Type 2 Diabetes Update Prevention and Treatment Bruce


Type 2 Diabetes Update Prevention and Treatment Bruce W. Bode, MD, FACE Atlanta Diabetes Associates Atlanta, Georgia
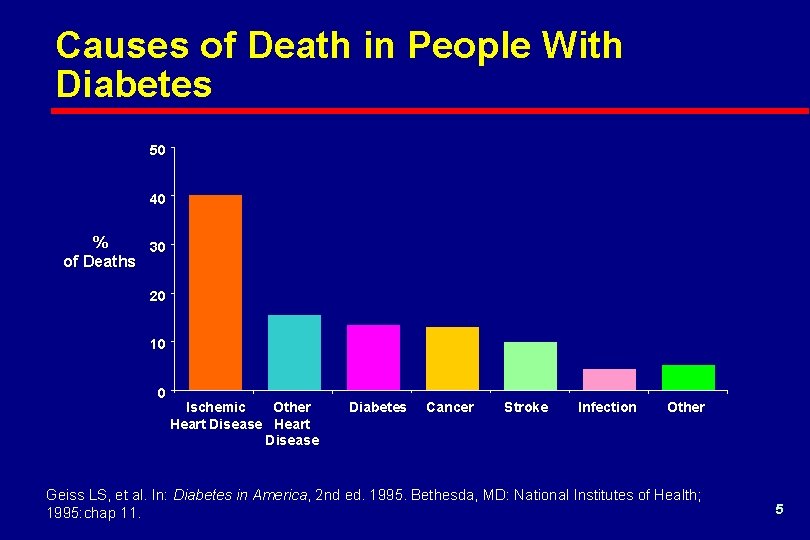
Causes of Death in People With Diabetes 50 40 % 30 of Deaths 20 10 0 Ischemic Other Heart Disease Diabetes Cancer Stroke Infection Other Geiss LS, et al. In: Diabetes in America, 2 nd ed. 1995. Bethesda, MD: National Institutes of Health; 1995: chap 11. 5
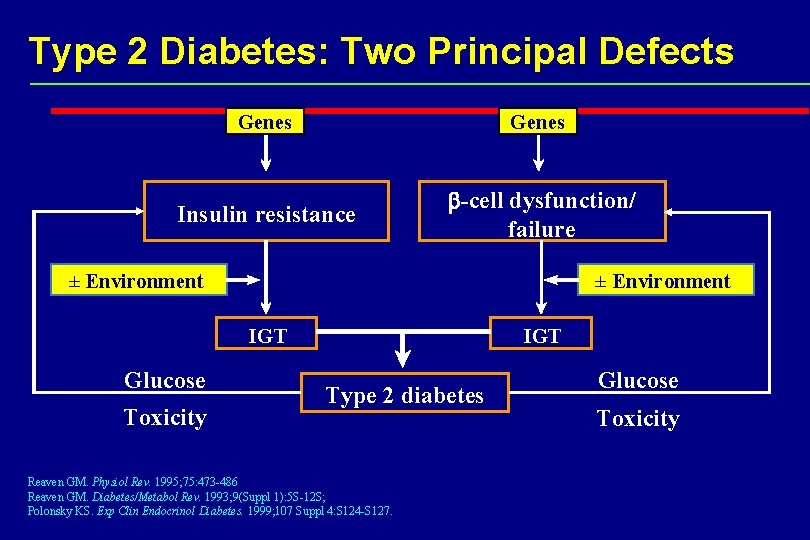
Type 2 Diabetes: Two Principal Defects Genes Insulin resistance b-cell dysfunction/ failure ± Environment IGT Glucose Toxicity IGT Type 2 diabetes Reaven GM. Physiol Rev. 1995; 75: 473 -486 Reaven GM. Diabetes/Metabol Rev. 1993; 9(Suppl 1): 5 S-12 S; Polonsky KS. Exp Clin Endocrinol Diabetes. 1999; 107 Suppl 4: S 124 -S 127. Glucose Toxicity
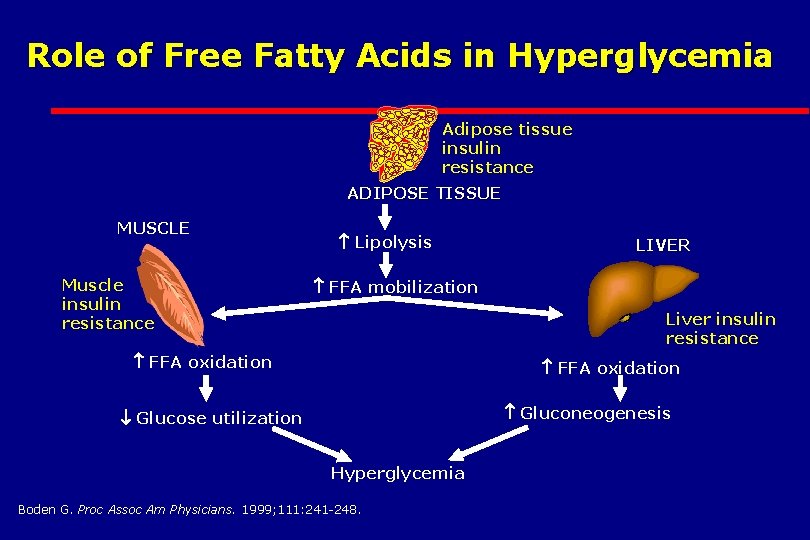
Role of Free Fatty Acids in Hyperglycemia Adipose tissue insulin resistance ADIPOSE TISSUE MUSCLE Muscle insulin resistance Lipolysis LIVER FFA mobilization Liver insulin resistance FFA oxidation Gluconeogenesis Glucose utilization Hyperglycemia Boden G. Proc Assoc Am Physicians. 1999; 111: 241 -248.
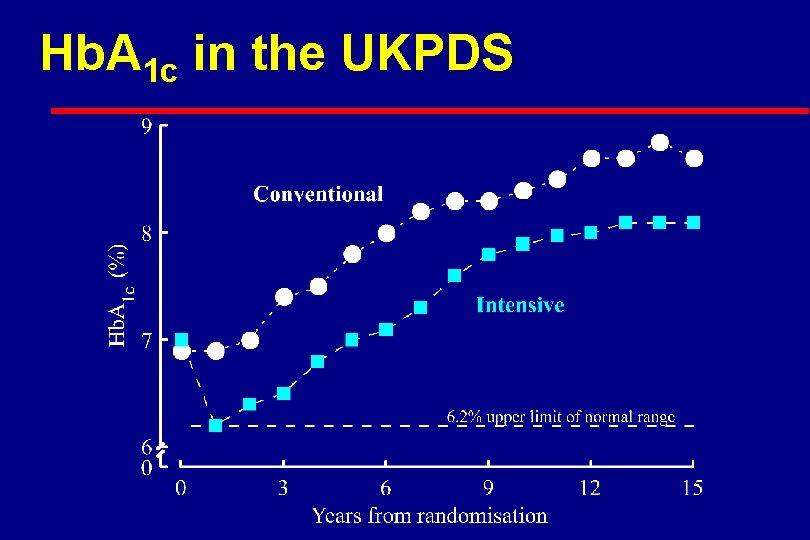
