Approccio Terapeutico Diabetologico La terapia ipoglicemizzante nella nefropatia

Approccio Terapeutico Diabetologico La terapia ipoglicemizzante nella nefropatia Giuseppe Penno Dipartimento di Medicina Clinica e Sperimentale Azienda Ospedaliera Universitaria di Pisa

Dichiarazione esplicita di trasparenza delle fonti di finanziamento e dei rapporti con soggetti portatori di interessi commerciali Il sottoscritto Dr. Giuseppe Moderatore in qualità di Penno Relatore ai sensi dell’art. 3. 3 sul Conflitto di Interessi, pag. 17 del Reg. Applicativo dell’Accordo Stato. Regione del 5 novembre 2009, per conto del Provider SID Società Italiana di Diabetologia dichiara che negli ultimi due anni ha avuto i seguenti rapporti anche di finanziamento con soggetti portatori di interessi commerciali in campo sanitario: Ely Lilly, Janssen, MSD 18 giugno 2015

Point by point: Type 1 Diabetes ü background ü effects of intervention EURODIAB IDDM PCS DCCT/EDIC Joslin Study on the Natural History of Microalbuminuria DCCT/EDIC FINNDIANE
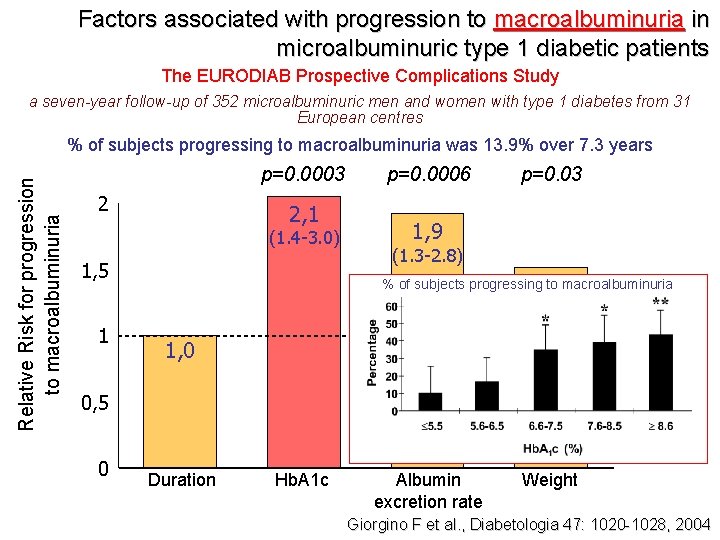
Factors associated with progression to macroalbuminuria in microalbuminuric type 1 diabetic patients The EURODIAB Prospective Complications Study a seven-year follow-up of 352 microalbuminuric men and women with type 1 diabetes from 31 European centres Relative Risk for progression to macroalbuminuria % of subjects progressing to macroalbuminuria was 13. 9% over 7. 3 years p=0. 0003 2 2, 1 (1. 4 -3. 0) 1, 5 p=0. 0006 p=0. 03 1, 9 (1. 3 -2. 8) % of subjects progressing 1, 5 to macroalbuminuria (1. 1 -2. 3) 1 1, 0 0, 5 0 Duration Hb. A 1 c Albumin excretion rate Weight Giorgino F et al. , Diabetologia 47: 1020 -1028, 2004
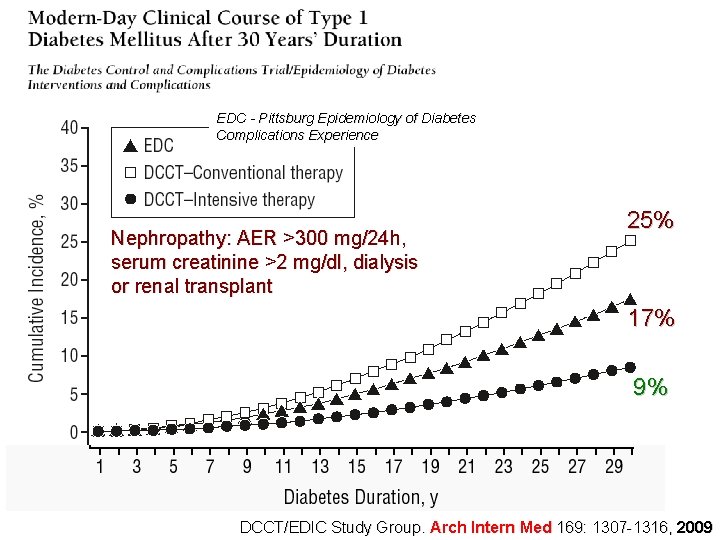
EDC - Pittsburg Epidemiology of Diabetes Complications Experience Nephropathy: AER >300 mg/24 h, serum creatinine >2 mg/dl, dialysis or renal transplant 25% 17% 9% DCCT/EDIC Study Group. Arch Intern Med 169: 1307 -1316, 2009
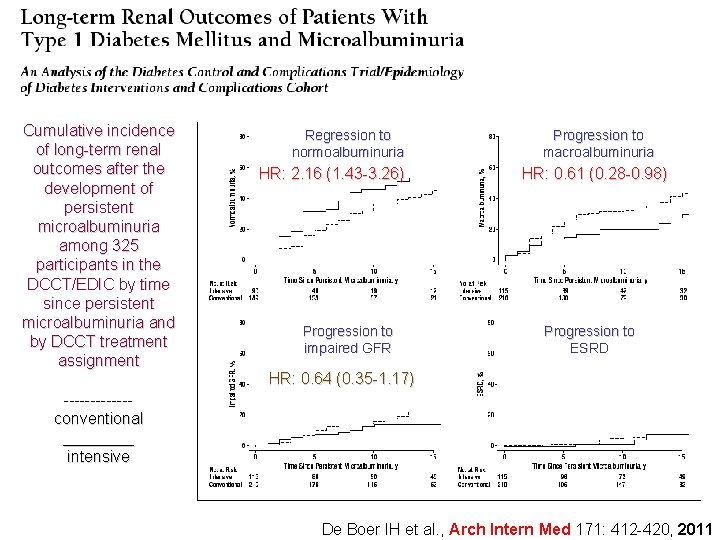
Cumulative incidence of long-term renal outcomes after the development of persistent microalbuminuria among 325 participants in the DCCT/EDIC by time since persistent microalbuminuria and by DCCT treatment assignment Regression to normoalbuminuria HR: 2. 16 (1. 43 -3. 26) Progression to impaired GFR Progression to macroalbuminuria HR: 0. 61 (0. 28 -0. 98) Progression to ESRD HR: 0. 64 (0. 35 -1. 17) ------conventional ____ intensive De Boer IH et al. , Arch Intern Med 171: 412 -420, 2011
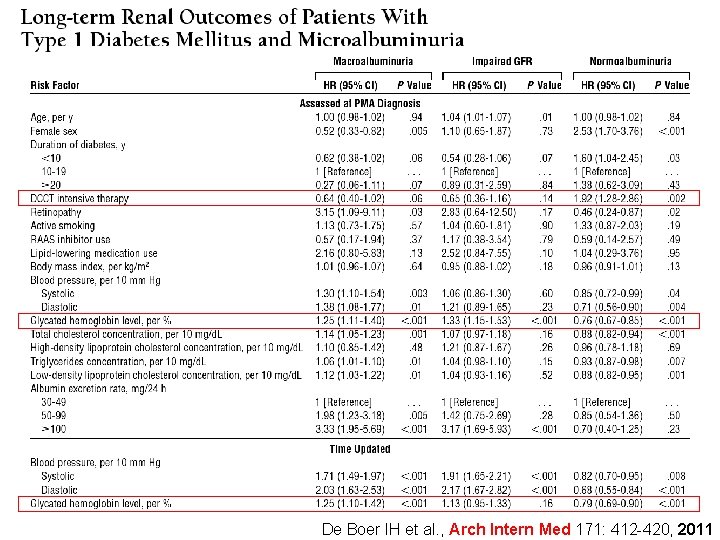
De Boer IH et al. , Arch Intern Med 171: 412 -420, 2011
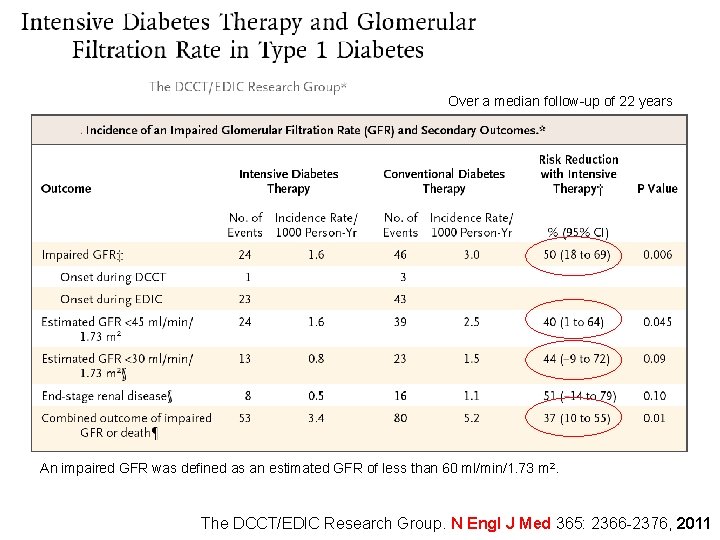
Over a median follow-up of 22 years An impaired GFR was defined as an estimated GFR of less than 60 ml/min/1. 73 m 2. The DCCT/EDIC Research Group. N Engl J Med 365: 2366 -2376, 2011
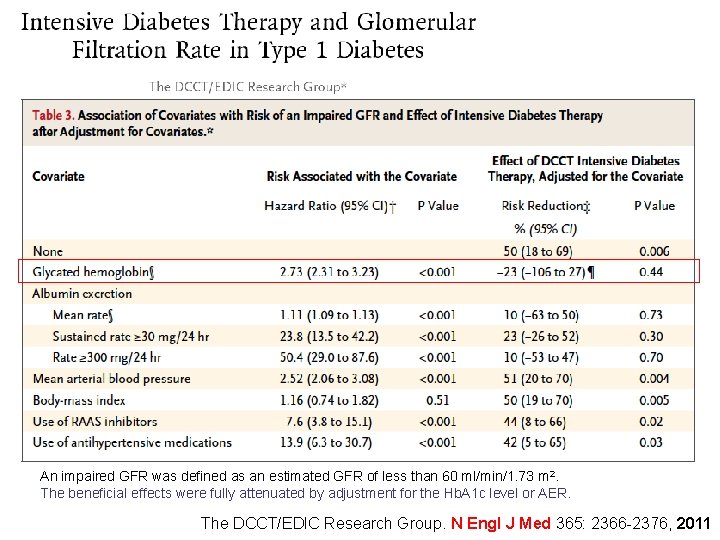
An impaired GFR was defined as an estimated GFR of less than 60 ml/min/1. 73 m 2. The beneficial effects were fully attenuated by adjustment for the Hb. A 1 c level or AER. The DCCT/EDIC Research Group. N Engl J Med 365: 2366 -2376, 2011

