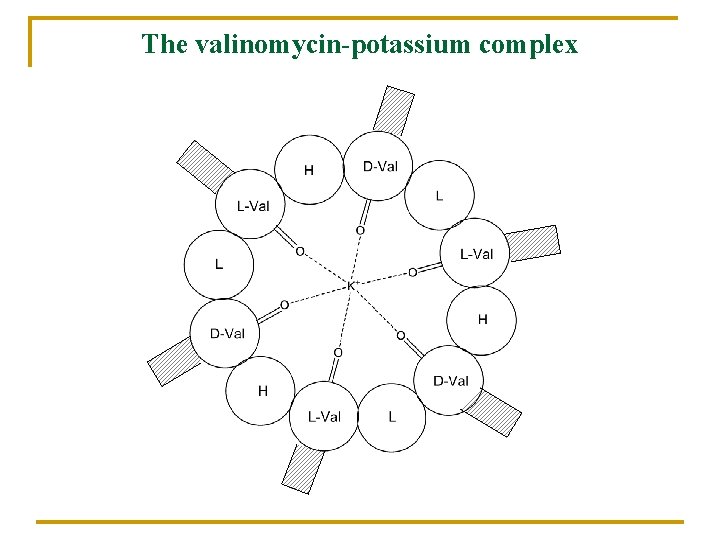
The valinomycin-potassium complex
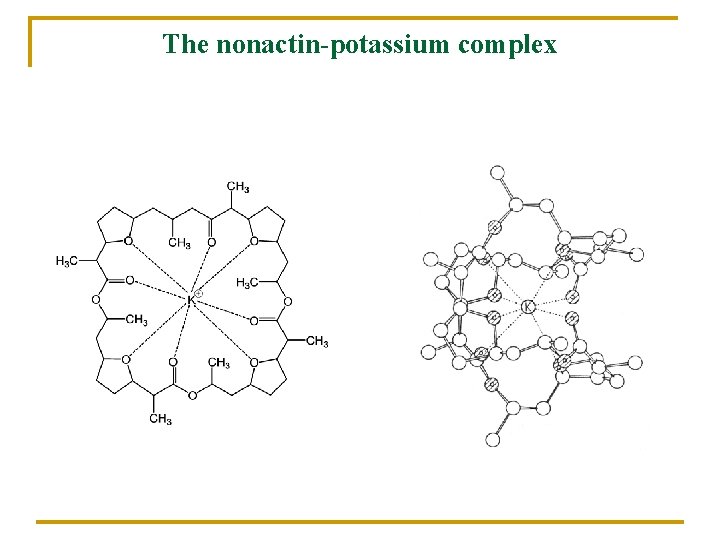
The nonactin-potassium complex
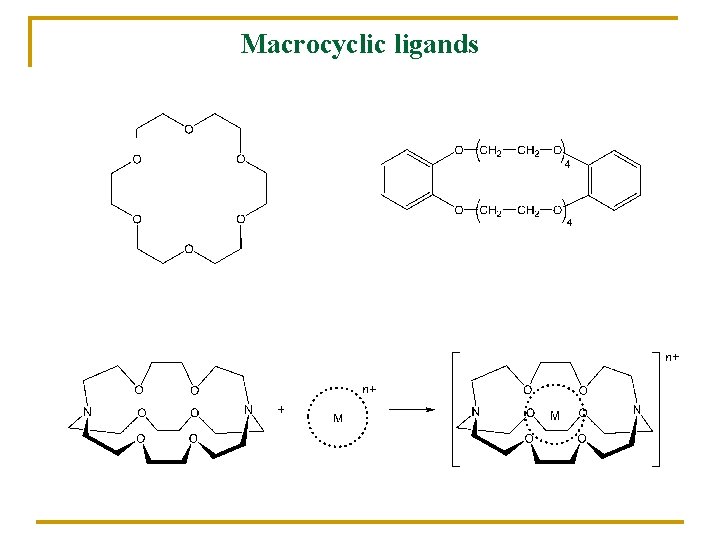
Macrocyclic ligands

Alkaline Earth Metals Terrestrial distribution: Be Mg Distribution in vivo: Be, Ba TOXIC! Sr (not particularly toxic) 90 Sr accumulates in bones Ca Sr Mg Ca Ba Ra
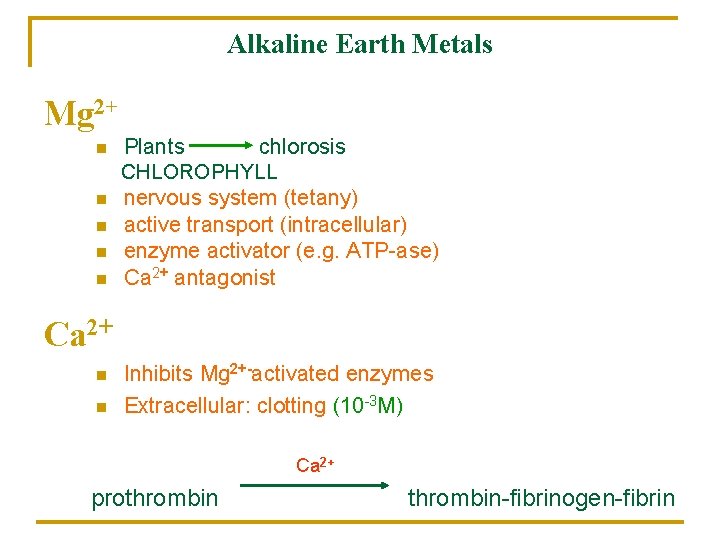
Alkaline Earth Metals Mg 2+ n n n Plants chlorosis CHLOROPHYLL nervous system (tetany) active transport (intracellular) enzyme activator (e. g. ATP-ase) Ca 2+ antagonist Ca 2+ n n Inhibits Mg 2+-activated enzymes Extracellular: clotting (10 -3 M) Ca 2+ prothrombin-fibrinogen-fibrin
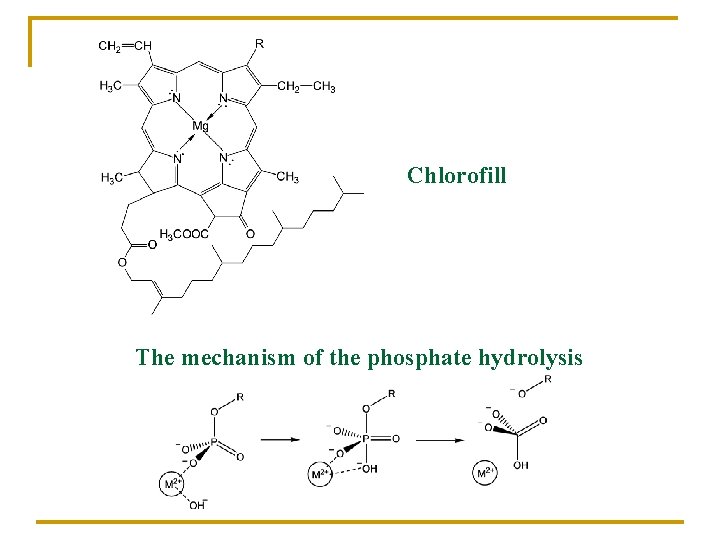
Chlorofill The mechanism of the phosphate hydrolysis

Transition Metals
Fe, Cu, Mo: Electron-transfer Redox proteins and enzymes Oxygen carrying proteins Nitrogen fixation Zn: Metalloenzymes Structure promoters Lewis acid Not a redox catalyst! Other metal ions: less well defined and more obscure roles