Hb. A 1 c in the UKPDS
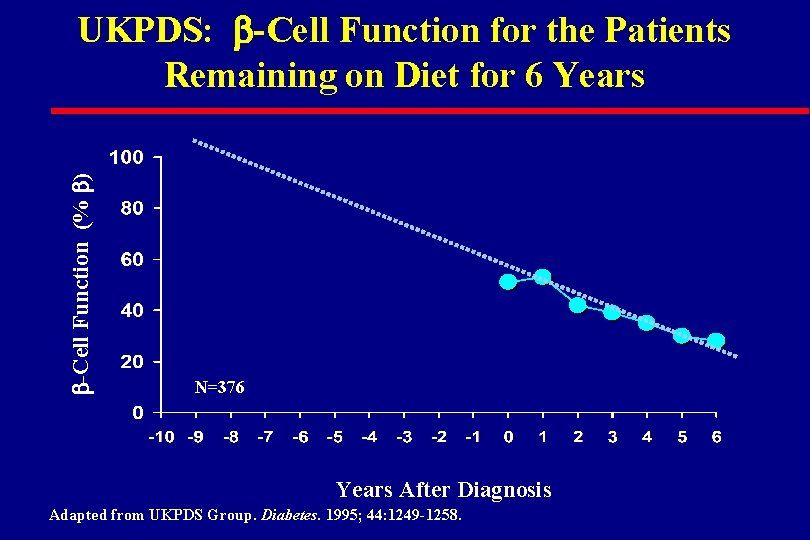
b-Cell Function (% b) UKPDS: b-Cell Function for the Patients Remaining on Diet for 6 Years N=376 Years After Diagnosis Adapted from UKPDS Group. Diabetes. 1995; 44: 1249 -1258.
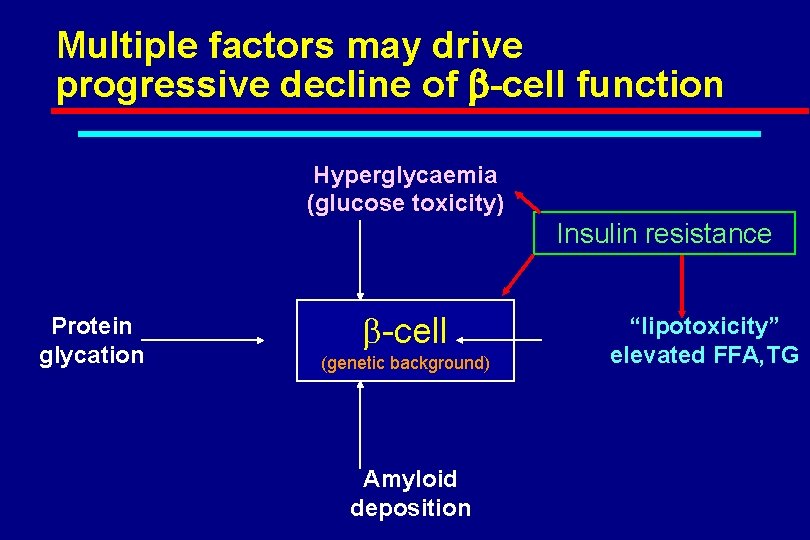
Multiple factors may drive progressive decline of b-cell function Hyperglycaemia (glucose toxicity) Insulin resistance Protein glycation b-cell (genetic background) Amyloid deposition “lipotoxicity” elevated FFA, TG
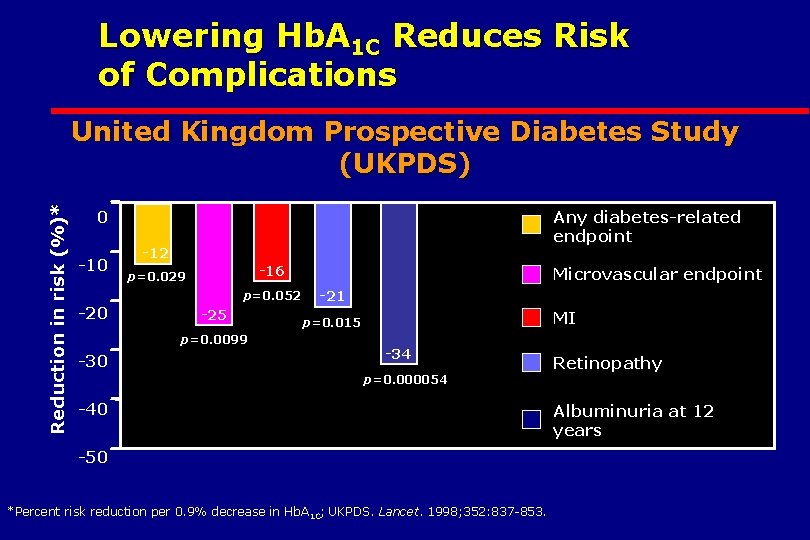
Lowering Hb. A 1 C Reduces Risk of Complications Reduction in risk (%)* United Kingdom Prospective Diabetes Study (UKPDS) Any diabetes-related endpoint 0 -12 -16 p=0. 029 p=0. 052 -20 -25 p=0. 0099 -30 Microvascular endpoint -21 MI p=0. 015 -34 Retinopathy p=0. 000054 -40 -50 *Percent risk reduction per 0. 9% decrease in Hb. A 1 C; UKPDS. Lancet. 1998; 352: 837 -853. Albuminuria at 12 years
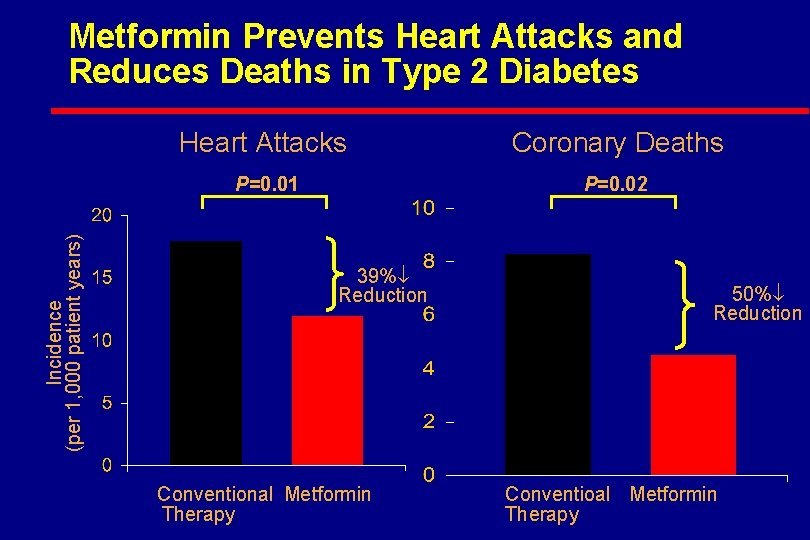
Incidence (per 1, 000 patient years) Metformin Prevents Heart Attacks and Reduces Deaths in Type 2 Diabetes Heart Attacks Coronary Deaths P=0. 01 P=0. 02 39% Reduction Conventional Metformin Therapy 50% Reduction Conventioal Metformin Therapy

