Diabetes Mellitus Zoya Minasyan RNMSNEdu Diabetes Mellitus A

Diabetes Mellitus Zoya Minasyan RN-MSN-Edu

Diabetes Mellitus �A chronic multisystem disease related to ◦ Abnormal insulin production ◦ Impaired insulin utilization ◦ Or both � 7 th leading cause of death in the USA � Leading cause of adult blindness, end-stage renal disease, and non traumatic lower limb amputation. � Major contributing factor ◦ Heart disease ◦ Stroke

Diabetes Mellitus Etiology and Pathophysiology ◦ Genetic ◦ Autoimmune ◦ Viral ◦ Environmental Regardless of it’s cause, Diabetes is primarily a disorder of glucose metabolism r/t absent or insufficient insulin supply and/or poor utilization of the insulin that is available.

Etiology and Pathophysiology � Two most common types ◦ Type 1 ◦ Type 2 � Other types ◦ Gestational ◦ Prediabetes ◦ Secondary diabetes

Etiology and Pathophysiology � Normal insulin metabolism ◦ Produced by the cells �Islets of Langerhans of the pancreas. ◦ Released continuously into bloodstream in small amounts; with larger amounts released after food. ◦ Stabilizes glucose range to 70 to 120 mg/d. L � Counter regulatory hormones-glucagon, epinephrine, growth hormone and cortisol work to oppose the effect of insulin; work to increase blood glucose level by stimulating glucose production and output by liver, and decreasing the movement of glucose into the cells.

Insulin ◦ Promotes glucose transport from bloodstream across cell membrane to cytoplasm of cell �Decreases glucose in the bloodstream ◦ ↑ insulin after a meal �Stimulates storage of glucose as glycogen in liver and muscle �Inhibits gluconeogenesis �Enhances fat deposition �↑ protein synthesis
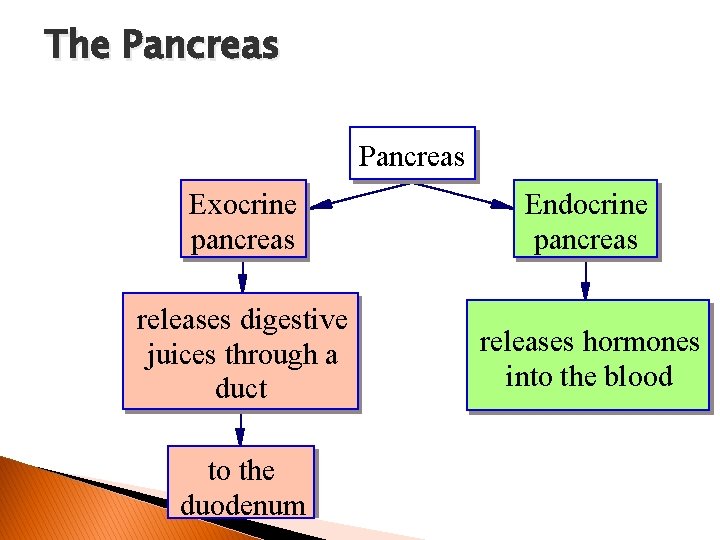
The Pancreas Exocrine pancreas Endocrine pancreas releases digestive juices through a duct releases hormones into the blood to the duodenum
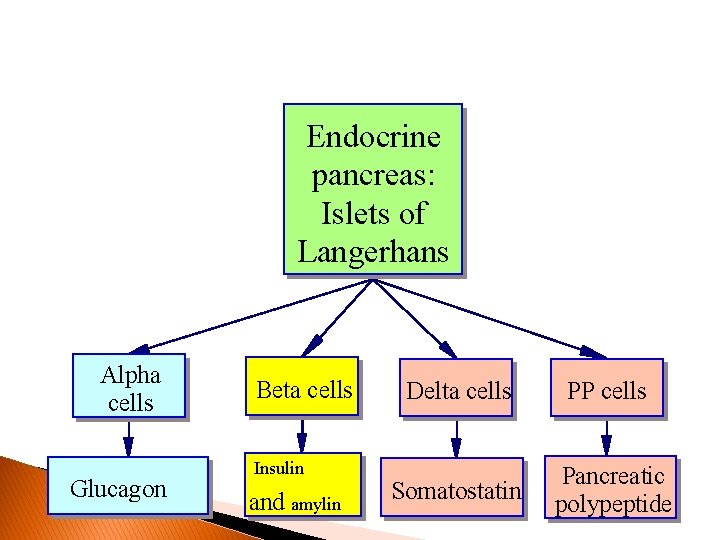
Endocrine pancreas: Islets of Langerhans Alpha cells Glucagon Beta cells Insulin and amylin Delta cells PP cells Somatostatin Pancreatic polypeptide