Fe(II), Fe(III): n Essential for ALL organisms n In plants: iron deficiency n In human body: 4 -5 g n Uptake: ~ 1 mg/day
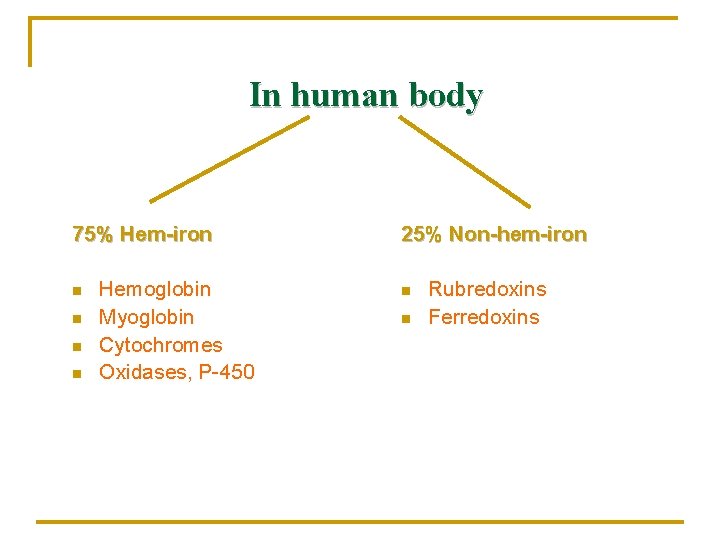
In human body 75% Hem-iron n n Hemoglobin Myoglobin Cytochromes Oxidases, P-450 25% Non-hem-iron n n Rubredoxins Ferredoxins
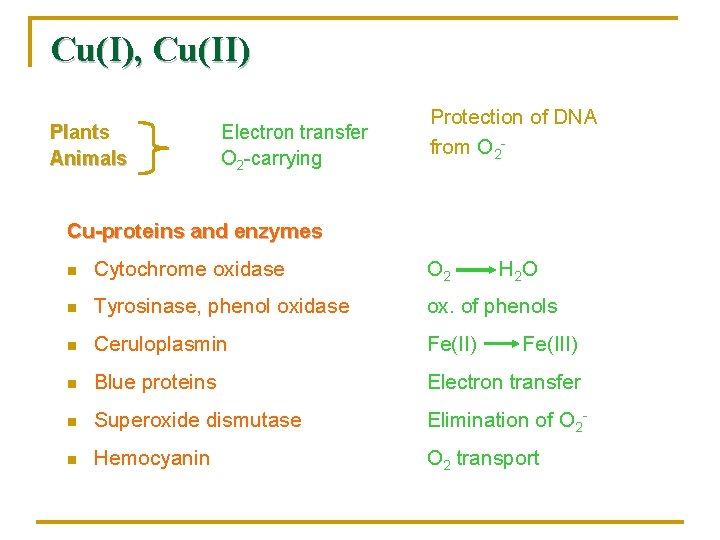
Cu(I), Cu(II) Plants Animals Electron transfer O 2 -carrying Protection of DNA from O 2 - Cu-proteins and