Renal Outcomes in patients with Type 1 Diabetes and Macroalbuminuria 159 patients with incident macroalbuminuria: follow-up 9 years e. GFR at the time of incident macroalbuminuria: 108± 20 ml/min/1. 73 m 2 Risk factors for the development of sustained e. GFR <60 among DCCT/EDIC study participants who developed macroalbuminuria De Boer IH et al. , J Am Soc Nephrol 25: 2342 -2350, 2014
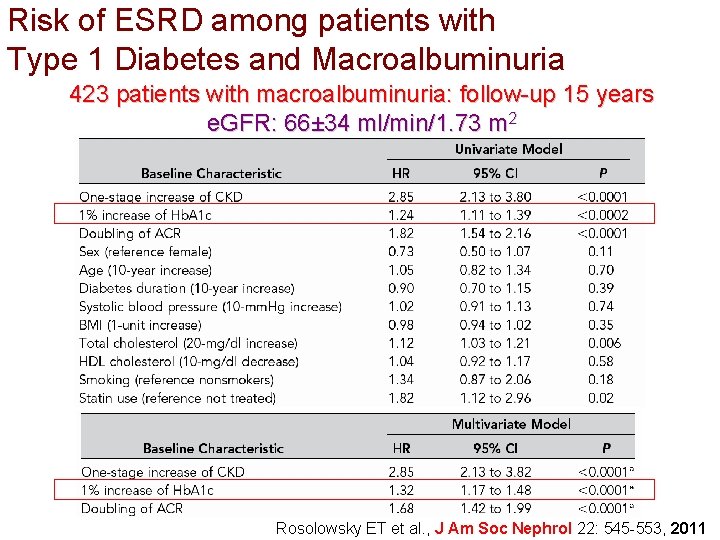
Risk of ESRD among patients with Type 1 Diabetes and Macroalbuminuria 423 patients with macroalbuminuria: follow-up 15 years e. GFR: 66± 34 ml/min/1. 73 m 2 Rosolowsky ET et al. , J Am Soc Nephrol 22: 545 -553, 2011

Risk of ESRD among patients with Type 1 Diabetes and Macroalbuminuria 592 patients with macroalbuminuria: follow-up 9. 9 years e. GFR: 52± 26 ml/min/1. 73 m 2; e. GFR <60 ml/min/1. 73 m 2: 60% Forsblom C et al. , J Am Soc Nephrol 22: 537 -544, 2011
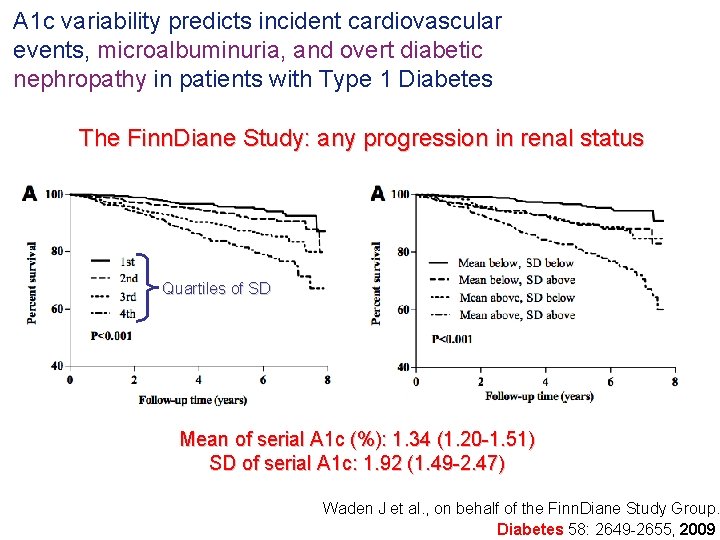
A 1 c variability predicts incident cardiovascular events, microalbuminuria, and overt diabetic nephropathy in patients with Type 1 Diabetes The Finn. Diane Study: any progression in renal status Quartiles of SD Mean of serial A 1 c (%): 1. 34 (1. 20 -1. 51) SD of serial A 1 c: 1. 92 (1. 49 -2. 47) Waden J et al. , on behalf of the Finn. Diane Study Group. Diabetes 58: 2649 -2655, 2009
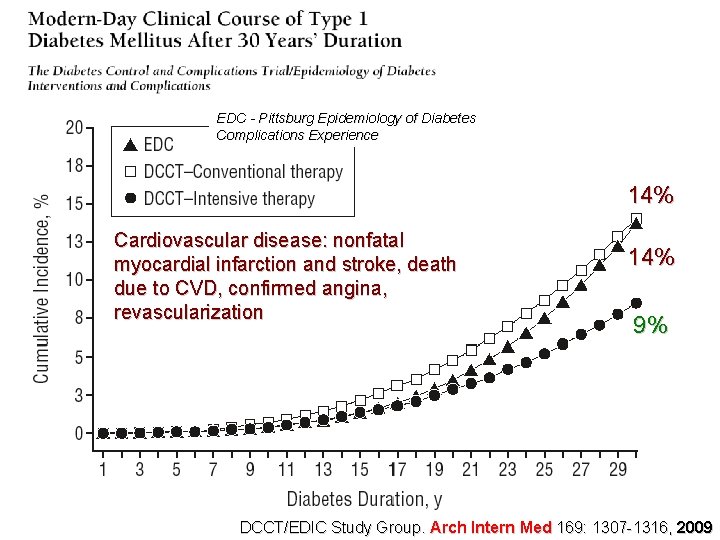
EDC - Pittsburg Epidemiology of Diabetes Complications Experience 14% Cardiovascular disease: nonfatal myocardial infarction and stroke, death due to CVD, confirmed angina, revascularization 14% 9% DCCT/EDIC Study Group. Arch Intern Med 169: 1307 -1316, 2009