Functions of Pancreatic Hormones � Glucagon: causes cells to release stored food into the blood � Insulin: allows cells to take up glucose from the blood � Amylin: slows glucose absorption in small intestine; suppresses glucagon secretion � Somatostatin: decreases GI activity; suppresses glucagon and insulin secretion


Question Which pancreatic hormone decreases blood glucose levels? a. Glucagon b. Insulin c. Amylin d. Somatostatin

Answer Insulin allows cells to take glucose from the blood and use it for energy/to make ATP. Because it stimulates movement of glucose out of the blood and into the cells, blood levels decrease when insulin is released. b.

Question Tell whether the following statement is true or false: Type 2 DM is more common than Type 1 DM.

Answer True Type 1 DM is autoimmune (juvenile diabetes is Type 1), and affects only 5% to 10% of the diabetic population. Type 2 DM is associated with risk factors like obesity, poor diet, and sedentary lifestyle; 90% to 95% of diabetics suffer from this type.

Insulin � Skeletal muscle and adipose tissues are Insulin-dependent tissues; have receptors for insulin � Other tissues (brain, liver, blood cells) do not directly depend on insulin for glucose transport; but require adequate glucose for normal fx. Liver has receptor sites for hepatic uptake of glucose and its conversion to glycogen.

Type 1 Diabetes Mellitus � “juvenile-onset” or “insulin-dependent” diabetes, occurs in people younger than 40 years of age, and in younger children. • Progressive destruction of pancreatic cells by body’s own T cells � Caused by ◦ Genetic predisposition �Related to human leukocyte antigens (HLAs) ◦ Exposure to a virus � Manifestations develop when pancreas can no longer produce insulin.

Type 1 Diabetes Mellitus Onset of Disease � Will require insulin � Diabetic ketoacidosis (DKA) ◦ Occurs in absence of insulin ◦ Life-threatening condition ◦ Results in metabolic acidosis

Prediabetes � Individuals already at risk for diabetes � Blood glucose is high but not high enough to be diagnosed as having diabetes � Characterized by ◦ Impaired fasting glucose (IFG) ◦ Impaired glucose tolerance (IGT) � IFG: Fasting glucose levels are 100 to 125 mg/d. L � IGT: 2 -Hour plasma glucose levels are between 140 and 199 mg/d. L � AIC is in range of 5. 7% to 6. 4%. � Must watch for diabetes symptoms ◦ Polyuria ◦ Polyphagia ◦ Polydipsia

Type 2 Diabetes Mellitus Etiology and Pathophysiology � Usually ◦ ◦ occurs in people over 35 years of age, overweight, increases with age, genetic basis.