enzymes n Cytochrome oxidase O 2 H 2 O n Tyrosinase, phenol oxidase ox. of phenols n Ceruloplasmin Fe(II) n Blue proteins Electron transfer n Superoxide dismutase Elimination of O 2 - n Hemocyanin O 2 transport Fe(III)
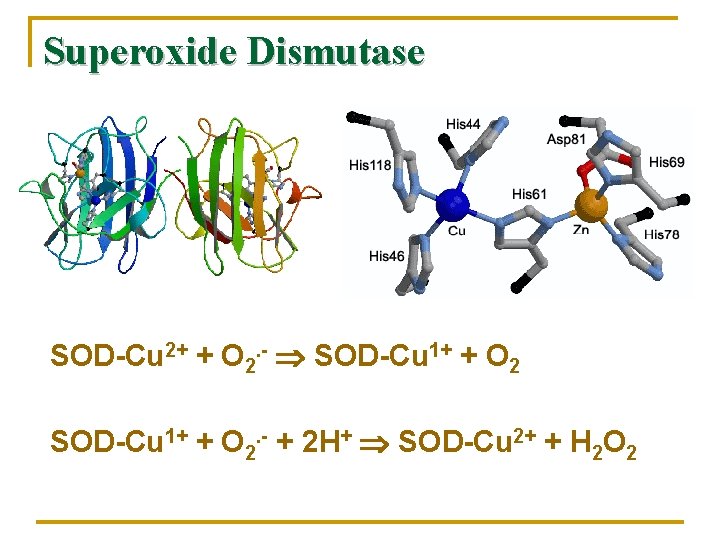
Superoxide Dismutase SOD-Cu 2+ + O 2. - SOD-Cu 1+ + O 2. - + 2 H+ SOD-Cu 2+ + H 2 O 2

Role of Zn 2+ : deficiency: n disturbances of repr. system n dwarfism n skin lesions n skeletal abnormalities
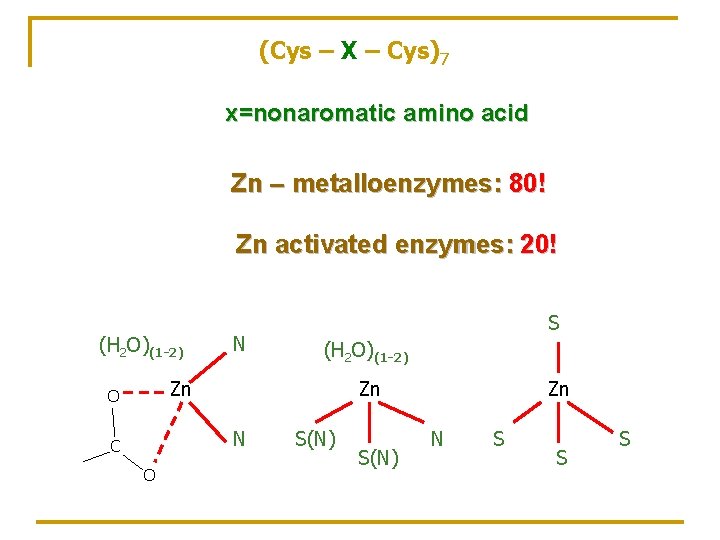