Diabetes Prevention Program l 3234 obese patients with IGT l BMI average 34; A 1 C 5. 9%, 55% Caucasion l 4 year study to compare diet and exercise to metformin, troglitazone or control l Troglitazone stopped at 8 months l Study ended after 3 years

Diabetes Prevention Program l 58% prevention with diet (low fat) and exercise (2. 5 hours per week) l 31% prevention with metformin (more effective if < 60 years old and obese) l Troglitazone patients equal to metformin group at three years and equal to the diet and exercise group at 8 months.
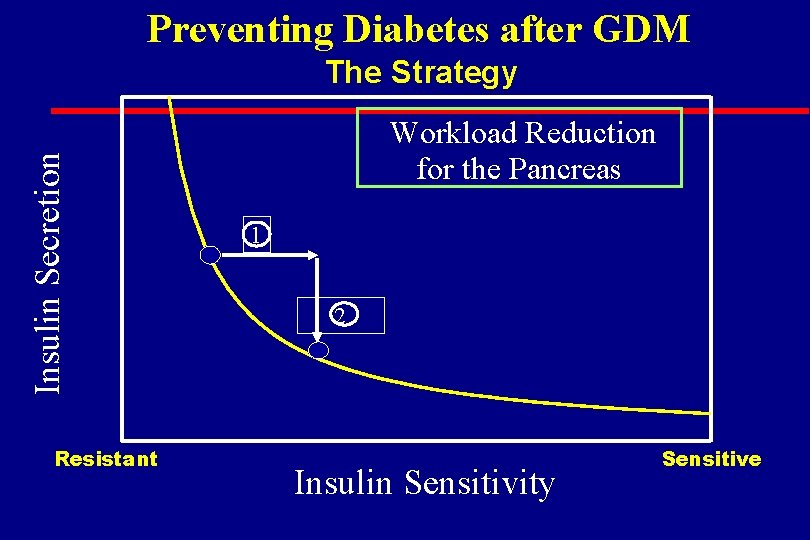
Preventing Diabetes after GDM Insulin Secretion The Strategy Resistant Workload Reduction for the Pancreas 1 2 Insulin Sensitivity Sensitive

Troglitazone In the Prevention Of Diabetes TRIPOD: A Test of “Workload Reduction” for B-cells Subjects Ø Non-pregnant, non-diabetic Hispanic women Ø Recent gestational diabetes (< 4 years) Ø 5 -year diabetes risk = 70% (o. GTT glucose sum) Procedures Ø Placebo vs 400 mg troglitazone daily Ø Fasting glucose every three months Ø o. GTT every year Ø iv. GTT at 0, 3, 24 months, Dx of diabetes and study end Main Outcome Variables Ø Diabetes incidence rates Azen et al: Contr Clin Trials, 1998

Preventing Diabetes: The “TRIPOD” Study People with Diabetes 60% 53% 40% RR=0. 44 Placebo 12. 3%/yr 20% 19% Troglitazone 0% 5. 4%/yr 0 10 20 30 40 50 Months on Study 60

Randomization Placebo DM Rate 12. 3%/yr Troglitazone Small Change in S I DM Rate 9. 8%/yr Large Change in S I Small Fall in Insulin Levels DM Rate 5. 8%/yr Large Fall in Insulin Levels DM Rate 0%/yr
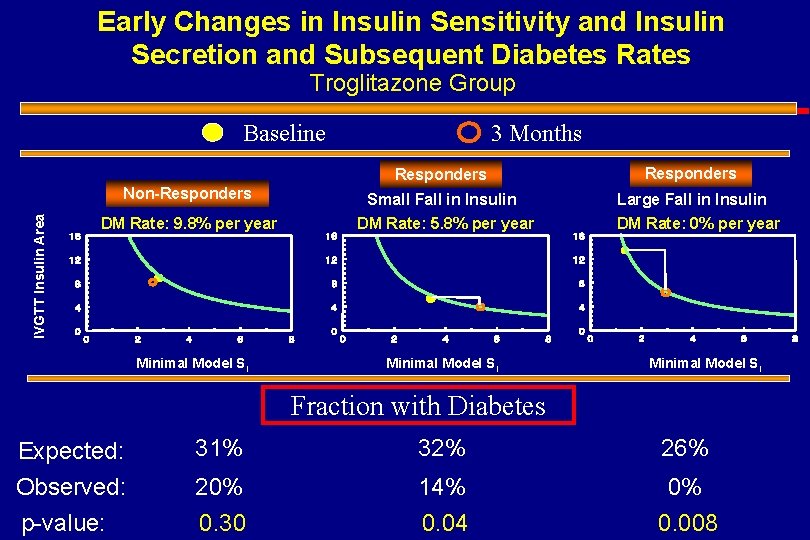
Early Changes in Insulin Sensitivity and Insulin Secretion and Subsequent Diabetes Rates Troglitazone Group Baseline Responders Large Fall in Insulin DM Rate: 9. 8% per year Small Fall in Insulin DM Rate: 5. 8% per year Minimal Model SI Non-Responders IVGTT Insulin Area 3 Months DM Rate: 0% per year Minimal Model SI Fraction with Diabetes Expected: Observed: 31% 32% 26% 20% 14% 0% p-value: 0. 30 0. 04 0. 008
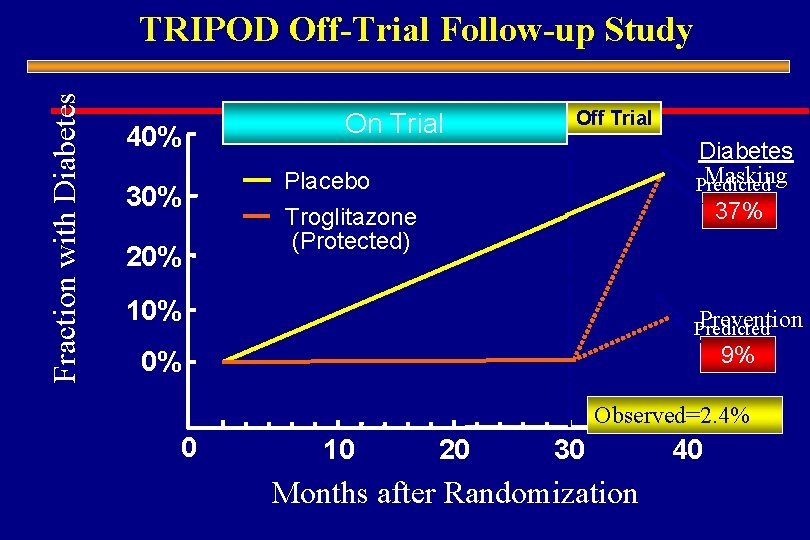
Fraction with Diabetes TRIPOD Off-Trial Follow-up Study 40% 30% 20% On Trial Off Trial Diabetes Masking Predicted 37% Placebo Troglitazone (Protected) 10% Prevention Predicted 9% 0% Observed=2. 4% 0 10 20 30 Months after Randomization 40