Type 2 Diabetes Mellitus Etiology and Pathophysiology � Four major metabolic abnormalities ◦ 1. Insulin resistance �Body tissues do not respond to insulin which results in hyperglycemia. ◦ 2. Pancreas ↓ ability to produce insulin �β cells fatigued from compensating �β-cell mass lost ◦ 3. Inappropriate glucose production from liver ◦ 4. Alteration in production of hormones

Type 2 Diabetes Mellitus Etiology and Pathophysiology Individuals with metabolic syndrome also known as syndrome X or insulin resistance syndrome are at increased risk for type 2 diabetes ◦ risk for cardiovascular disease and diabetes characterized by insulin resistance Individuals with metabolic syndrome ◦ Elevated insulin levels, ↑ triglycerides, LDLs, ↓ HDLs, hypertension ◦ Risk factors �Central obesity, sedentary lifestyle, urbanization, certain ethnicities(African Americans, Native Americans, Hispanics, Asians)

Type 2 Diabetes Mellitus Onset of Disease � Gradual onset � Person may go many years with undetected hyperglycemia. � Osmotic fluid/electrolyte loss from hyperglycemia may become severe. ◦ Hyperosmolar coma(will discuss later)

Gestational Diabetes � Develops during pregnancy � Detected at 24 to 28 weeks of gestation � Usually normal glucose levels at 6 weeks post partum � Increased risk for cesarean delivery, and neonatal complications � Increased risk for developing type 2 in 5 to 10 years � Therapy: First- nutritional, second -insulin

Specific Types of Diabetes � Results from ◦ Another medical condition � Cushing syndrome � Hyperthyroidism � Pancreatitis � Parenteral nutrition � Cystic fibrosis ◦ Treatment of a medical condition that causes abnormal blood glucose level � Corticosteroids (Prednisone) � Thiazides � Phenytoin (Dilantin) � Atypical antipsychotics (clozapine) Usually resolves when underlying condition treated � Drugs that can alter blood glucose levels are listed in Table 49 -8. �

Clinical Manifestations Type 1 Diabetes Mellitus � Classic symptoms ◦ Polyuria (frequent urination) ◦ Polydipsia (excessive thirst) ◦ Polyphagia (excessive hunger) � Weight loss � Weakness � Fatigue

Clinical Manifestations Type 2 Diabetes Mellitus � Nonspecific symptoms ◦ May have classic symptoms of type 1 � Fatigue � Recurrent infection � Recurrent vaginal yeast infection � Prolonged wound healing � Visual changes

Diabetes Mellitus Diagnostic Studies � Four methods of diagnosis � Hemoglobin A 1 C test 1. 2. 3. 4. AIC ≥ 6. 5% Fasting plasma glucose level >126 mg/d. L Random plasma glucose measurement ≥ 200 mg/d. L Two-hour OGTT(oral glucose tolerance test) level ≥ 200 mg/d. L when a glucose load of 75 g is used ◦ In 2010, recommended to be used as a diagnostic test ◦ Useful in determining glycemic levels over time ◦ Shows the amount of glucose attached to hemoglobin molecules over RBC life span, approximately 120 days ◦ Ideal goal � ADA ≤ 7. 0% � American College of Endocrinology <6. 5% ◦ Normal A 1 C reduces risks of retinopathy, nephropathy, and neuropathy.

Diabetes Mellitus � Goals of diabetes management Decrease symptoms Promote well being Prevent acute complications Delay onset and progression of long term complications ◦ Patient teaching ◦ ◦ �Self-monitoring of blood glucose ◦ Nutritional therapy ◦ Drug therapy ◦ Exercise

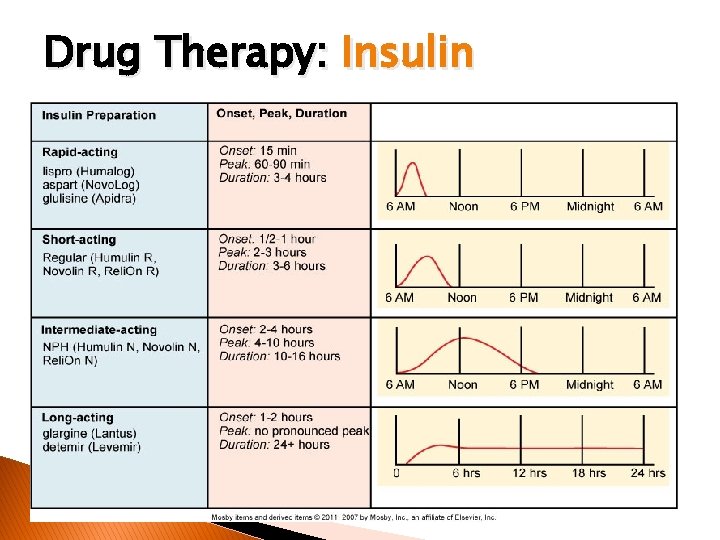
Drug Therapy: Insulin ◦ Insulin differ with regard to onset, peak action, and duration. �Characterized as rapid-acting, short-acting, intermediate-acting, and long-acting ◦ Rapid-acting: Lispro (Humalog), Aspart (Novolog), and glulisine (Apidra) ◦ Short-acting: Regular ◦ Intermediate-acting: NPH ◦ Long-acting: Glargine (Lantus), detemir (Levemir)
Drug Therapy: Insulin