(Cys – X – Cys)7 x=nonaromatic amino acid Zn – metalloenzymes: 80! Zn activated enzymes: 20! (H 2 O)(1 -2) N S (H 2 O)(1 -2) Zn O Zn N C O S(N) Zn N S S S

Function of Zn in metalloenzymes 1. Structure-promoter 2. Substrate binder 3. Lewis acid
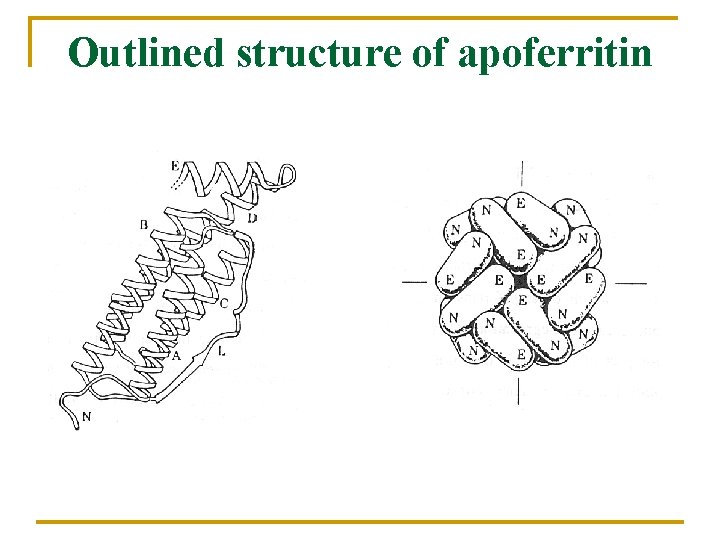
Outlined structure of apoferritin
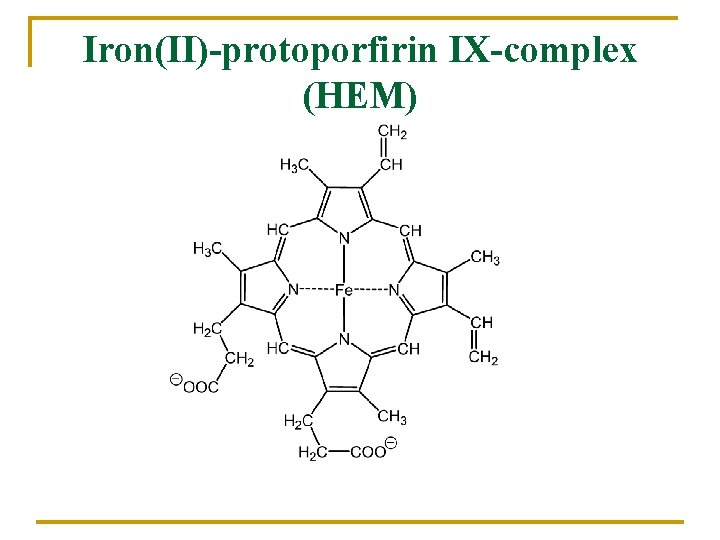
Iron(II)-protoporfirin IX-complex (HEM)
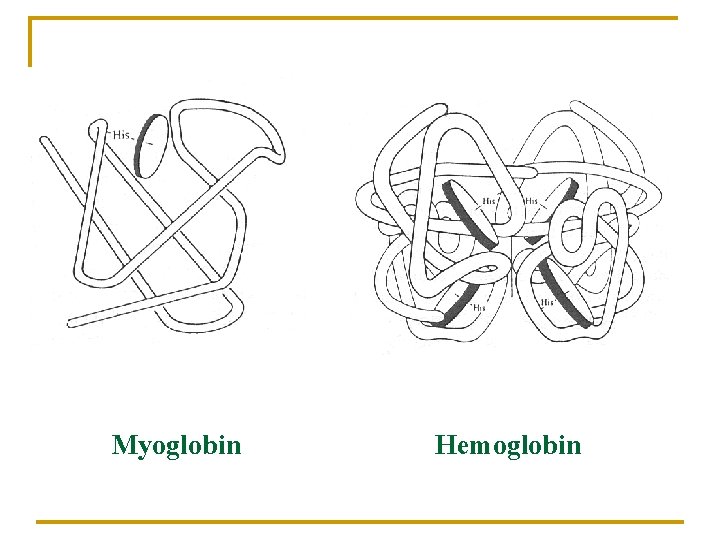
Myoglobin Hemoglobin