TRIPOD: Conclusion Reducing secretory demands placed on pancreatic B-cells by chronic insulin resistance can delay or prevent the onset of type 2 diabetes. “B-cell rest = B-cell protection”

Management of Type 2 DM Step Therapy l Diet l Exercise l Sulfonylurea or Metformin l Add Alternate Agent l Add hs NPH vs TZD l Switch to Mixed Insulin bid l Switch to Multiple Dose Insulin Prone to Failure from Negative Reinforcement Misscheduling, Mismanagement

Management of Type 2 DM Stumble Therapy l WAG Diet l Golf Cart Exercise l Sample of the Week Medication – Interrupted – Not Combined l Poor Understanding of Goals l Poor Monitoring Hb. A 1 c >8% (If Seen)

Consider A New Treatment Paradigm l Treatment designed to correct the dual impairments l Vigorous effort to meet glycemic targets l Simultaneous rather than sequential therapy l Combination therapy from the outset l Early step-wise titrations to meet glycemic targets

ACE / AACE Targets for Glycemic Control Hb. A 1 c < 6. 5 % Fasting/preprandial glucose< 110 mg/d. L Postprandial glucose < 140 mg/d. L ACE / AACE Consensus Conference, Washington DC August 2001

Goals in Management of Type 2 Diabetes l Fasting BG < 110 mg/d. L l Post-meal < 140 mg/d. L l Hb. A 1 c < 6. 5% l Blood Pressure < 130/80 l LDL < 100 mg/dl l HDL > 45 mg/dl
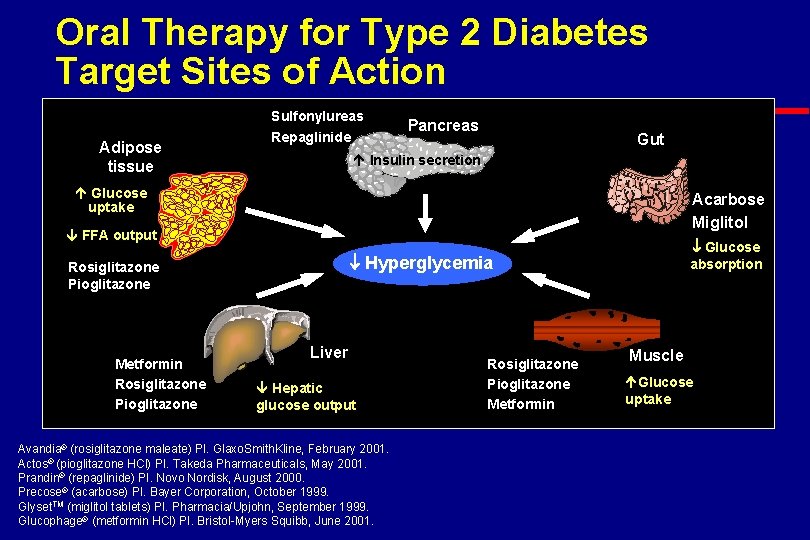
Oral Therapy for Type 2 Diabetes Target Sites of Action Adipose tissue Sulfonylureas Repaglinide Pancreas Gut Insulin secretion Glucose uptake Acarbose Miglitol FFA output Hyperglycemia Rosiglitazone Pioglitazone Metformin Rosiglitazone Pioglitazone Glucose absorption Liver Hepatic glucose output Avandia® (rosiglitazone maleate) PI. Glaxo. Smith. Kline, February 2001. Actos® (pioglitazone HCl) PI. Takeda Pharmaceuticals, May 2001. Prandin® (repaglinide) PI. Novo Nordisk, August 2000. Precose® (acarbose) PI. Bayer Corporation, October 1999. Glyset. TM (miglitol tablets) PI. Pharmacia/Upjohn, September 1999. Glucophage® (metformin HCl) PI. Bristol-Myers Squibb, June 2001. Rosiglitazone Pioglitazone Metformin Muscle Glucose uptake

Visceral Fat Distribution Normal versus Type 2 Diabetes Normal Courtesy of Wilfred Y. Fujimoto, MD. Type 2 Diabetes
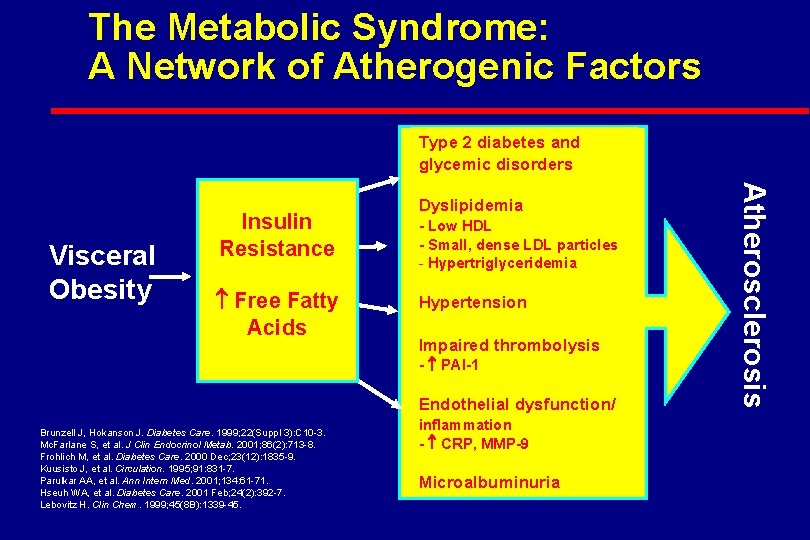
The Metabolic Syndrome: A Network of Atherogenic Factors Type 2 diabetes and glycemic disorders Free Fatty Acids - Low HDL - Small, dense LDL particles - Hypertriglyceridemia Hypertension Impaired thrombolysis - PAI-1 Endothelial dysfunction/ Brunzell J, Hokanson J. Diabetes Care. 1999; 22(Suppl 3): C 10 -3. Mc. Farlane S, et al. J Clin Endocrinol Metab. 2001; 86(2): 713 -8. Frohlich M, et al. Diabetes Care. 2000 Dec; 23(12): 1835 -9. Kuusisto J, et al. Circulation. 1995; 91: 831 -7. Parulkar AA, et al. Ann Intern Med. 2001; 134: 61 -71. Hseuh WA, et al. Diabetes Care. 2001 Feb; 24(2): 392 -7. Lebovitz H. Clin Chem. 1999; 45(8 B): 1339 -45. inflammation - CRP, MMP-9 Microalbuminuria Atherosclerosis Visceral Obesity Insulin Resistance Dyslipidemia