Drug Therapy: Insulin � Insulin preparations ◦ Rapid-acting (bolus) �Lispro, aspart, glulisine �Injected 0 to 15 minutes before meal �Onset of action 15 minutes ◦ Short-acting (bolus) �Regular �Injected 30 to 45 minutes before meal �Onset of action 30 to 60 minutes ◦ Long-acting (basal) �Injected once a day at bedtime or in the morning �Released steadily and continuously �No peak action �Cannot be mixed with any other insulin or solution

Drug Therapy: Insulin � Storage of insulin ◦ Do not heat/freeze. ◦ In-use vials may be left at room temperature up to 4 weeks. ◦ Extra insulin should be refrigerated. ◦ Avoid exposure to direct sunlight. ◦ Administration of insulin �Cannot be taken orally �Subcutaneous injection for self-administration �IV administration �Fastest absorption from abdomen, followed by arm, thigh, and buttock �Abdomen is the preferred site. �Rotate injections within one particular site. �Do not inject in site to be exercised.


Drug Therapy: Insulin � Administration of insulin ◦ Hand washing ◦ Do not recap needle ◦ 45 - to 90 -degree angle, depending on fat thickness of the patient ◦ Insulin pens preloaded with insulin are now available.
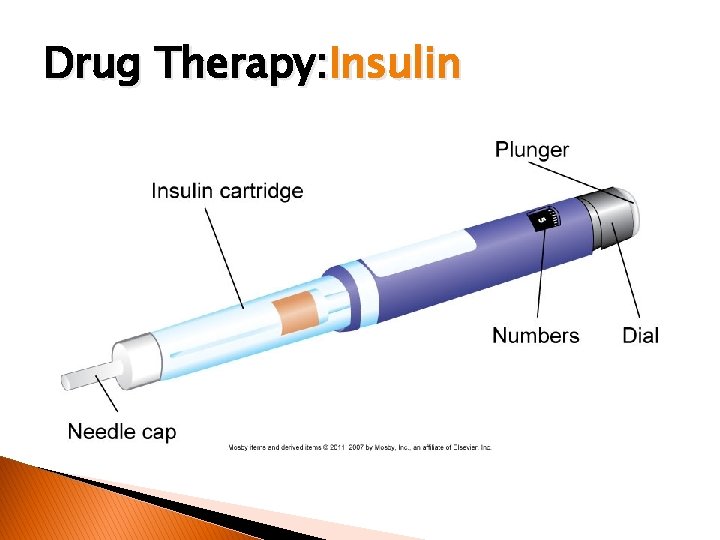
Drug Therapy: Insulin

Drug Therapy: Insulin �Insulin pump ◦ Continuous subcutaneous infusion; Battery-operated device ◦ Connected via plastic tubing to a catheter inserted into subcutaneous tissue in abdominal wall

Drug Therapy: Insulin � Problems ◦ ◦ ◦ with insulin therapy Hypoglycemia Allergic reaction Lipodystrophy(atrophy of SQ tissue) Somogyi effect( during the sleep hrs-decline in blood glucose in respond to insulin; counterregulatory hormones causes gluconeogenesis which causes rebound hyperglycemia and ketosis at night and in the morning; sweats or nightmares, and high blood glucose in am(recheck BG at 2 and 4 pm again) Dawn phenomenon( growth hormone and cortisol: hyperglycemia in the morning)