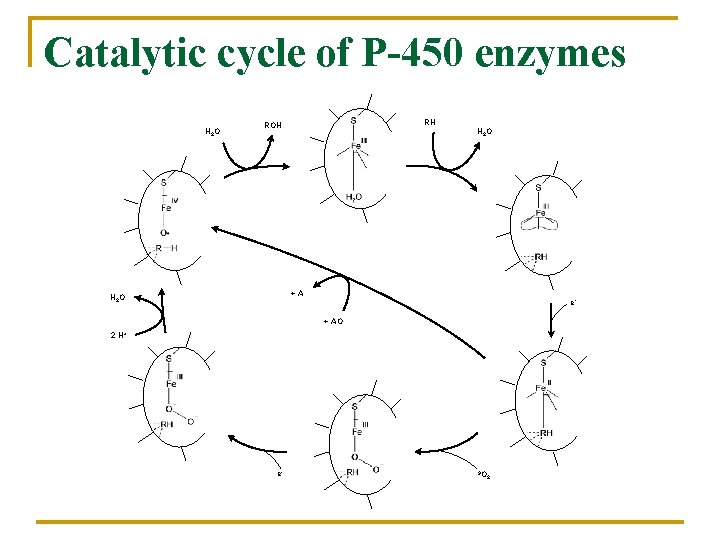
Catalytic cycle of P-450 enzymes H 2 O RH ROH H 2 O +A H 2 O e+ AO 2 H+ e- 3 O 2
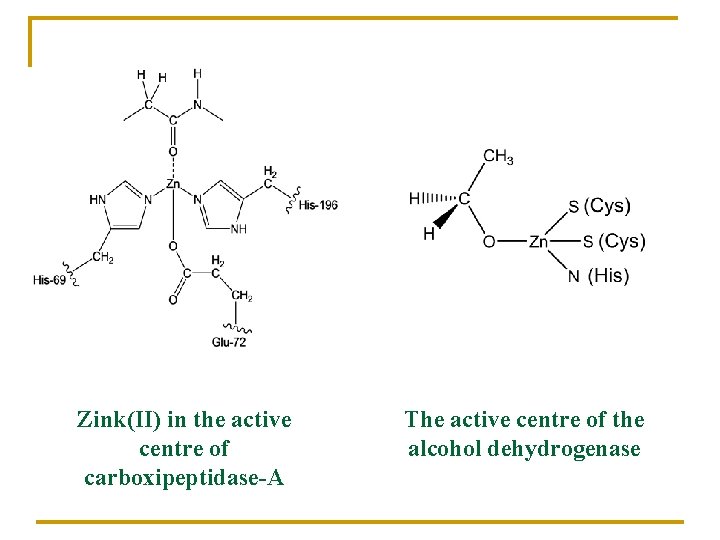
Zink(II) in the active centre of carboxipeptidase-A The active centre of the alcohol dehydrogenase
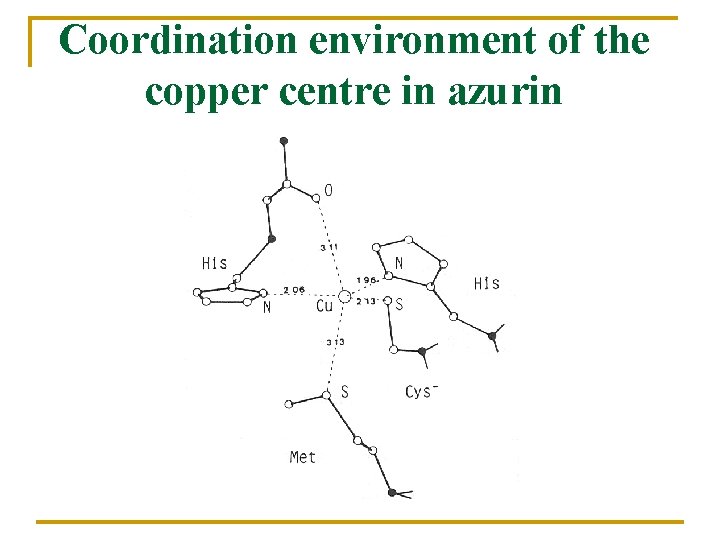
Coordination environment of the copper centre in azurin
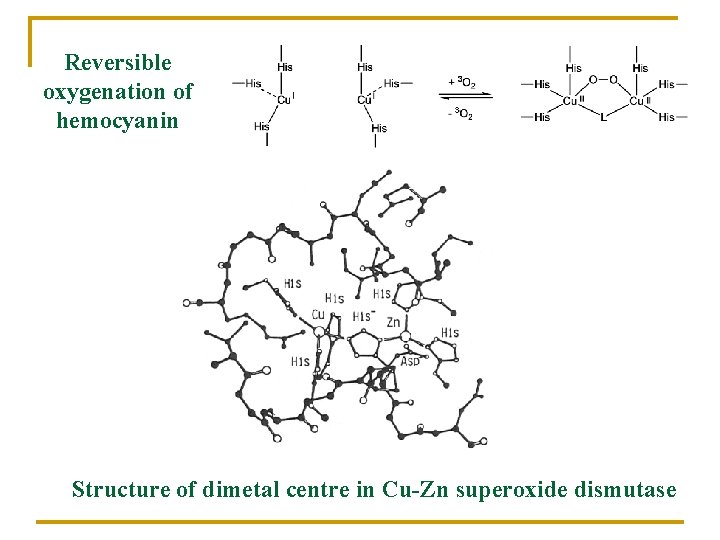
Reversible oxygenation of hemocyanin Structure of dimetal centre in Cu-Zn superoxide dismutase
