INORGANIC CHEMISTRY BASIC CONCEPTS OF CHEMISTRY Chemistry Chemistry

INORGANIC CHEMISTRY

BASIC CONCEPTS OF CHEMISTRY Chemistry: Chemistry is the branch of science that deals with the composition, structure and properties of matter. Chemistry is called the science of atoms and molecule

Branches of Chemistry Organic Chemistry -This branch deals with study of carbon compounds especially hydrocarbons and their derivatives. Inorganic Chemistry-This branch deals with the study of compounds of all other elements except carbon. It largely concerns itself with the study of minerals found in the Earth's crust. Physical Chemistry-The explanation of fundamental principles governing various chemical phenomena is the main concern of this branch. It is basically concerned with laws and theories of the different branches of chemistry.

Industrial Chemistry-The chemistry involved in industrial processes is studied under this branch. Analytical Chemistry-This branch deals with the qualitative and quantitative analysis of various substances. Biochemistry-This branch deals with the chemical changes going on in the bodies of living organisms; plants and animals. Nuclear Chemistry-Nuclear reactions, such as nuclear fission, nuclear fusion, transmutation processes etc. are studied under this branch.

SCIENTIFC METHOD The scientific method is not a rigid set of rules, but a general guide for investigation or logical approach to finding answer to question. 1. Curiosity or a quest for knowledge 2. Design and do experiment, collect data and information, establish facts, and make observation 3. Organize and find relationship between information or data. Formulate a law, a statement of the relationships between facts data, and observation

Matter and Its Properties all matter takes up space (has volume) and has mass: a measure of the amount of matter. Objects that have a large amount of matter will have a large mass, and objects with a small amount of matter will have a small mass.

Basic Building Blocks of Matter all matter is made up of atoms and/or molecules. atom: the smallest unit of an element that maintains the properties of that element: a pure substance made of only one kind of atom. molecule: a substance that is comprised of two or more elements that are chemically bonded. compound: a substance that is comprised of two or more different elements that are chemically bonded.

Classification of Matter all matter can be classified as either a pure substance or a mixture. • pure substances -> elements & compounds • mixtures -> a blend of two or more kinds of matter, each of which retains its own identity and properties. 1. Homogeneous mixtures: uniform in composition. ex: salt water solution 2. Heterogeneous mixtures: not uniform in composition. ex: muddy water

Mixtures can be separated physically. 1. Filtration 2. Decanting (“pouring off”) 3. Paper chromatography 4. Centrifugation Pure substances differ from mixtures in the following ways: 1. Every sample of a pure substance has exactly the same characteristic properties. 2. Every sample of a given pure substance has exactly the same composition. 3. Separating Mixture Separating Ink
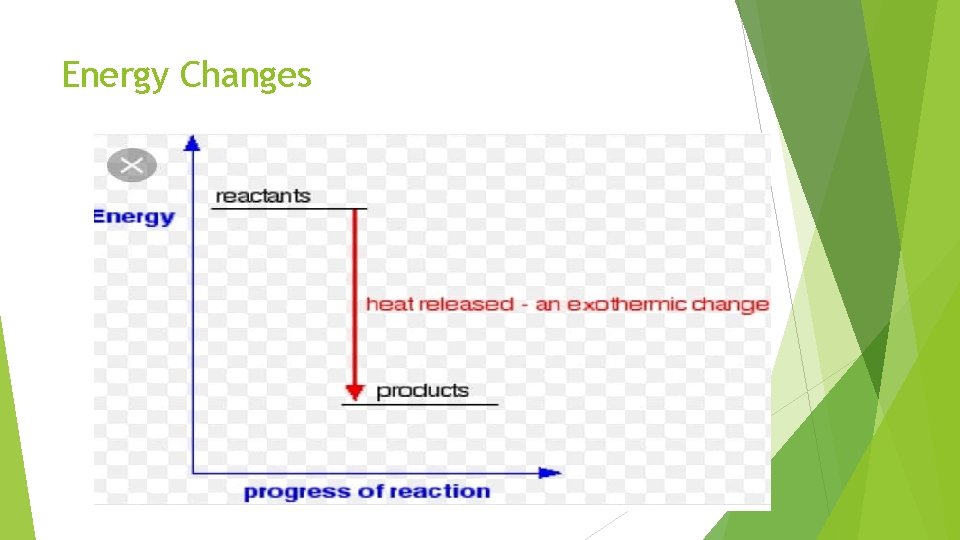
Energy Changes
- Slides: 10