Microvascular Complications of Diabetes Mellitus Harrison 19 e

Microvascular Complications of Diabetes Mellitus Harrison 19 e Brownlee M Nature (2001) 2016. 09. 05 학생집담회 내과 3년차 문선준

Introduction Diabetes : leading cause of new blindness, renal failure, nontraumatic L/E amputation Diabetes related complications are similar for type 1 and 2 DM. T 2 DM : long asymptomatic period of hyperglycemia before Dx. Many have complications at the time of Dx. Fortunately, early detection, aggressive glycemic control can reduce Cx.

Diabetes related complications Microvascular Macrovascular Other ü Eye disease Retinopathy (nonproliferative/proliferative) Macular edema ü Nephropathy (albuminuria and declining renal function) ü Neuropathy Sensory and motor (mono- and polyneuropathy) Autonomic ü Coronary heart disease ü Peripheral arterial disease ü Cerebrovascular disease ü Gastrointestinal (gastroparesis, diarrhea) ü Genitourinary (uropathy/sexual dysfunction) ü Dermatologic ü Infectious ü Cataracts ü Glaucoma ü Cheiroarthropathy ü Periodontal disease ü Hearing loss

Glycemic control and complication Several clinical studies Diabetes Control and Complications Trial (DCCT) Epidemiology of Diabetes Intervention and Complications (EDIC) United Kingdom Prospective Diabetes Study (UKPDS) Action to Control Cardiovascular Risk in Diabetes (ACCORD) Action in Diabetes and Vascular Disease (ADVANCE)

Glycemic control and complication Causative role of chronic hyperglycemia. Definite for microvascular Cx. , but not conclusive for macrovascular Cx. Duration and degree of hyperglycemia correlate with Cx. Intensive glycemic control is beneficial in all forms of DM. BP control is critical, especially in type 2 DM. Not all individuals with diabetes develop diabetes-related Cx. eg. Despite long-standing DM, some never develop nephropathy or retinopathy. Suggesting a genetic susceptibility for developing particular Cx. Legacy effect or metabolic memory Positive impact of a period of improved glycemic control on later ds.
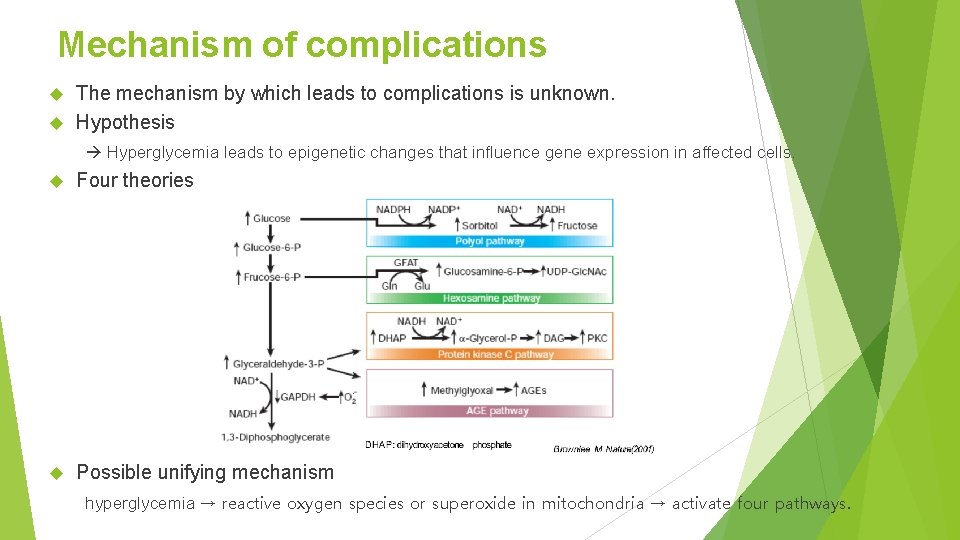
Mechanism of complications The mechanism by which leads to complications is unknown. Hypothesis Hyperglycemia leads to epigenetic changes that influence gene expression in affected cells. Four theories Possible unifying mechanism hyperglycemia → reactive oxygen species or superoxide in mitochondria → activate four pathways.