Drug Therapy Oral Agents ◦ Sulfonylureas ◦ ◦ � ↑ insulin production from pancreas � ↓ chance of prolonged hypoglycemia �Ex: glipizide (Glucotrol, Glucotrol XL), glyburide (Micronase, Dia. Beta, Glynase), and glimepiride (Amaryl). Meglitinides � Increase insulin production from pancreas � Taken 30 minutes before each meal up to time of meal � Should not be taken if meal skipped �Ex: repaglinide (Prandin) and nateglinide (Starlix). Biguanide � Reduce glucose production by liver � Enhance insulin sensitivity at tissues � Improve glucose transport into cells � Do not promote weight gain �Ex: Metformin (Glucophage) α-glucosidase inhibitors � “Starch blockers” � Slow down absorption of carbohydrate in small intestine � Ex: Acarbose (Precose) and miglitol (Glyset) Thiazaolidinediones �↑ glucose uptake in muscle and ↓glucose production �Ex: pioglitazone (Actos) and rosiglitazone (Avandia).

Drug Therapy Oral Agents � Dipeptidyl peptidase-4 (DDP-4) inhibitor ◦ Slows the inactivation of incretin hormones(Gastric hormones-Ex: gastrin, secretin…) ◦ Potential for hypoglycemia ◦ Ex: Sitagliptin (Januvia) � � � Saxagliptin (Onglyza) Amylin analog ◦ ◦ Hormone secreted by cells of pancreas Administered subcutaneously: thigh or abdomen Slows gastric emptying Ex: Pramlintide (Symlin) ◦ ◦ ◦ ◦ Synthetic peptide Stimulates release of insulin from cells Subcutaneous injection Suppresses glucagon secretion Reduces food intake Slows gastric emptying Not to be used with insulin Ex: Byetta Incretin mimetic -adrenergic blockers ◦ Mask symptoms of hypoglycemia � Thiazide/loop diuretics ◦ Can potentiate hyperglycemia � By inducing potassium loss

Diabetes Nutritional Therapy � American Diabetes Association (ADA) ◦ Overall goal �Assist people in making changes in nutrition and exercise habits that will lead to improved metabolic control. ◦ Meal plan is based on individual’s usual food intake and is balanced with insulin and exercise patterns.

Diabetes: Nutritional Therapy � Carbohydrates ◦ Sugars, starches, and fiber ◦ minimum of 130 g/day; whole grains, along with fruits, vegetables, and low-fat milk, should be included. � � � Fats ◦ Less than 200 mg/day of cholesterol and trans fats Protein Alcohol ◦ ◦ High in calories No nutritive value Detrimental effects on liver Can cause severe hypoglycemia • Alcohol inhibits gluconeogenesis (breakdown of glycogen to glucose) by the liver. This can cause severe hypoglycemia in patients on insulin or oral hypoglycemic medications that increase insulin secretion. • Moderate alcohol consumption can sometimes be safely incorporated into the meal plan if blood glucose levels are well controlled, and if the patient is not taking medications that will cause adverse effects.

Diabetes � Exercise ◦ Essential part of diabetes management ◦ ↑ insulin receptor sites ◦ Lowers blood glucose levels ◦ Contributes to weight loss • The ADA recommends that individuals with diabetes should perform at least 150 minutes per week of a moderate-intensity aerobic physical activity. • The ADA also encourages those with type 2 diabetes to perform resistance training 3 times a week in the absence of contraindications. ◦ Several small carbohydrate snacks can be taken every 30 minutes during exercise to prevent hypoglycemia. ◦ Best done after meals ◦ Exercise plans should be started after medical clearance ◦ Slowly with gradual progression ◦ Should be individualized ◦ Monitor blood glucose levels before, during, and after exercise.