Type 1 diabetes: take home messages l There is strong evidence that onset of albuminuria and impairment of GFR may be prevented or delayed in patients with type 1 diabetes, reinforcing the importance of early glycemic control. l There is also strong evidence that progression of albuminuria and risk of long-term renal outcomes (even in patients with macroalbuminuria or reduced e. GFR) may be prevented or delayed by the improved glycemic control. l In type 1 diabetes, intensive glucose management aimed to near-normal glycemia is mandatory. l This deeply affects the clinical course of both micro(renal) and macrovascular complications.
Point by point: Type 2 Diabetes ü background ü effects of intervention ü role of treatments AACE ADA/EASD
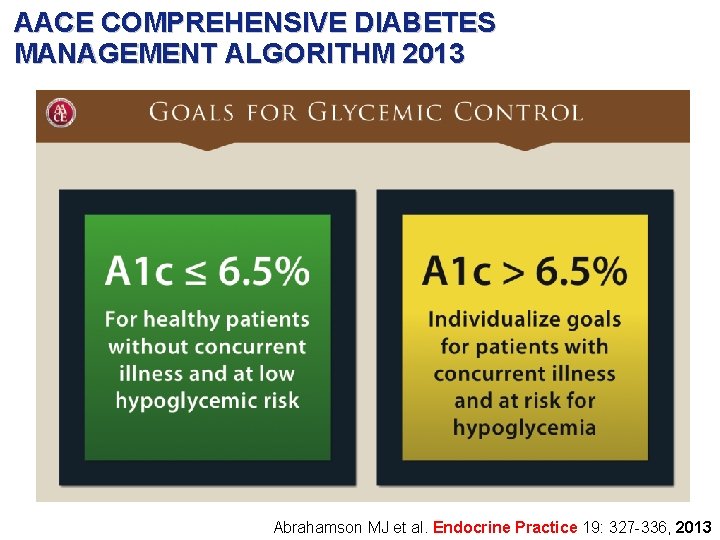
AACE COMPREHENSIVE DIABETES MANAGEMENT ALGORITHM 2013 Abrahamson MJ et al. Endocrine Practice 19: 327 -336, 2013
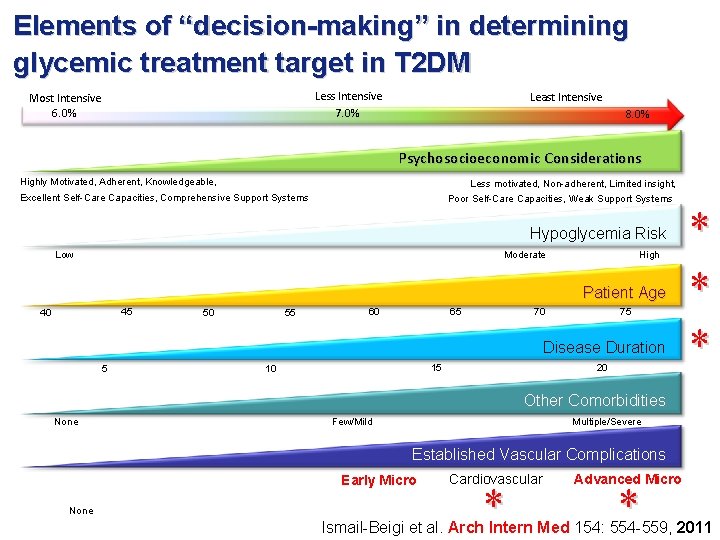
Elements of “decision-making” in determining glycemic treatment target in T 2 DM Less Intensive 7. 0% Most Intensive 6. 0% Least Intensive 8. 0% Psychosocioeconomic Considerations Highly Motivated, Adherent, Knowledgeable, Less motivated, Non-adherent, Limited insight, Excellent Self-Care Capacities, Comprehensive Support Systems Poor Self-Care Capacities, Weak Support Systems Hypoglycemia Risk Low Moderate High Patient Age 45 40 55 50 60 70 65 75 Disease Duration 5 20 15 10 * * * Other Comorbidities None Few/Mild Multiple/Severe Established Vascular Complications Early Micro None Cardiovascular * Advanced Micro * Ismail-Beigi et al. Arch Intern Med 154: 554 -559, 2011
Point by point: Type 2 Diabetes ü background ü effects of intervention ü role of treatments UKPDS PROACTIVE ADOPT ADVANCE ACCORD VADT AACE others …. ADA/EASD