Ophthalmologic Cx. – clinical feature DM – leading cause of blindness of adults. 25 times more likely Severe vision loss d/t diabetic retinopathy, macular edema Nonproliferative diabetic retinopathy (NPDR) Late in the 1 st decade or early in the 2 nd decade. Retinal vascular microaneurysm, blot hemorrhages, cotton-wool spots. Pathophysiology : loss of retinal pericytes, increased retinal vascular permeability, alterations in retinal blood flow, abnormal retinal microvasculature → retinal ischemia

Ophthalmologic Cx. – clinical feature Proliferative diabetic retinopathy (PDR) Neovascularization in response to retinal hypoxemia (near the optic n. , macula) → vitreous hemorrhage, fibrosis, retinal detachment. More severe NPDR, more chance to develop PDR Macular edema Occur in the context of NPDR or PDR. 25% chance of moderate visual loss in 3 yrs. Fluorescent angiography , optical coherence tomography (OCT) : detect macular edema.
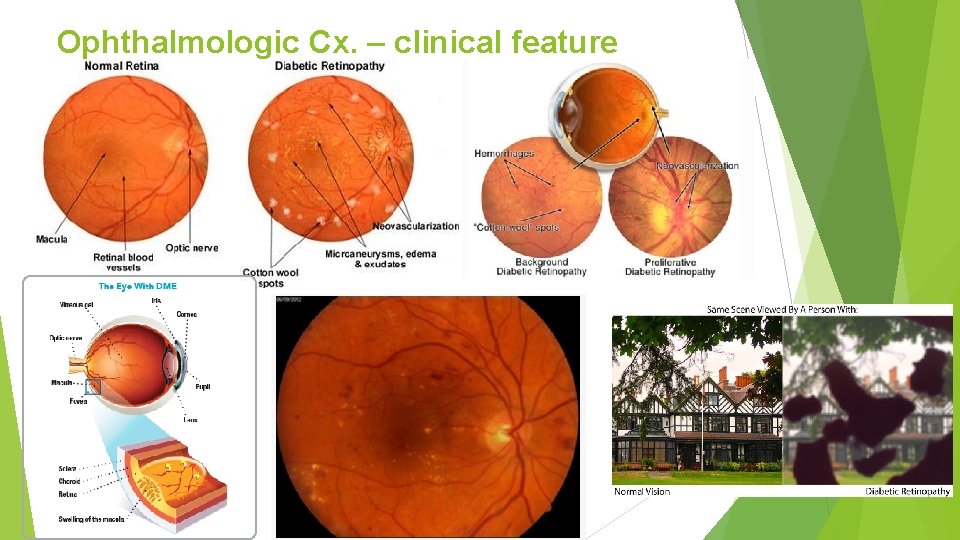
Ophthalmologic Cx. – clinical feature

Ophthalmologic Cx. – management Prevention – most effective therapy Intensive glycemic control, BP control → delay development or progression Prophylactic laser photocoagulation (successful in preserving vision) During the 1 st 6 -12 mo. of improved glycemic control, diabetic retinopathy may transiently worsen. → but finally effective. Once advanced retinopathy is present → glycemic control became less benefit Photocoagulation PDR : panretinal laser photocoagulation Macular edema : focal laser photocoagulation, anti-vascular endothelial GF tx.

Renal Cx. – clinical feature Diabetic nephropathy– leading cause of CKD and ESRD. Commonly have diabetic retinopathy Possible mechanism Soluble factors GF, angiotensin II, endothelin, AGEs Hemodynamic alterations glomerular hyperfiltration or hyperperfusion, increased glomerular capillary pressure Structural changes in glomerulus increased ECM, basement membrane thickening, mesangial expansion, fibrosis Ø Smoking accelerates decline in renal function. Ø Genetic or environmental susceptibility – d/t only 20 -40% of diabetes patients develop nephropathy

Renal Cx. – natural course (after onset of DM) first year : Glomerular hyperperfusion, renal hypertrophy → GFR ▲ During 5 yr : thickening of the glomerular basement memb, glomerular hypertrophy, mesangial volume expansion → GFR returns to normal. After 5 -10 yrs, begin to excrete albumin in the urine Microalbuminuria : 30 -299 mg/d (24 h collection), 30 -299 ug/mg (spot urine) Macroalbuminuria : >300 mg/d (24 h collection), >300 ug/mg (spot urine) ~50% of microalbuminuria progress to macroalbuminuria over 10 yr. Once macroalbuminuria present GFR ▼ steadily, 50% reach ESRD in 7 -10 yrs. Pathologic changes are likely irreversible.