Nursing Management Nursing Assessment � Past health history ◦ Viral infections ◦ Medications ◦ Recent surgery � Obesity � Weight � Thirst � Hunger � Poor loss healing • Kussmaul respirations (Rapid, deep breathing)

Nursing Management Nursing Diagnoses � Ineffective self-health management � Risk for injury � Risk for infection � Powerlessness � Imbalanced nutrition: More than body requirements

Nursing Management Planning � Overall goals ◦ Active patient participation ◦ Few or no episodes of acute hyperglycemic emergencies or hypoglycemia ◦ Maintain normal blood glucose levels. ◦ Prevent or delay chronic complications. ◦ Lifestyle adjustments with minimal stress

Nursing Management Nursing Implementation � Health promotion ◦ Identify those at risk, routine screening � Acute intervention for ◦ Hypoglycemia; Diabetic ketoacidosis; Hyperosmolar hyperglycemic nonketotic syndrome. ◦ Stress of illness and surgery �↑ blood glucose level �Continue taking oral agents and insulin. �Frequent monitoring of blood glucose �Patients undergoing surgery or radiologic procedures requiring contrast medium should hold their metformin on day of surgery and to 48 hour � Ambulatory ◦ ◦ ◦ and home care Overall goal is to reach an optimal level of independence Insulin therapy and oral agents Personal hygiene Medical identification and travel card Patient and family teaching �Educate on disease process, physical activity, medications, monitoring blood glucose, diet, resources.

Medical Alert

Nursing Management Evaluation � Knowledge � Balance of nutrition � Immune status � Health benefits � No injuries

Acute Complications � Diabetic ketoacidosis (DKA) � Hyperosmolar hyperglycemic syndrome (HHS) � Hypoglycemia � Table 49 -17 compares the manifestations, causes, management, and prevention of hyperglycemia and hypoglycemia(page 1242)

Diabetic ketoacidosis (DKA) ◦ Caused by profound deficiency of insulin �Characterized by �Hyperglycemia, Ketosis, Acidosis, Dehydration �Most likely occurs in type 1 ◦ Precipitating factors �Illness, Infection, Inadequate insulin dosage, Undiagnosed type 1, Poor self-management, and Neglect ◦ When supply of insulin insufficient �Glucose cannot be properly used for energy. �Body breaks down fat stores. �Ketones are by-products of fat metabolism. �Alter p. H balance, causing metabolic acidosis �Ketone bodies excreted in urine �Electrolytes become depleted.

DKA (cont’d) ◦ Signs and symptoms �Lethargy/weakness: early symptoms �Dehydration �Poor skin turgor �Dry mucous membranes �Tachycardia �Orthostatic hypotension �Abdominal pain �Anorexia, vomiting �Kussmaul respirations �Rapid deep breathing �Attempt to reverse metabolic acidosis �Sweet fruity odor ◦ Serious condition �Must be treated promptly

DKA (cont’d) Acute Complications ◦ Airway management �Oxygen administration ◦ Correct fluid/electrolyte imbalance �IV infusion 0. 45% or 0. 9% Na. Cl �Restore urine output. �Raise blood pressure. � When blood glucose levels approach 250 mg/d. L � 5% dextrose added to regimen �Prevent hypoglycemia. �Potassium replacement �Sodium bicarbonate ◦ Insulin therapy �Withheld until fluid resuscitation has begun. �Bolus followed by insulin drip

Hyperosmolar hyperglycemic syndrome (HHS) Life-threatening syndrome Less common than DKA Often occurs in patients older than 60 years with type 2 DM Patient has enough circulating insulin that ketoacidosis does not occur. ◦ Neurologic manifestations occur because of ↑ serum osmolality. ◦ Usually history of ◦ ◦ � Inadequate fluid intake � Increasing mental depression � Polyuria ◦ Laboratory values � Blood glucose >400 mg/d. L � Increase in serum osmolality � Absent/minimal ketone bodies ◦ Medical emergency; high mortality rate ◦ Therapy similar to DKA, except HHS requires greater fluid replacement