Thiazolidinediones: Mode of Action Peroxisome Proliferator-Activated Receptors l PPARg – Affects glucose, lipid and protein metabolism l PPARa – Affects lipoprotein metabolism (some TZDs) Saltiel & Olefsky. Diabetes 1996; 45: 1661– 9
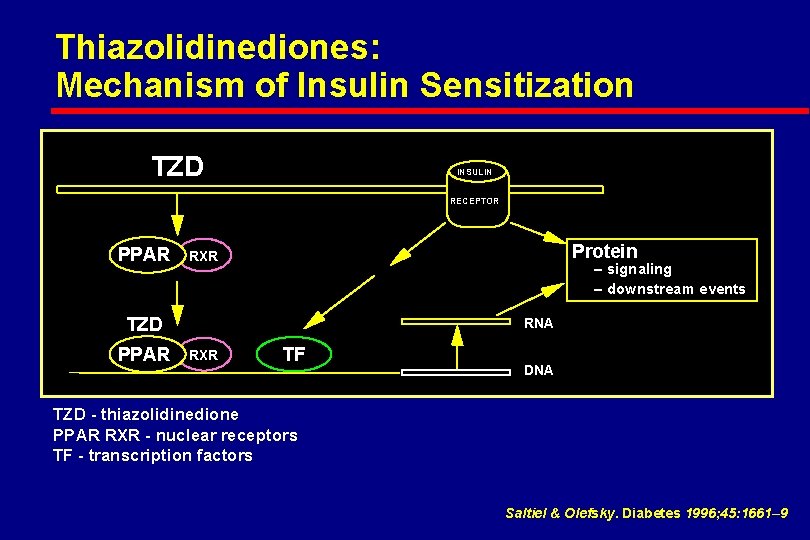
Thiazolidinediones: Mechanism of Insulin Sensitization TZD INSULIN RECEPTOR PPAR Protein RXR – signaling – downstream events TZD PPAR RNA RXR TF DNA TZD - thiazolidinedione PPAR RXR - nuclear receptors TF - transcription factors Saltiel & Olefsky. Diabetes 1996; 45: 1661– 9