Renal Cx. – management Glycemic control Once macroalbuminuria + → role of glycemic control is unclear. At later phase of declining renal function → insulin requirements ▼ Strict BP control Target : <140/90 mm. Hg Reduce albumin excretion and slowing the decline in renal function ACEi or ARB No benefit of intervention prior to onset of microalbuminuria Mechanism : reducing intraglomerular pr. , inhibition of angiotensin-driven sclerosing pathway.


Chronic Complications of DM Microvascular Eye disease Retinopathy (nonproliferative/proliferative) Macular edema Neuropathy Sensory and motor (mono- and polyneuropathy) Autonomic Nephropathy Macrovascular Coronary heart disease Peripheral arterial disease Cerebrovascular disease
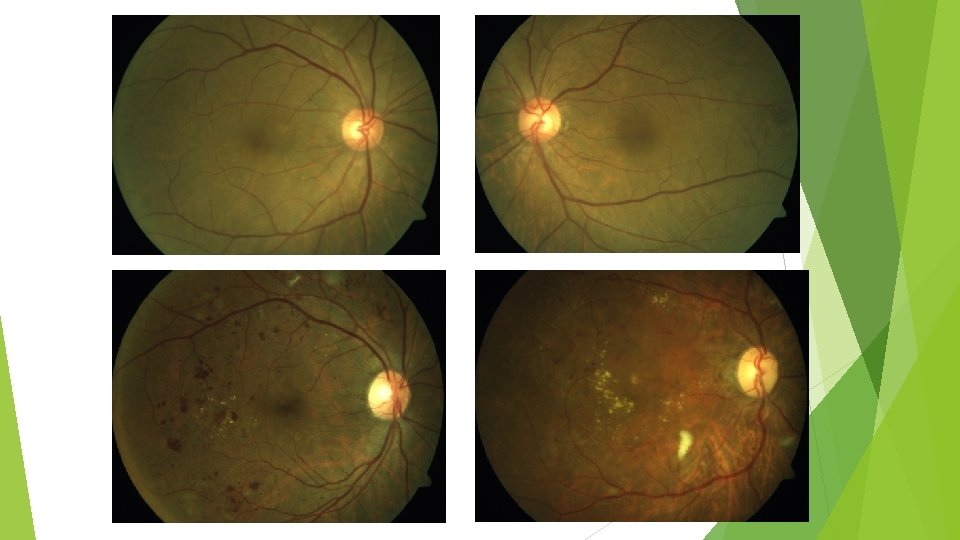
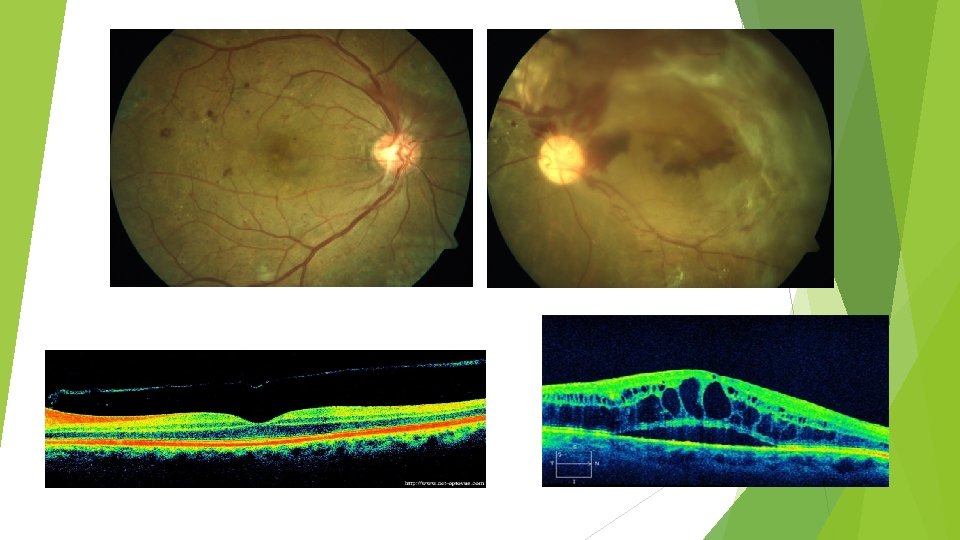
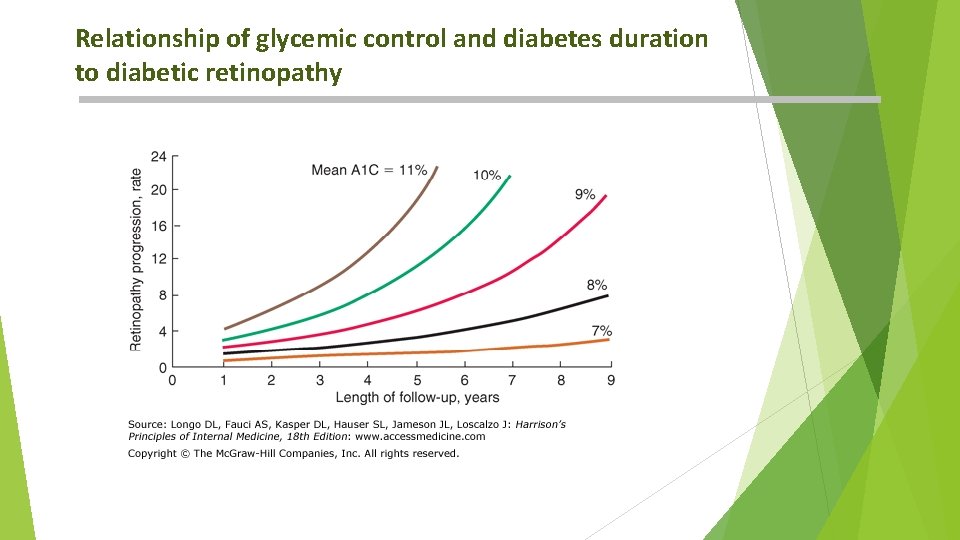