Nursing management DKA/HHS �Administration � IV fluids �Insulin therapy �Electrolytes �Assessment �Renal status �Cardiopulmonary status �Level of consciousness �Signs of potassium imbalance �Cardiac monitoring �Vital signs

Hypoglycemia ◦ Low blood glucose �Blood glucose level less than 70 mg/d. L ◦ Common manifestations �Confusion �Irritability �Diaphoresis �Tremors �Hunger �Weakness �Visual disturbances ◦ Untreated can progress to loss of consciousness, seizures, coma, and death • The balance between blood glucose and insulin can be disrupted by the administration of too much insulin or medication, the ingestion of too little food, delaying the time of eating, and performing unusual amounts of exercise.

Hypoglycemia (cont’d) �Check blood glucose �If <70 mg/d. L, begin treatment �If >70 mg/d. L, investigate further for cause of signs/symptoms �If monitoring equipment not available, treatment should be initiated �If alert enough to swallow � 15 to 20 g of a simple carbohydrate � 4 to 6 oz fruit juice �Regular soft drink �Avoid foods with fat �Decrease absorption of sugar �Gels or tablets containing specific amounts of glucose are convenient for carrying. • Avoid large quantities of quick-acting carbohydrates, so that rapid fluctuation to hyperglycemia does not occur.

Hypoglycemia (cont’d) ◦ Treatment �If alert enough to swallow �Recheck blood sugar 15 minutes after treatment. �Repeat until blood sugar >70 mg/d. L. �Patient should eat regularly scheduled meal/snack to prevent rebound hypoglycemia. �Check blood sugar again 45 minutes after treatment. Patient not alert enough to swallow �Administer 1 mg of glucagon IM or subcutaneously. �Side effect: Rebound hypoglycemia �Have patient ingest a complex carbohydrate after recovery. �In acute care settings � 20 to 50 m. L of 50% dextrose IV push

Angiopathy ◦ Macrovascular �Diseases of large and medium-sized blood vessels �one of the leading causes of diabetes-related deaths �Tight glucose control may delay atherosclerotic process. �Risk factors �Obesity �Smoking �Hypertension �High fat intake �Sedentary lifestyle

Angiopathy ◦ Microvascular �Result from thickening of vessel membranes in capillaries and arterioles �In response to chronic hyperglycemia �Is specific to diabetes, unlike macrovascular ◦ Areas most noticeably affected �Eyes (retinopathy) �Kidneys (nephropathy) �Skin (dermopathy) ◦ Clinical manifestations usually appear after 10 to 20 years of diabetes. ◦ Microvascular damage to retina �Result of chronic hyperglycemia

Angiopathy � Diabetic retinopathy (cont’d) ◦ Nonproliferative �Most common form �Partial occlusion of small blood vessels in retina � Causes development of microaneurysms � Capillary fluid leaks out. � Retinal edema and eventually hard exudates or intr-aretinal hemorrhages occur ◦ Proliferative �Most severe form �Involves retina and vitreous �When retinal capillaries become occluded � Body forms new blood vessels � Vessels are extremely fragile and hemorrhage easily � Produce vitreous contraction � Retinal detachment can occur

Diabetic retinopathy (cont’d) ◦ Treatment �Laser photocoagulation �Most common �Laser destroys ischemic areas of retina �Prevents further visual loss �Vitrectomy �Aspiration of blood, membrane, and fibers inside the eye

Diabetic nephropathy ◦ damage to small blood vessels that supply the glomeruli of the kidney ◦ Leading cause of end-stage renal disease � Risk factors for the development of diabetic nephropathy include hypertension, genetic predisposition, smoking, and chronic hyperglycemia.