Thiazolidinediones: Rationale for Type 2 Diabetes Therapy l Proven characteristics – Target insulin resistance, a core defect – Improve glycemic control – Do not cause hypoglycemia – Improve lipid profile (pioglitazone and troglitazone) l Potential benefits – Preservation of pancreatic b-cell function – Prevention of progression from impaired glucose tolerance to type 2 diabetes – Improvement in cardiovascular outcomes Saltiel & Olefsky. Diabetes 1996; 45: 1661– 9 Sonnenberg and Kotchen. Curr Opin Nephrol Hypertens 1998; 7(5): 551– 5
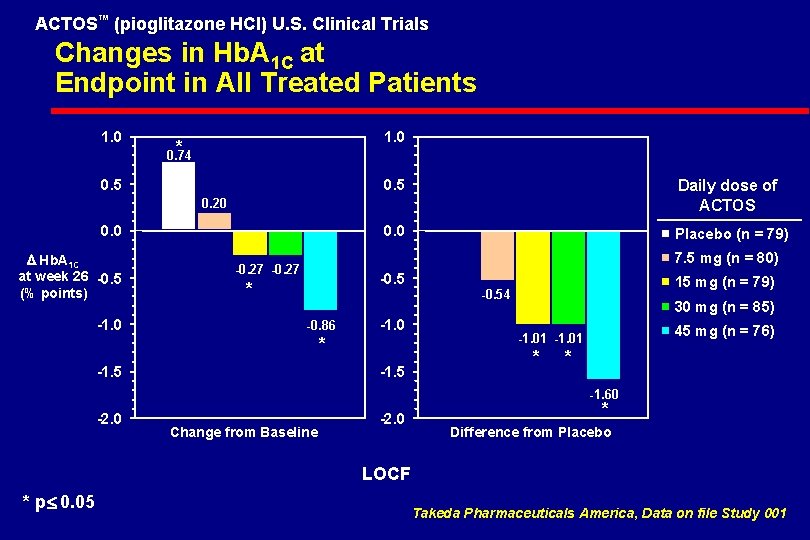
ACTOS™ (pioglitazone HCl) U. S. Clinical Trials Changes in Hb. A 1 C at Endpoint in All Treated Patients 1. 0 * 0. 74 0. 5 Daily dose of ACTOS 0. 0 Placebo (n = 79) 0. 20 0. 0 D Hb. A 1 C at week 26 -0. 5 (% points) -1. 0 7. 5 mg (n = 80) -0. 27 -0. 5 * 15 mg (n = 79) -0. 54 -0. 86 -1. 0 * -1. 5 30 mg (n = 85) 45 mg (n = 76) -1. 01 * * -1. 60 -2. 0 Change from Baseline -2. 0 * Difference from Placebo LOCF * p£ 0. 05 Takeda Pharmaceuticals America, Data on file Study 001
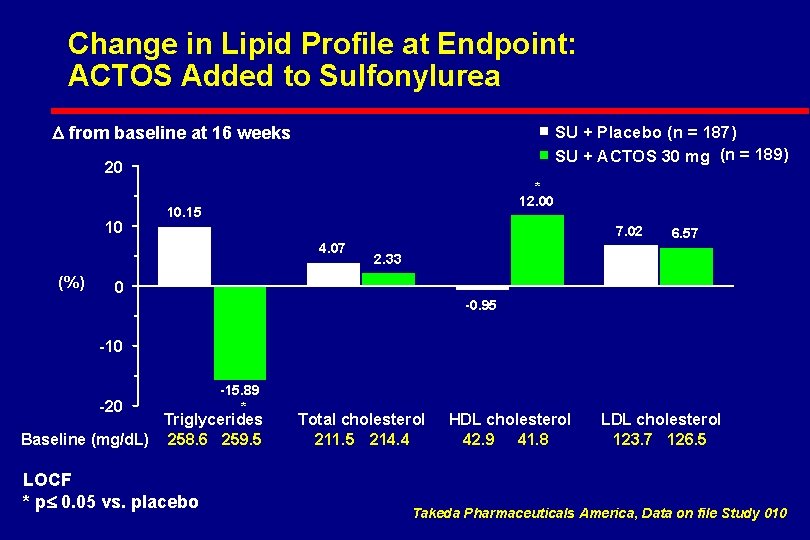
Change in Lipid Profile at Endpoint: ACTOS Added to Sulfonylurea D from baseline at 16 weeks SU + Placebo (n = 187) SU + ACTOS 30 mg (n = 189) 20 * 10 12. 00 10. 15 7. 02 4. 07 (%) 6. 57 2. 33 0 -0. 95 -10 -20 -15. 89 * Triglycerides Baseline (mg/d. L) 258. 6 259. 5 LOCF * p£ 0. 05 vs. placebo Total cholesterol 211. 5 214. 4 HDL cholesterol 42. 9 41. 8 LDL cholesterol 123. 7 126. 5 Takeda Pharmaceuticals America, Data on file Study 010
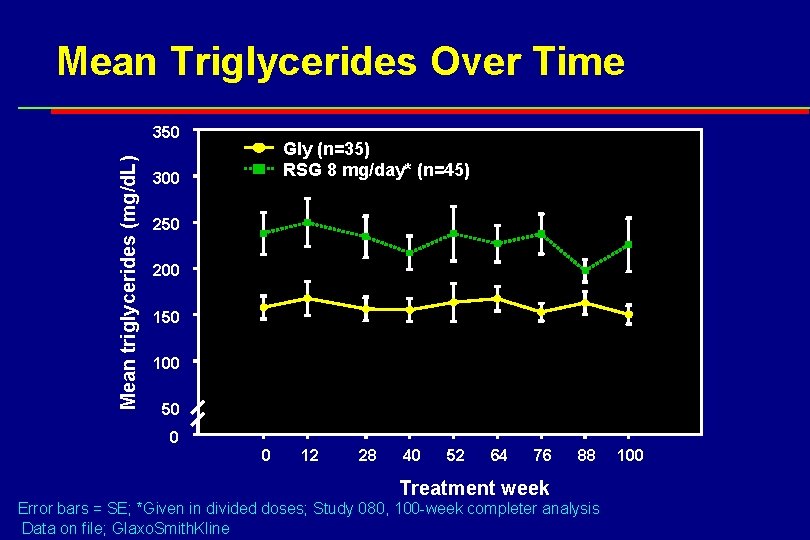
Mean Triglycerides Over Time Mean triglycerides (mg/d. L) 350 Gly (n=35) RSG 8 mg/day* (n=45) 300 250 200 150 100 50 0 0 12 28 40 52 64 76 88 Treatment week Error bars = SE; *Given in divided doses; Study 080, 100 -week completer analysis Data on file; Glaxo. Smith. Kline 100
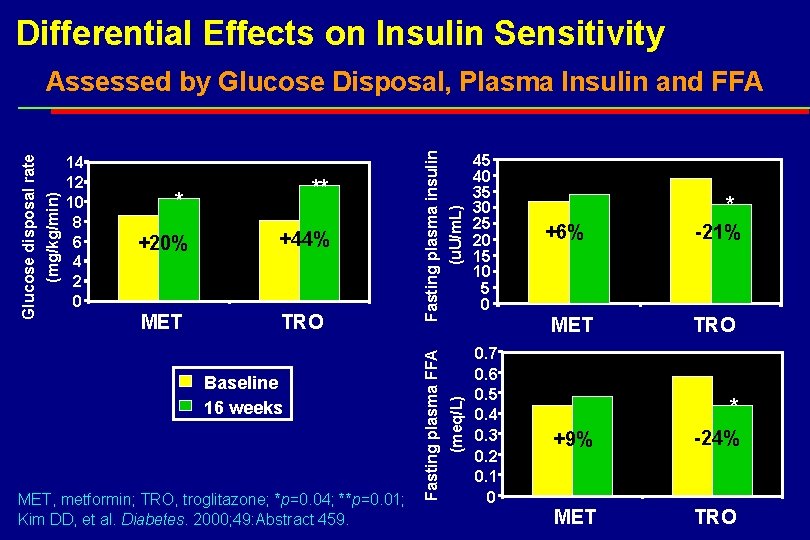
Differential Effects on Insulin Sensitivity +20% +44% MET TRO Baseline 16 weeks MET, metformin; TRO, troglitazone; *p=0. 04; **p=0. 01; Kim DD, et al. Diabetes. 2000; 49: Abstract 459. 45 40 35 30 25 20 15 10 5 0 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 (meq/L) ** * Fasting plasma insulin (u. U/m. L) 14 12 10 8 6 4 2 0 Fasting plasma FFA (mg/kg/min) Glucose disposal rate Assessed by Glucose Disposal, Plasma Insulin and FFA +6% MET * -21% TRO * +9% -24% MET TRO
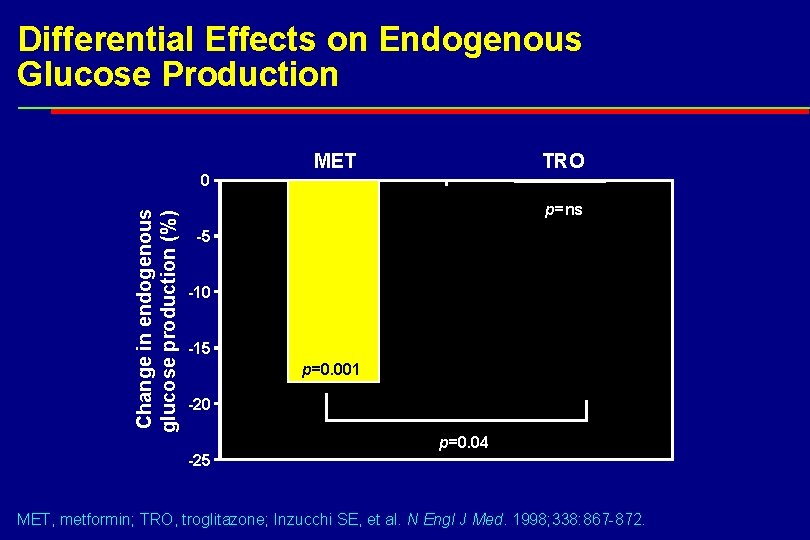
Differential Effects on Endogenous Glucose Production Change in endogenous glucose production (%) 0 MET TRO p=ns -5 -10 -15 p=0. 001 -20 p=0. 04 -25 MET, metformin; TRO, troglitazone; Inzucchi SE, et al. N Engl J Med. 1998; 338: 867 -872.
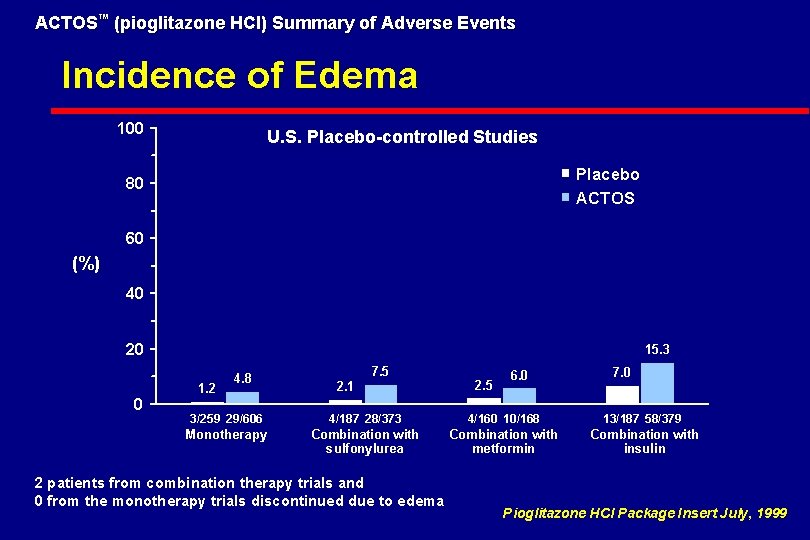
ACTOS™ (pioglitazone HCl) Summary of Adverse Events Incidence of Edema 100 U. S. Placebo-controlled Studies Placebo ACTOS 80 60 (%) 40 20 0 15. 3 1. 2 4. 8 7. 5 2. 1 2. 5 6. 0 7. 0 3/259 29/606 4/187 28/373 4/160 10/168 13/187 58/379 Monotherapy Combination with sulfonylurea Combination with metformin Combination with insulin 2 patients from combination therapy trials and 0 from the monotherapy trials discontinued due to edema Pioglitazone HCl Package Insert July, 1999