Relationship of glycemic control and diabetes duration to diabetic retinopathy
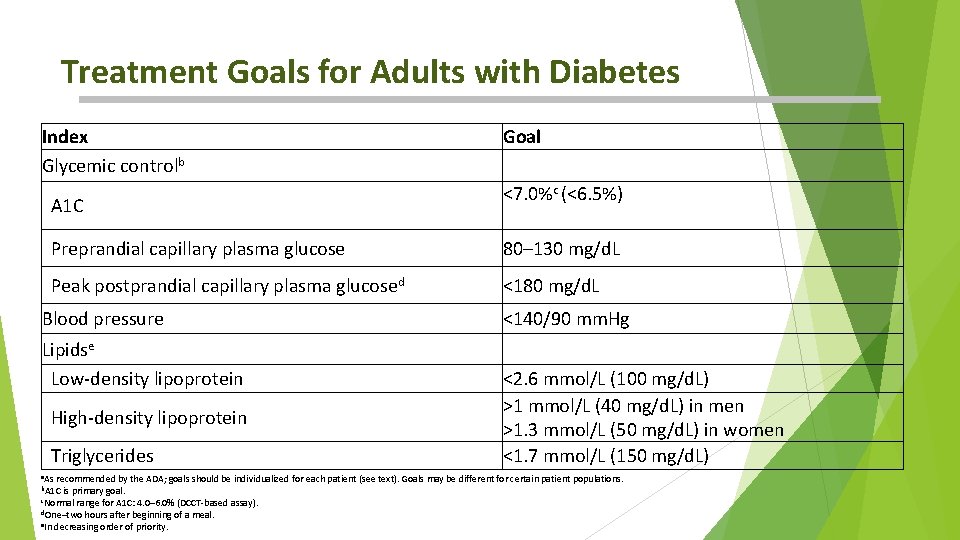
Treatment Goals for Adults with Diabetes Index Glycemic controlb A 1 C Goal <7. 0%c (<6. 5%) Preprandial capillary plasma glucose 80– 130 mg/d. L Peak postprandial capillary plasma glucosed <180 mg/d. L Blood pressure <140/90 mm. Hg Lipidse Low-density lipoprotein <2. 6 mmol/L (100 mg/d. L) >1 mmol/L (40 mg/d. L) in men >1. 3 mmol/L (50 mg/d. L) in women <1. 7 mmol/L (150 mg/d. L) High-density lipoprotein Triglycerides a. As recommended by the ADA; goals should be individualized for each patient (see text). Goals may be different for certain patient populations. b. A 1 C is primary goal. c. Normal range for A 1 C: 4. 0– 6. 0% (DCCT-based assay). d. One–two hours after beginning of a meal. e. In decreasing order of priority.
- Slides: 19