Questions addressed in RCT of Type 2 diabetes treatment Question 1: Does treatment-directed lowering Hb. A 1 c (below 6. 0 to 6. 5%) reduce CV endpoints UKPDS P Long-term follow-up ACCORD P, S ADVANCE P, S VADT P, S After nearly 10 years of follow-up, patients with type 2 diabetes who had been randomly assigned to intensive glucose control for 5. 6 years had 8. 6 fewer major cardiovascular events per 1000 person-years than those assigned to standard therapy, but no improvement was seen in the rate of overall survival. N Engl J Med 2015 Jun 4; 372: 2197 -2206. P, primary prevention; S, secondary prevention
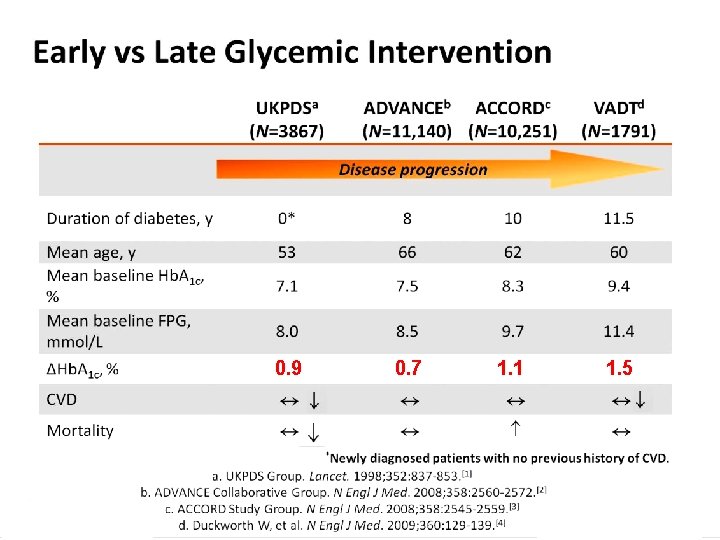
0. 9 0. 7 1. 1 1. 5
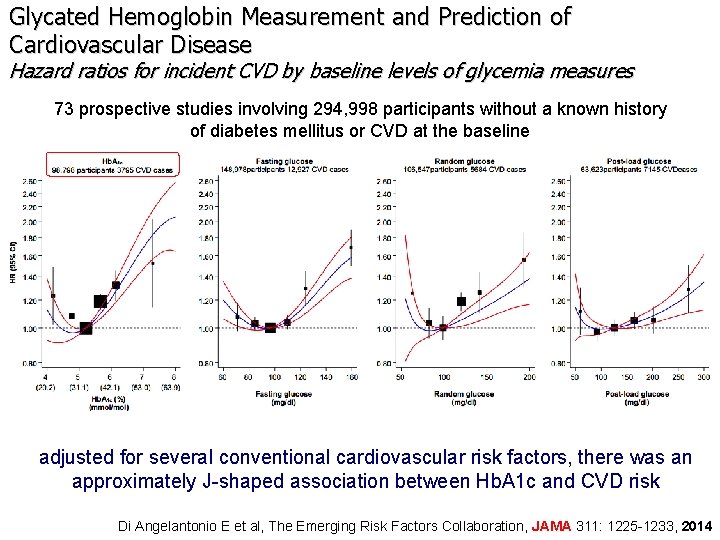
Glycated Hemoglobin Measurement and Prediction of Cardiovascular Disease Hazard ratios for incident CVD by baseline levels of glycemia measures 73 prospective studies involving 294, 998 participants without a known history of diabetes mellitus or CVD at the baseline adjusted for several conventional cardiovascular risk factors, there was an approximately J-shaped association between Hb. A 1 c and CVD risk Di Angelantonio E et al, The Emerging Risk Factors Collaboration, JAMA 311: 1225 -1233, 2014
Questions addressed in RCT of Type 2 diabetes treatment Question 1: Does treatment-directed lowering Hb. A 1 c (below 6. 0 to 6. 5%) reduce microvascular endpoints UKPDS P Long-term follow-up ACCORD P, S ADVANCE P, S VADT P, S P, primary prevention; S, secondary prevention
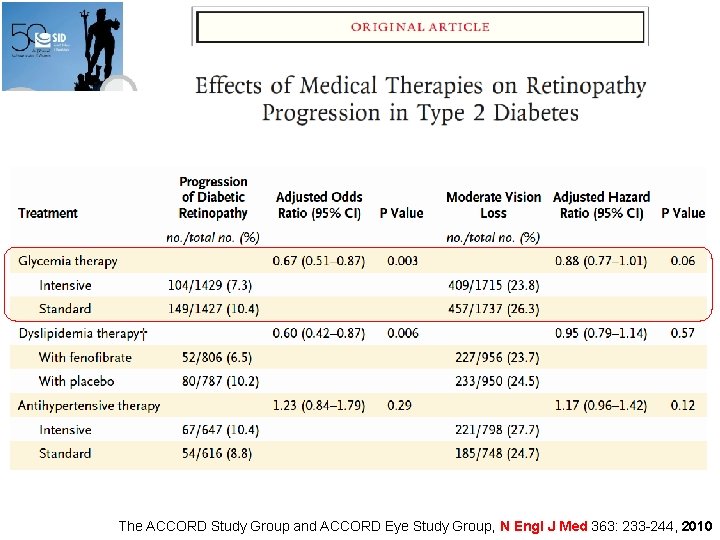
The ACCORD Study Group and ACCORD Eye Study Group, N Engl J Med 363: 233 -244, 2010
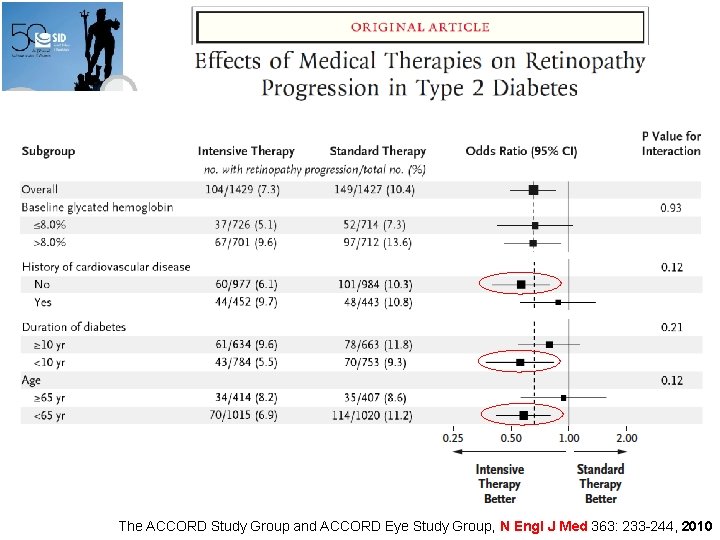
The ACCORD Study Group and ACCORD Eye Study Group, N Engl J Med 363: 233 -244, 2010
Questions addressed in RCT of Type 2 diabetes treatment Question 1: Does treatment-directed lowering Hb. A 1 c (below 6. 0 to 6. 5%) reduce renal endpoints UKPDS P Long-term follow-up ACCORD P, S ADVANCE P, S VADT P, S P, primary prevention; S, secondary prevention
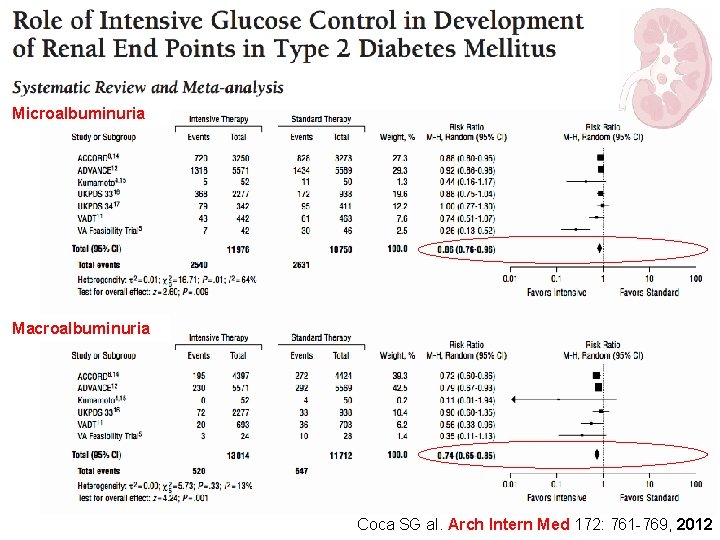
Microalbuminuria Macroalbuminuria Coca SG al. Arch Intern Med 172: 761 -769, 2012
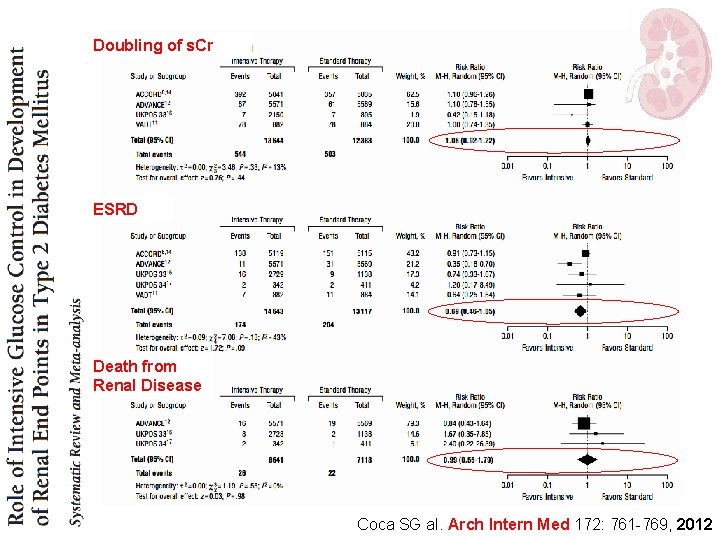
Doubling of s. Cr ESRD Death from Renal Disease Coca SG al. Arch Intern Med 172: 761 -769, 2012