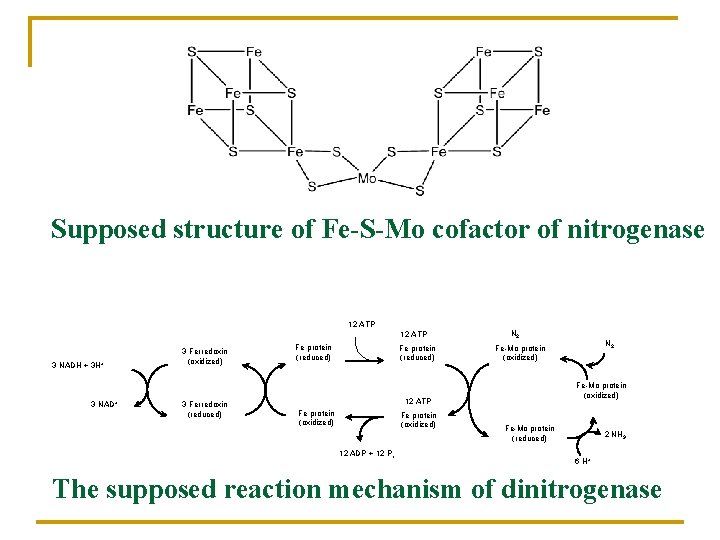
Supposed structure of Fe-S-Mo cofactor of nitrogenase 12 ATP 3 NADH + 3 H+ 3 NAD+ 3 Ferredoxin (oxidized) 3 Ferredoxin (reduced) Fe protein (reduced) N 2 Fe-Mo protein (oxidized) 12 ATP Fe protein (oxidized) 12 ADP + 12 Pi N 2 Fe-Mo protein (oxidized) Fe-Mo protein (reduced) 2 NH 3 6 H+ The supposed reaction mechanism of dinitrogenase
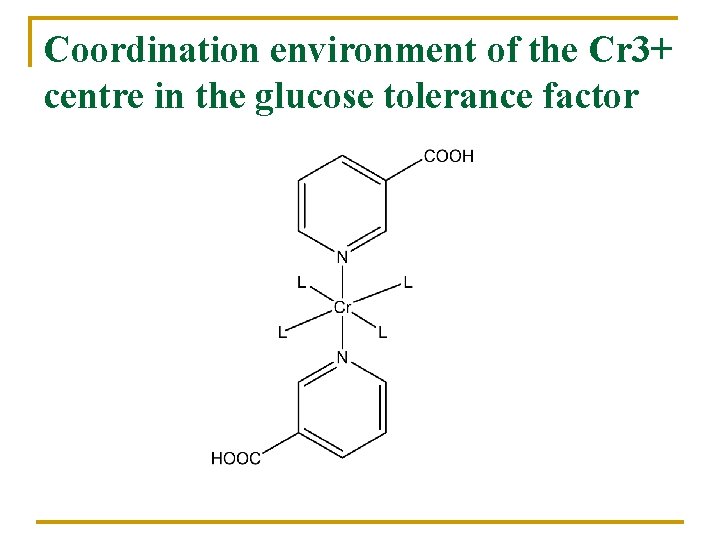
Coordination environment of the Cr 3+ centre in the glucose tolerance factor
- Slides: 35