Patient Case 1 l Patient JM DOB 11/30/1943 l Problem List: 1. Diabetes Mellitus, onset 1981, age 38 a. Insulin therapy, 10/88 through 3/92, A 1 c 9. 0% C-peptide of 5. 1 b. Metformin and Glyburide therapy. A 1 c 7. 7%- 8. 9% c. Troglitazone therapy 400 mg/day, 5/97 through 3/99: A 1 c 8. 9% to 7. 1% then to 8. 1% d. Pioglitazone added 12/99: A 1 c 8. 1% to 6. 3 -6. 5% range.

Case 1 Cont’d 2. Hypertriglyceridemia l Simvastatin therapy with TG of 402 mg/d. L l Pioglitazone added l TG levels fell to 140 mg/d. L

Patient Case 2 l Patient CM DOB 11/17/1935 l Problem List: 1. Type 2 Diabetes, Onset 1993, age 59 a. Troglitazone added to Metformin and Repaglinide, 3/98: A 1 c 9. 6% to 8. 1% b. Pioglitazone 45 mg added 1/26/00, A 1 c 8. 1% to 7. 3%, avg glucose 121 mg/d. L TG decreased from 206 mg/d. L to 118 mg/d. L Tchol 164 mg/d. L to 130 mg/d. L, LDL 82 mg/d. L to 58 mg/d. L, HDL 41 to 46. 2. CHD: status post PTCA 1989, 1999.

Patient Case 3 l Patient EM; DOB 9/27/1932 1. Type 2 DM, Onset 1995, age 63 Insulin therapy since onset, post 15 lbs wt loss to 185 lbs Height 71 inches Glucose well controlled on NPH and Regular bid until 5/99, A 1 c 8. 7% Changed to NPH at am and hs with pre-meal Lispro. A 1 c to 7. 0% on 54 - 81 units/day. Weight 229 pounds Desires insulin pump therapy C-peptide 3. 7

Patient Case 3 Cont’d l Changed to Pioglitazone 30 mg/day, Glimepiride 4 mg/d, off insulin. l A 1 c down 6. 2%. l TG decreased from 546 to 182 to 79 mg/d. L. l LDL 191 to 132 (Atorvastatin 10 mg/day).
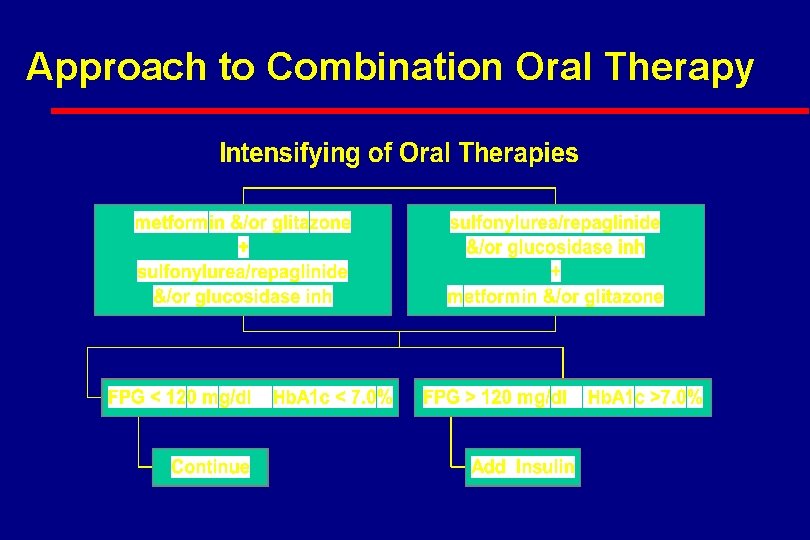
Approach to Combination Oral Therapy

Insulin Therapy in Type 2 Diabetes Indications l Significant hyperglycemia at presentation l Hyperglycemia on maximal doses of oral agents l Decompensation – – Acute injury, stress, infection, myocardial ischemia Severe hyperglycemia with ketonemia and/or ketonuria Uncontrolled weight loss Use of diabetogenic medications (eg, corticosteroids) l Surgery l Pregnancy l Renal or hepatic disease