Type 2 diabetes: take home message 1 Intensive glucose control reduces the risk for microalbuminuria and macroalbuminuria, but evidence is lacking that intensive glycemic control reduces the risk for significant clinical renal outcomes, such as doubling of the serum creatinine level, ESRD, or death from renal disease during the years of follow-up of the trials. Coca SG al. Arch Intern Med 172: 761 -769, 2012
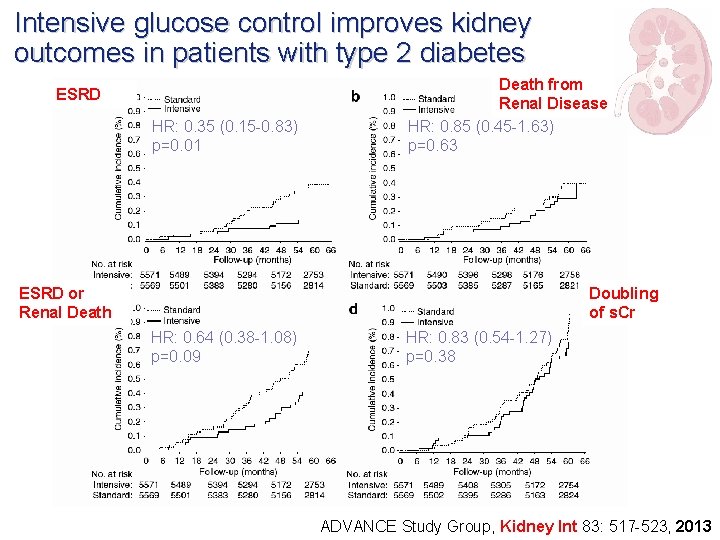
Intensive glucose control improves kidney outcomes in patients with type 2 diabetes ESRD HR: 0. 35 (0. 15 -0. 83) p=0. 01 Death from Renal Disease HR: 0. 85 (0. 45 -1. 63) p=0. 63 ESRD or Renal Death Doubling of s. Cr HR: 0. 64 (0. 38 -1. 08) p=0. 09 HR: 0. 83 (0. 54 -1. 27) p=0. 38 ADVANCE Study Group, Kidney Int 83: 517 -523, 2013
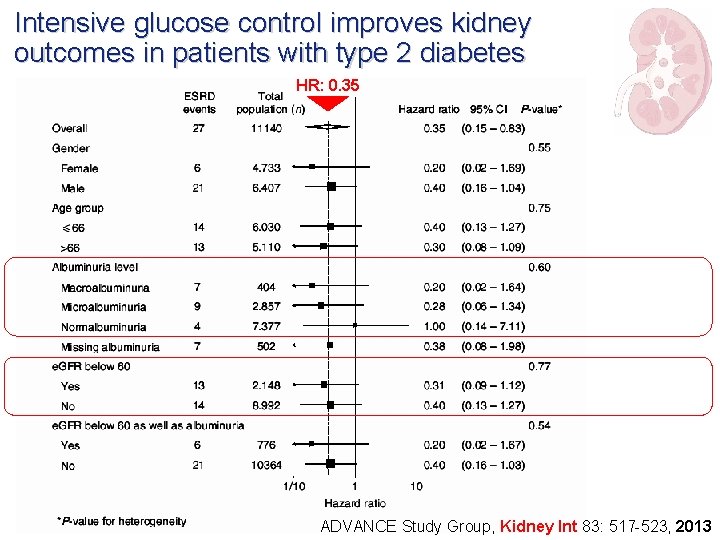
Intensive glucose control improves kidney outcomes in patients with type 2 diabetes HR: 0. 35 ADVANCE Study Group, Kidney Int 83: 517 -523, 2013
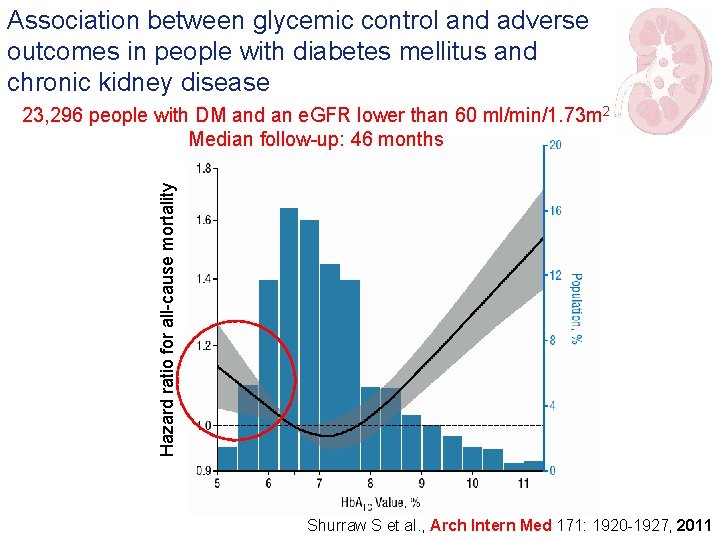
Association between glycemic control and adverse outcomes in people with diabetes mellitus and chronic kidney disease Hazard ratio for all-cause mortality 23, 296 people with DM and an e. GFR lower than 60 ml/min/1. 73 m 2 Median follow-up: 46 months Shurraw S et al. , Arch Intern Med 171: 1920 -1927, 2011

Type 2 diabetes: take home message 2 These findings suggest that appropriate and timely control of Hb. A 1 c level in people with DM and CKD may be more important than previously realized. The excess risk of kidney failure associated with a higher Hb. A 1 c was similar irrespective of kidney function. However, intensive glycemic control (Hb. A 1 c <6. 5%) may be associated with increased mortality.