Diabetic neuropathy � More than 60% of nontraumatic amputations in the United States occur in people with diabetes. • Sensory versus autonomic neuropathy ◦ Sensory neuropathy �Distal symmetric �Most common form �Affects hands and/or feet bilaterally �Characteristics include �Loss of sensation, abnormal sensations, pain. The pain is often described as burning, cramping, crushing, or tearing. �Usually worse at night �Foot injury and ulcerations can occur without the patient having pain. �Can cause atrophy of small muscles of hands/feet
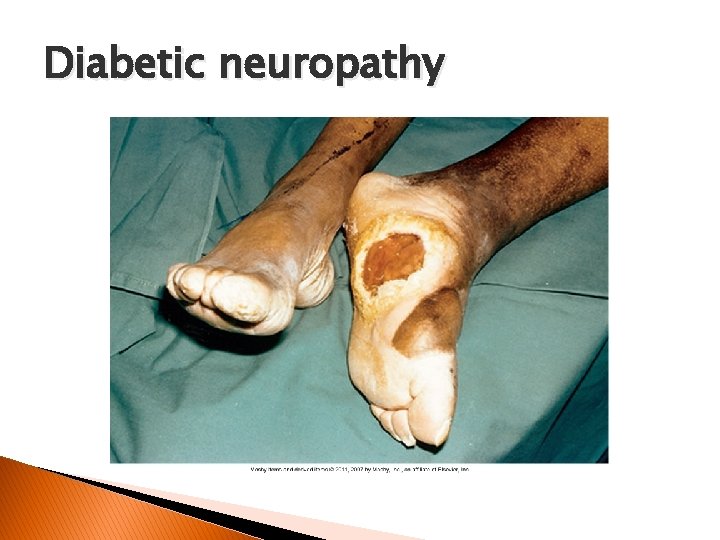
Diabetic neuropathy

Diabetic neuropathy ◦ Autonomic �Can affect nearly all body systems �Complications �Gastroparesis �Delayed gastric emptying �Cardiovascular abnormalities ◦ Foot complications �Most common cause of hospitalization in diabetes �Result from combination of microvascular and macrovascular diseases
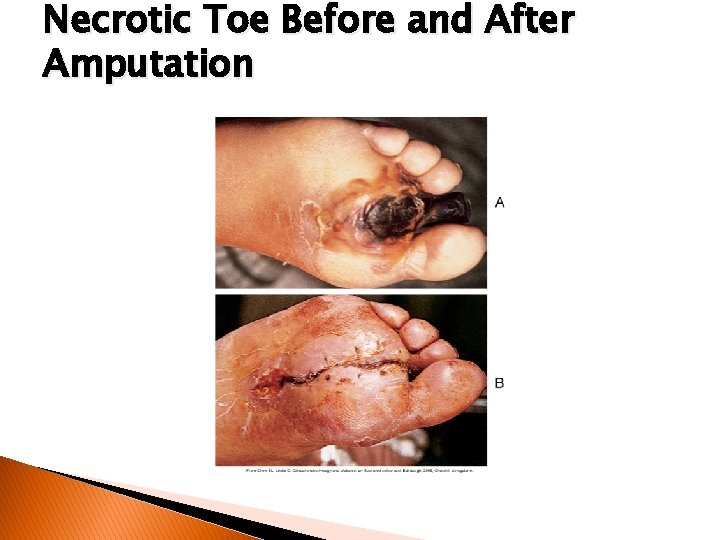
Necrotic Toe Before and After Amputation

Complications of foot and lower extremity (cont’d) ◦ Risk factors �Sensory neuropathy �Peripheral arterial disease ◦ Other contributors �Smoking �Clotting abnormalities. �Impaired immune function �Autonomic neuropathy

Infection ◦ Diabetic individuals more susceptible to infection ◦ Defect in mobilization of inflammatory cells ◦ Impairment of phagocytosis by neutrophils and monocytes ◦ Loss of sensation may delay detection.

Gerontologic Considerations • process of aging is associated with a reduction in β-cell function, decreased insulin sensitivity, and altered carbohydrate metabolism. • diabetes has been found to contribute to a greater rate of decline in cognitive function. � Recognize limitations in physical activity and visual acuity � Education based on individual’s needs, using slower pace.
- Slides: 69