Mimicking Nature The Basal/Bolus Insulin Concept 6 -16

Treat to Target Study: Glargine vs NPH Added to Oral Therapy of Type 2 Diabetes l Type 2 DM on 1 or 2 oral agents (SU, MET, TZD) l Age 30 to 70 l BMI 26 to 40 l A 1 C 7. 5 to 10% and FPG > 140 mg/d. L l Anti GAD negative l Willing to enter a 24 week randomized, open labeled study Riddle et al, Diabetes June 2002, Abstract 457 -p

Treat to Target Study: Glargine vs NPH Added to Oral Therapy of Type 2 Diabetes l Add 10 units Basal insulin at bedtime (NPH or Glargine) l Continue current oral agents l Titrate insulin weekly to fasting BG < 100 mg/d. L - if 100 -120 mg/d. L, increase 2 units - if 120 -140 mg/d. L, increase 4 units - if 140 -160 mg/d. L, increase 6 units - if 160 -180 mg/d. L, increase 8 units Riddle et al, Diabetes June 2002, Abstract 457 -p

Treat to Target Study; A 1 C Decrease Riddle et al, Diabetes June 2002, Abstract 457 -p
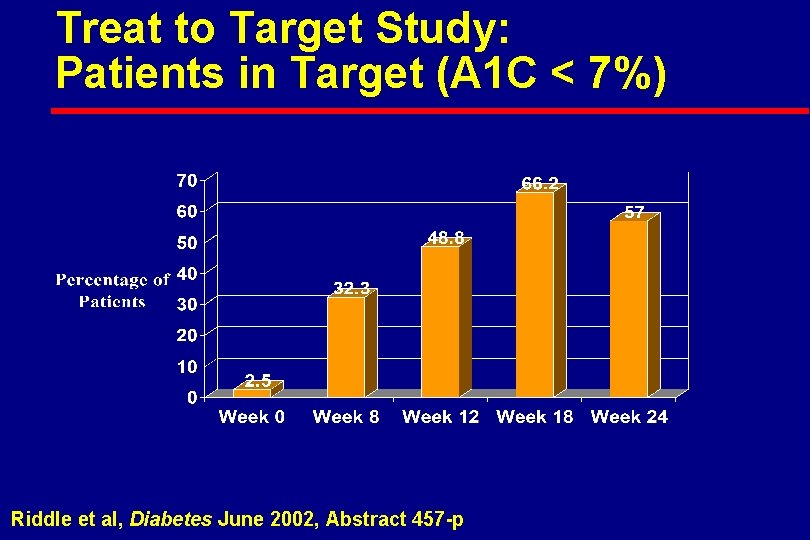
Treat to Target Study: Patients in Target (A 1 C < 7%) Riddle et al, Diabetes June 2002, Abstract 457 -p

Treat to Target Study: Glargine vs NPH Added to Oral Therapy of Type 2 Diabetes l Nocturnal Hypoglycemia reduced by 40% in the Glargine group (532 events) vs NPH group (886 events) Riddle et al, Diabetes June 2002, Abstract 457 -p

Advancing Basal/Bolus Insulin l Indicated when FBG acceptable but – Hb. A 1 c > 7% or > 6. 5% and/or – SMBG before dinner > 140 mg/d. L l Insulin options – To glargine or NPH, add mealtime aspart / lispro – To suppertime 70/30, add morning 70/30 – Consider insulin pump therapy l Oral agent options – Usually stop sulfonylurea – Continue metformin for weight control – Continue glitazone for glycemic stability?

Insulin Monotherapy vs Combination l 88 Type 2 patients on insulin monotherapy Baseline A 1 C 8. 7% l Randomized for 4 months to: Monotherapy with titration to A 1 C 5. 6% Metformin 1000 mg BID (no titration) Troglitazone 600 mg per day (no titration) Strowig et al, Diabetes Care 25, 10, October 2002
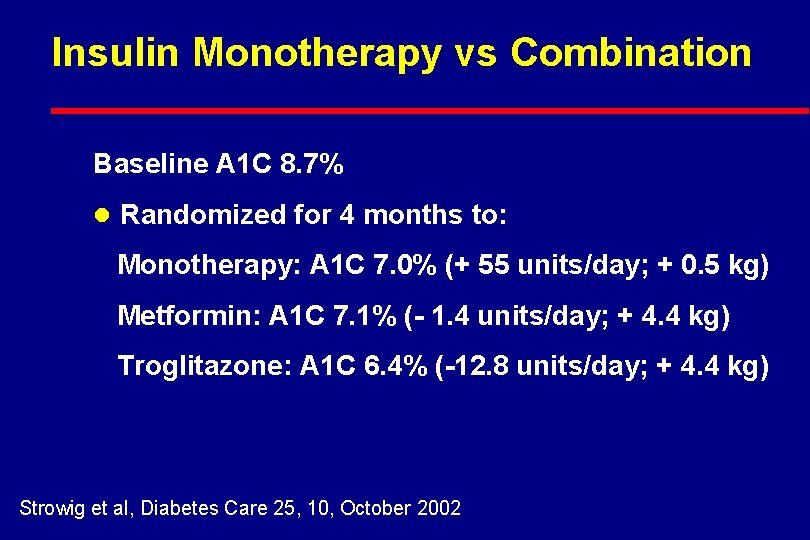
Insulin Monotherapy vs Combination Baseline A 1 C 8. 7% l Randomized for 4 months to: Monotherapy: A 1 C 7. 0% (+ 55 units/day; + 0. 5 kg) Metformin: A 1 C 7. 1% (- 1. 4 units/day; + 4. 4 kg) Troglitazone: A 1 C 6. 4% (-12. 8 units/day; + 4. 4 kg) Strowig et al, Diabetes Care 25, 10, October 2002

Multiple Daily Injections l Starting dose 0. 2 x wgt. in lbs. l Wgt. 180 lbs which = 36 units l Bolus dose (lispro/aspart) = 20% of starting dose at each meal, which = 7 units ac (tid) l Basal dose (glargine) = 40% of starting dose at HS, which = 14 units at HS l Correction bolus = (BG - 100)/ SF, where SF = 1500/total daily dose = 1500/36 = 40

Correction Bolus Formula Current BG - Ideal BG Glucose Correction factor Example: – Current BG: – Ideal BG: 220 mg/dl 100 mg/dl – Glucose Correction Factor: 40 mg/dl 220 - 100 =3. 0 u 40

Strategies to Improve Glycemic Control: Type 2 Diabetes l Monitor glycemic targets – Fasting and postprandial glucose, Hb. A 1 c l Self-monitoring of blood glucose is essential l Nutrition and activity are cornerstones of therapy l Combinations of pharmacologic agents are often necessary to achieve glycemic targets

Conclusion Intensive therapy is the best way to treat patients with diabetes
- Slides: 56