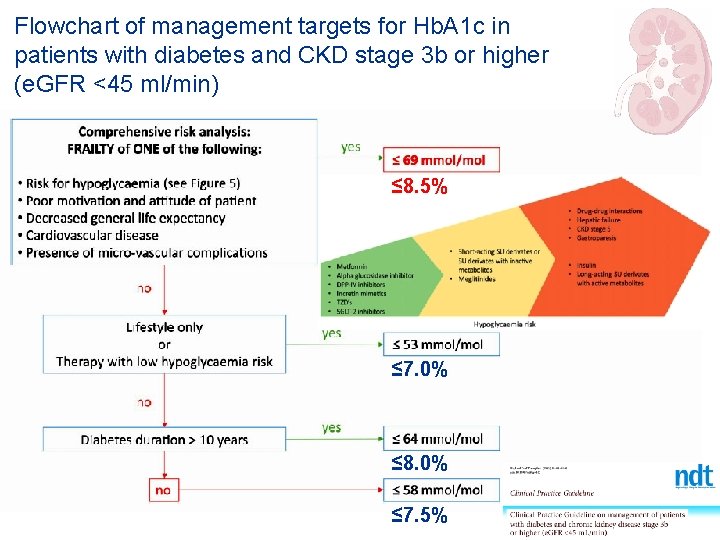
Flowchart of management targets for Hb. A 1 c in patients with diabetes and CKD stage 3 b or higher (e. GFR <45 ml/min) ≤ 8. 5% ≤ 7. 0% ≤ 8. 0% ≤ 7. 5%

Point by point: Type 2 Diabetes ü background ü effects of intervention ü role of treatments CAROLINA, LEADER EXSCEL, REWIND SUSTAIN 6, TOSCA IT SAVOR-TIMI EXAMINE TECOS ELIXA ORIGIN AACE UKPDS PROACTIVE ADOPT ADVANCE ACCORD VADT others …. ADA/EASD
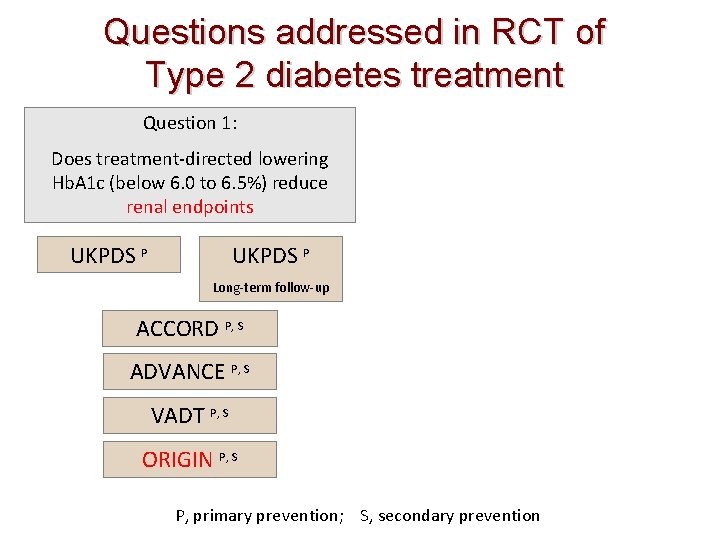
Questions addressed in RCT of Type 2 diabetes treatment Question 1: Does treatment-directed lowering Hb. A 1 c (below 6. 0 to 6. 5%) reduce renal endpoints UKPDS P Long-term follow-up ACCORD P, S ADVANCE P, S VADT P, S ORIGIN P, S P, primary prevention; S, secondary prevention
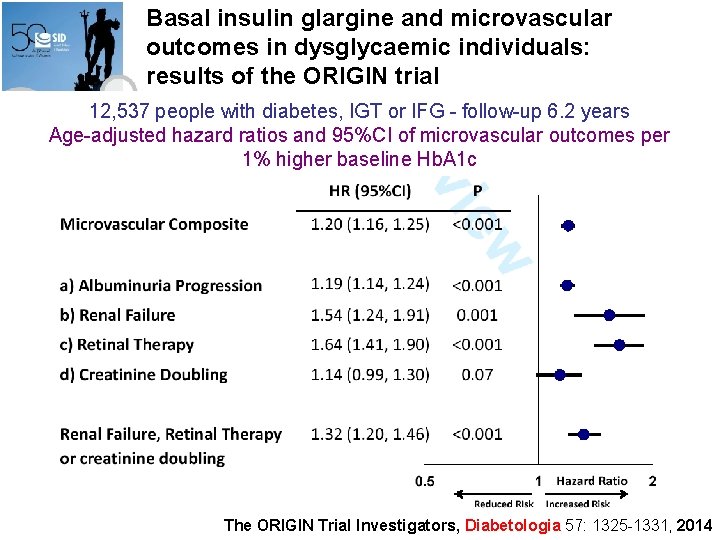
Basal insulin glargine and microvascular outcomes in dysglycaemic individuals: results of the ORIGIN trial 12, 537 people with diabetes, IGT or IFG - follow-up 6. 2 years Age-adjusted hazard ratios and 95%CI of microvascular outcomes per 1% higher baseline Hb. A 1 c The ORIGIN Trial Investigators, Diabetologia 57: 1325 -1331, 2014
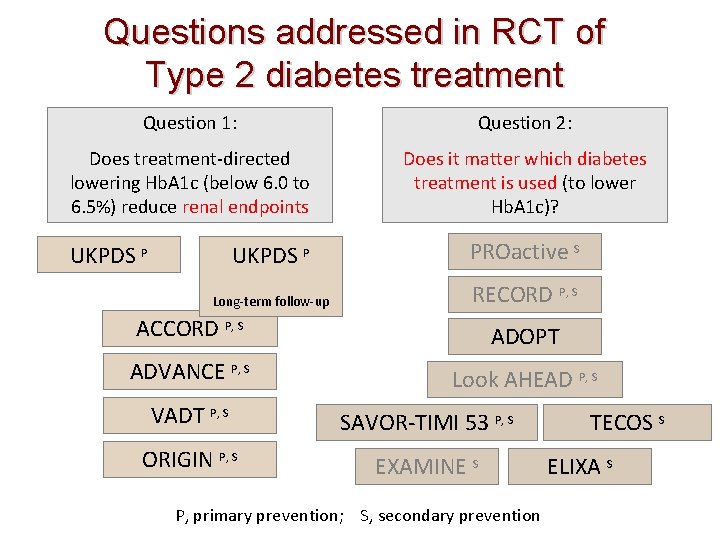
Questions addressed in RCT of Type 2 diabetes treatment Question 1: Question 2: Does treatment-directed lowering Hb. A 1 c (below 6. 0 to 6. 5%) reduce renal endpoints Does it matter which diabetes treatment is used (to lower Hb. A 1 c)? UKPDS P PROactive S Long-term follow-up RECORD P, S UKPDS P ACCORD P, S ADOPT ADVANCE P, S Look AHEAD P, S VADT P, S SAVOR-TIMI 53 P, S ORIGIN P, S EXAMINE S P, primary prevention; S, secondary prevention TECOS S ELIXA S
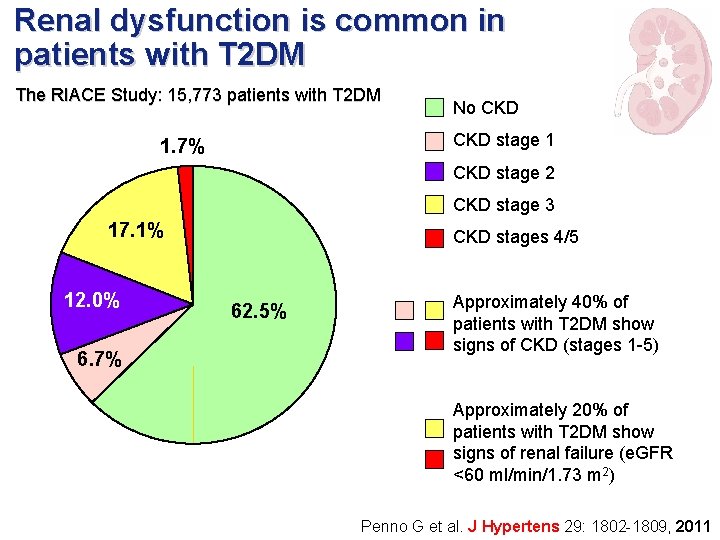
Renal dysfunction is common in patients with T 2 DM The RIACE Study: 15, 773 patients with T 2 DM No CKD stage 1 1. 7% CKD stage 2 CKD stage 3 17. 1% 12. 0% 6. 7% CKD stages 4/5 62. 5% Approximately 40% of patients with T 2 DM show signs of CKD (stages 1 -5) Approximately 20% of patients with T 2 DM show signs of renal failure (e. GFR <60 ml/min/1. 73 m 2) Penno G et al. J Hypertens 29: 1802 -1809, 2011
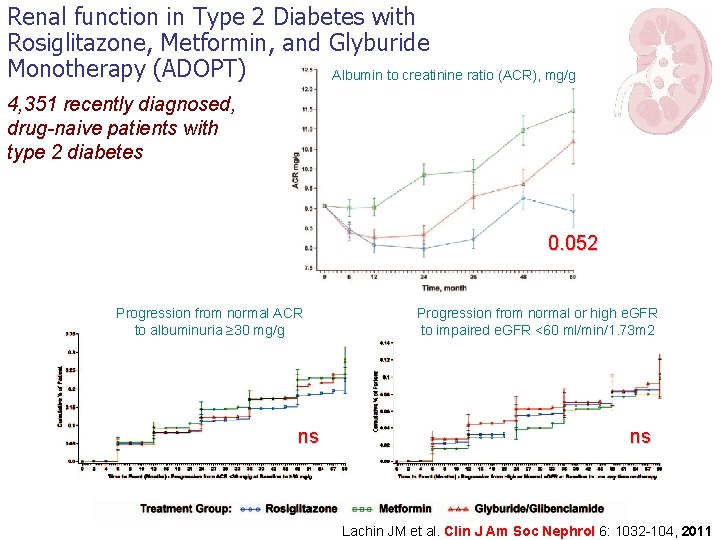
Renal function in Type 2 Diabetes with Rosiglitazone, Metformin, and Glyburide Monotherapy (ADOPT) Albumin to creatinine ratio (ACR), mg/g 4, 351 recently diagnosed, drug-naive patients with type 2 diabetes 0. 052 Progression from normal ACR to albuminuria ≥ 30 mg/g ns Progression from normal or high e. GFR to impaired e. GFR <60 ml/min/1. 73 m 2 ns Lachin JM et al. Clin J Am Soc Nephrol 6: 1032 -104, 2011
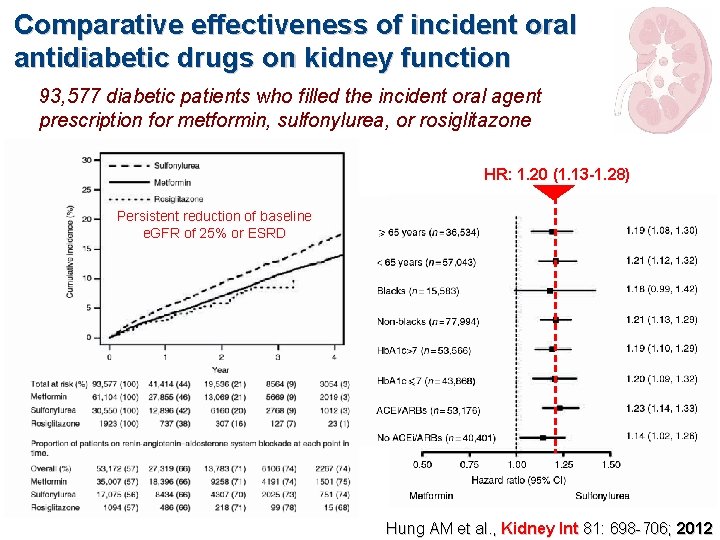
Comparative effectiveness of incident oral antidiabetic drugs on kidney function 93, 577 diabetic patients who filled the incident oral agent prescription for metformin, sulfonylurea, or rosiglitazone HR: 1. 20 (1. 13 -1. 28) Persistent reduction of baseline e. GFR of 25% or ESRD Hung AM et al. , Kidney Int 81: 698 -706; 2012

Type 2 diabetes: take home message 3 l In type 2 diabetes, targeting intensive glycaemic control reduces the risk of microvascular complications l At this time, it is difficult to draw conclusions about the comparative effectiveness of type 2 diabetes medication on microvascular outcomes, in particular on renal ones because of low quality or insufficient evidence. Hemmingsen B et al. , BMJ 343: d 6898 doi, 2011
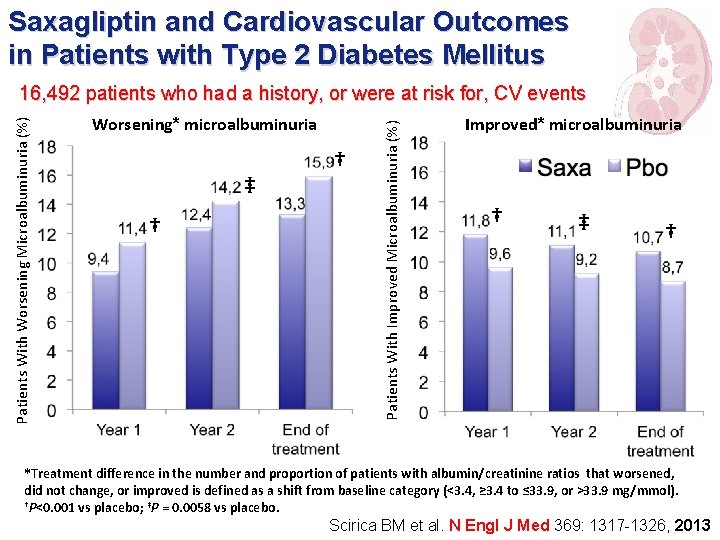
Saxagliptin and Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus Worsening* microalbuminuria ‡ † † Patients With Improved Microalbuminuria (%) Patients With Worsening Microalbuminuria (%) 16, 492 patients who had a history, or were at risk for, CV events Improved* microalbuminuria † ‡ † *Treatment difference in the number and proportion of patients with albumin/creatinine ratios that worsened, did not change, or improved is defined as a shift from baseline category (<3. 4, ≥ 3. 4 to ≤ 33. 9, or >33. 9 mg/mmol). †P<0. 001 vs placebo; ‡P = 0. 0058 vs placebo. Scirica BM et al. N Engl J Med 369: 1317 -1326, 2013
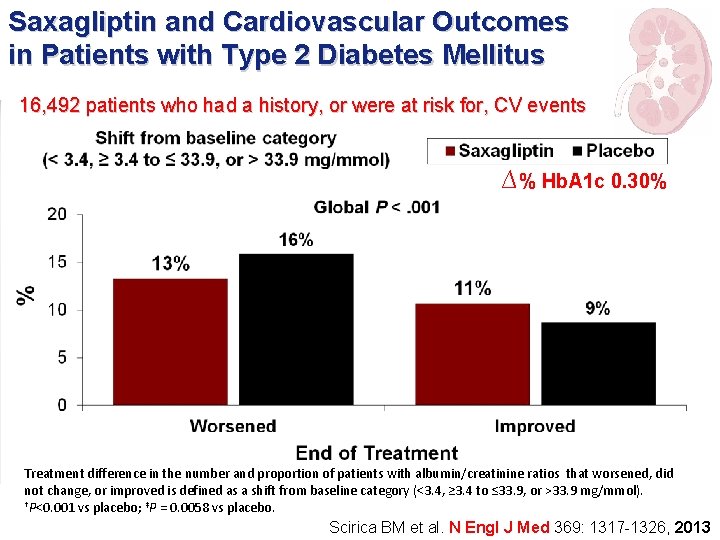
Saxagliptin and Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus 16, 492 patients who had a history, or were at risk for, CV events ∆% Hb. A 1 c 0. 30% Treatment difference in the number and proportion of patients with albumin/creatinine ratios that worsened, did not change, or improved is defined as a shift from baseline category (<3. 4, ≥ 3. 4 to ≤ 33. 9, or >33. 9 mg/mmol). †P<0. 001 vs placebo; ‡P = 0. 0058 vs placebo. Scirica BM et al. N Engl J Med 369: 1317 -1326, 2013
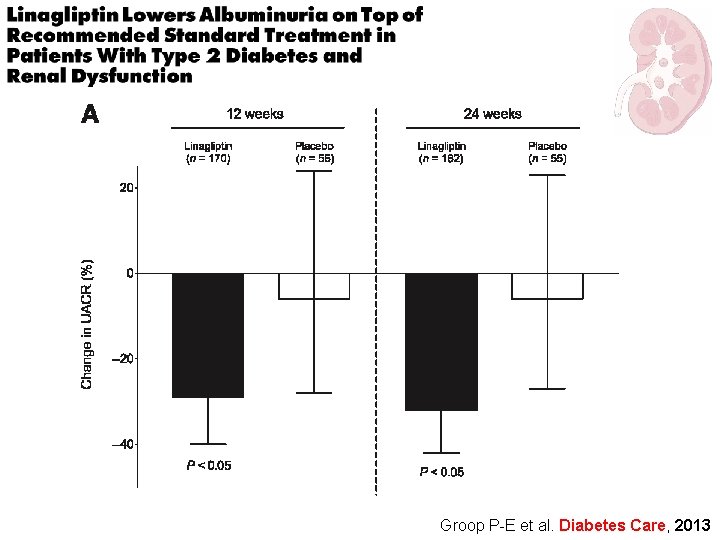
Groop P-E et al. Diabetes Care, 2013
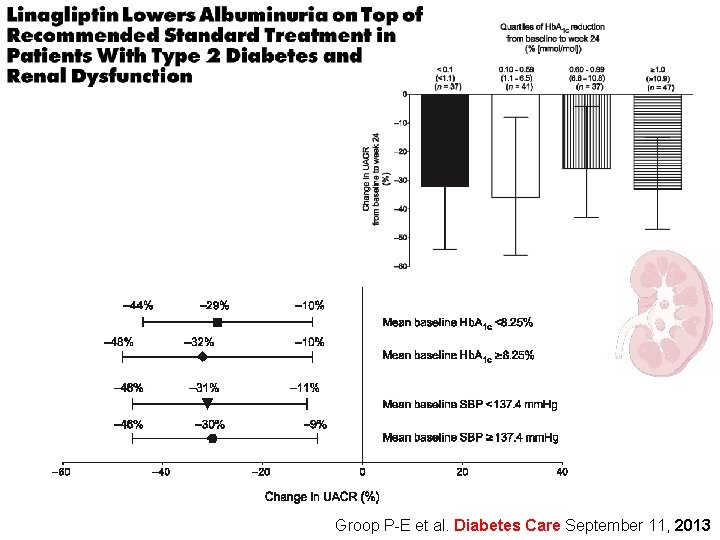
Groop P-E et al. Diabetes Care September 11, 2013
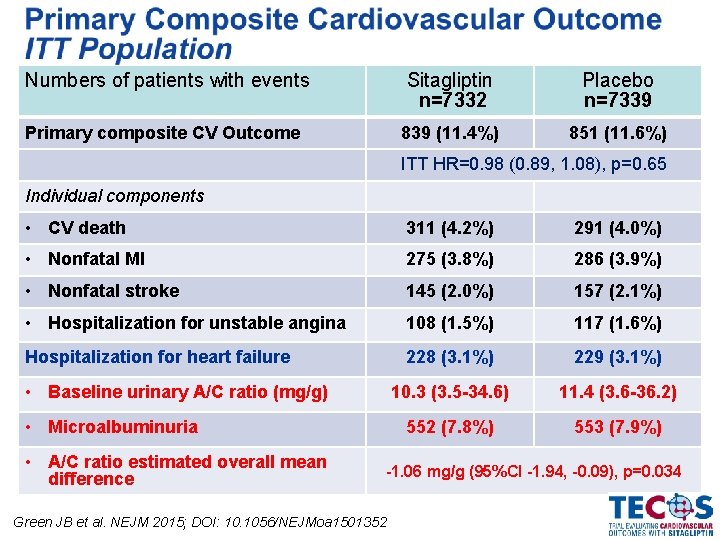
Numbers of patients with events Sitagliptin n=7332 Placebo n=7339 Primary composite CV Outcome 839 (11. 4%) 851 (11. 6%) ITT HR=0. 98 (0. 89, 1. 08), p=0. 65 Individual components • CV death 311 (4. 2%) 291 (4. 0%) • Nonfatal MI 275 (3. 8%) 286 (3. 9%) • Nonfatal stroke 145 (2. 0%) 157 (2. 1%) • Hospitalization for unstable angina 108 (1. 5%) 117 (1. 6%) Hospitalization for heart failure 228 (3. 1%) 229 (3. 1%) 10. 3 (3. 5 -34. 6) 11. 4 (3. 6 -36. 2) 552 (7. 8%) 553 (7. 9%) • Baseline urinary A/C ratio (mg/g) • Microalbuminuria • A/C ratio estimated overall mean difference -1. 06 mg/g (95%CI -1. 94, -0. 09), p=0. 034 Green JB et al. NEJM 2015; DOI: 10. 1056/NEJMoa 1501352
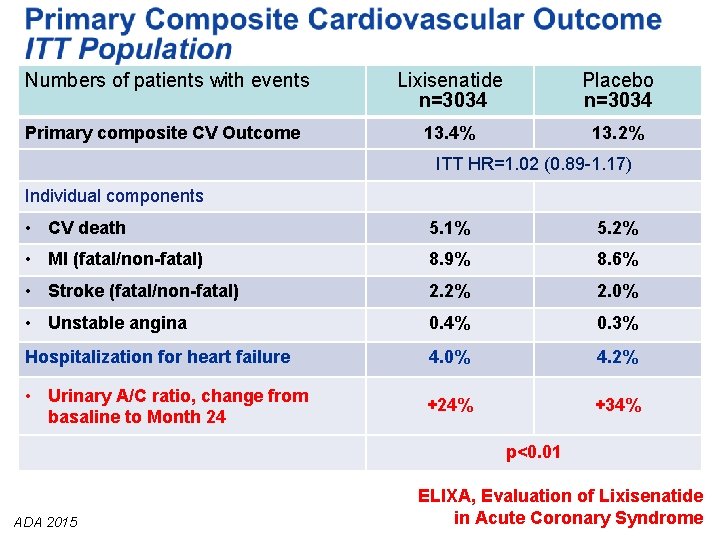
Numbers of patients with events Lixisenatide n=3034 Placebo n=3034 Primary composite CV Outcome 13. 4% 13. 2% ITT HR=1. 02 (0. 89 -1. 17) Individual components • CV death 5. 1% 5. 2% • MI (fatal/non-fatal) 8. 9% 8. 6% • Stroke (fatal/non-fatal) 2. 2% 2. 0% • Unstable angina 0. 4% 0. 3% Hospitalization for heart failure 4. 0% 4. 2% • Urinary A/C ratio, change from basaline to Month 24 +24% +34% p<0. 01 ADA 2015 ELIXA, Evaluation of Lixisenatide in Acute Coronary Syndrome
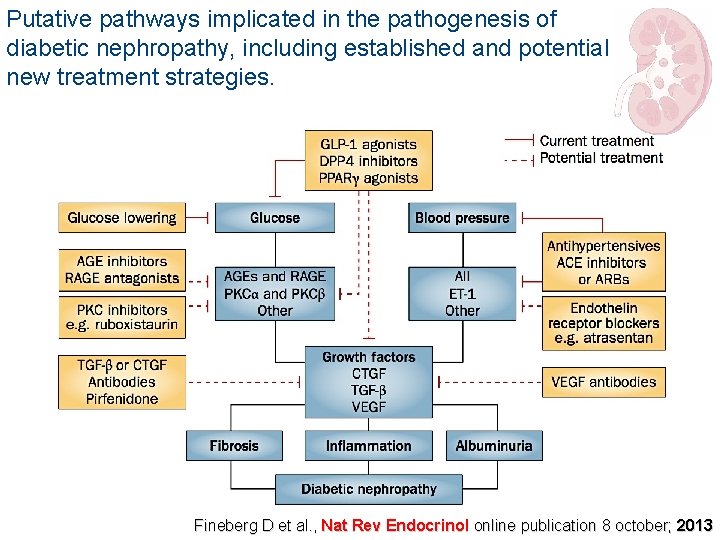
Putative pathways implicated in the pathogenesis of diabetic nephropathy, including established and potential new treatment strategies. Fineberg D et al. , Nat Rev Endocrinol online publication 8 october; 2013

Managing diabetic patients with moderate or severe renal impairment …. . Russo E et al. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 6: 1 -10, 2013

Scheen AJ. Expert Opin Drug Metab Toxicol 9: 529 -550, 2013

Scheen AJ. Expert Opin Drug Metab Toxicol 9: 529 -550, 2013

Managing diabetic patients with moderate or severe renal impairment …. . Russo E et al. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 6: 1 -10, 2013 (modified)
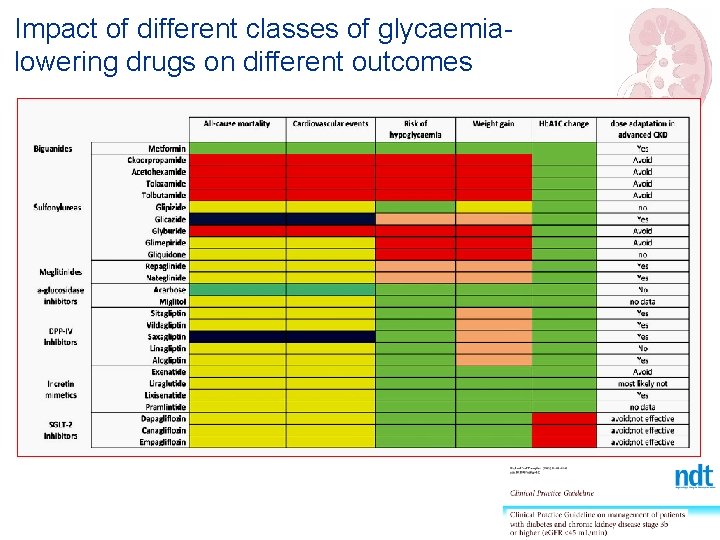
Impact of different classes of glycaemialowering drugs on different outcomes

DPP-4 provide reliable Hb. A 1 c reductions across different stages of declining renal function Adjusted mean change in Hb. A 1 c (%) from baseline at week 24 Change from baseline Hb. A 1 c by degree of renal impairment Placebo corrected, adjusted mean change from baseline at 24 weeks of treatment e. GFR No renal impairment ≥ 90 m. L/min -0. 01 Mild renal impairment 60 to <90 m. L/min -0. 06 Moderate renal impairment 30 to <60 m. L/min -0. 13 -0. 61 -0. 60 -0. 57 -0. 63 -0. 69 -0. 70 n= 406 1006 272 715 29 80 Mean baseline Hb. A 1 c (%) 8. 2 8. 1 8. 2 8. 3 p-value between group difference (versus placebo) <0. 0001 Pre-specified sub-group analysis on pooled data from 4 pivotal phase III randomized placebo-controlled trials: treatment in monotherapy, add-on to metformin + SU, initial combination with pioglitazone. Placebo Linagliptin placebo-corrected 0. 0023 Cooper, ADA 2011 Poster 1068
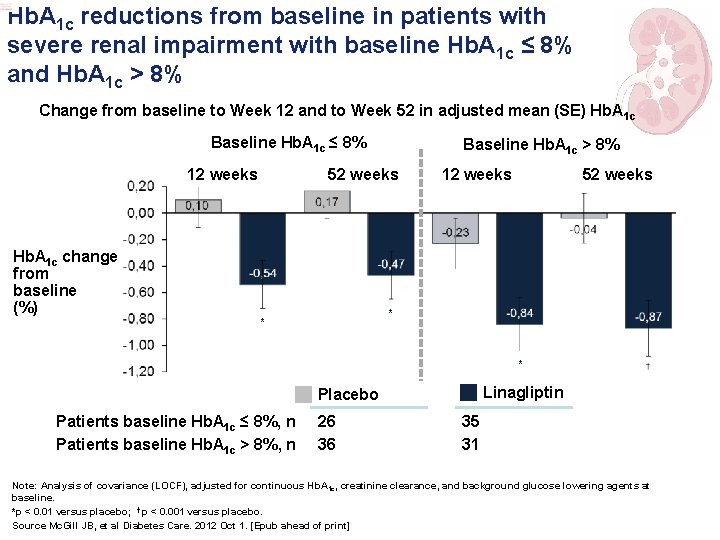
Hb. A 1 c reductions from baseline in patients with severe renal impairment with baseline Hb. A 1 c ≤ 8% and Hb. A 1 c > 8% Change from baseline to Week 12 and to Week 52 in adjusted mean (SE) Hb. A 1 c Baseline Hb. A 1 c ≤ 8% 12 weeks Hb. A 1 c change from baseline (%) Baseline Hb. A 1 c > 8% 52 weeks 12 weeks 52 weeks * * – 0. 7*** * Linagliptin Placebo Patients baseline Hb. A 1 c ≤ 8%, n Patients baseline Hb. A 1 c > 8%, n 26 36 35 31 Note: Analysis of covariance (LOCF), adjusted for continuous Hb. A 1 c, creatinine clearance, and background glucose lowering agents at baseline. *p < 0. 01 versus placebo; † p < 0. 001 versus placebo. Source Mc. Gill JB, et al Diabetes Care. 2012 Oct 1. [Epub ahead of print]
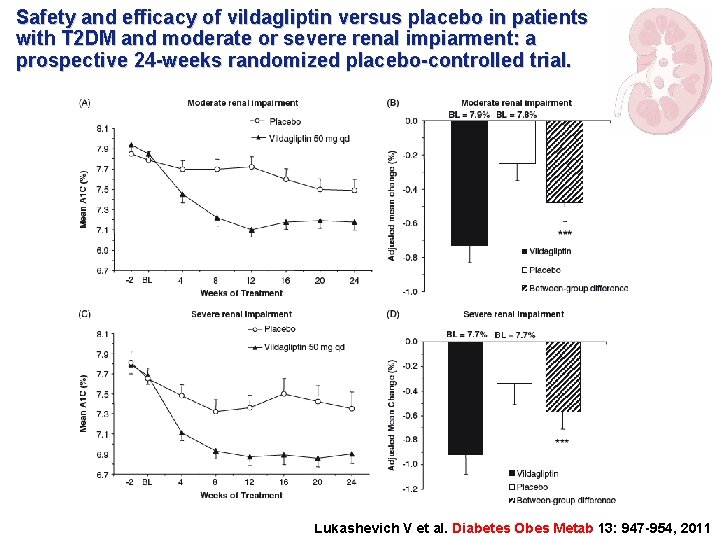
Safety and efficacy of vildagliptin versus placebo in patients with T 2 DM and moderate or severe renal impiarment: a prospective 24 -weeks randomized placebo-controlled trial. Lukashevich V et al. Diabetes Obes Metab 13: 947 -954, 2011
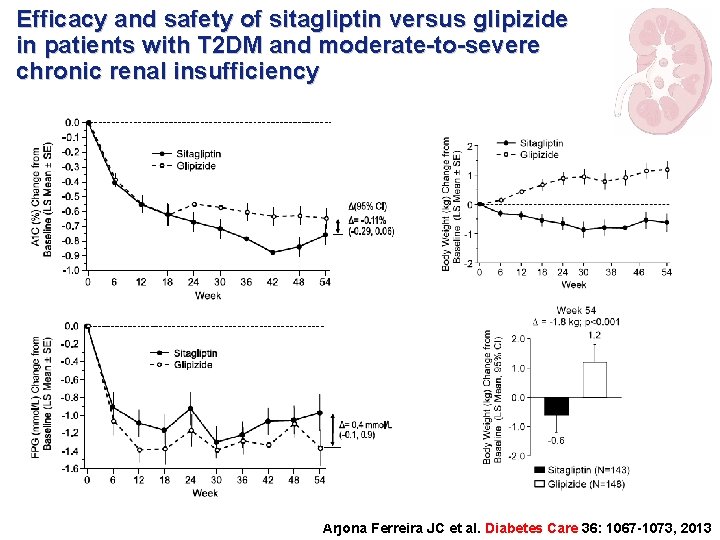
Efficacy and safety of sitagliptin versus glipizide in patients with T 2 DM and moderate-to-severe chronic renal insufficiency Arjona Ferreira JC et al. Diabetes Care 36: 1067 -1073, 2013
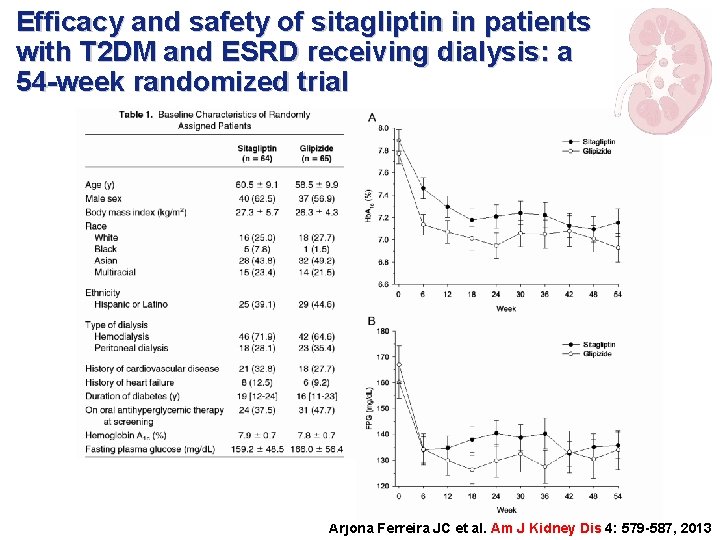
Efficacy and safety of sitagliptin in patients with T 2 DM and ESRD receiving dialysis: a 54 -week randomized trial Arjona Ferreira JC et al. Am J Kidney Dis 4: 579 -587, 2013
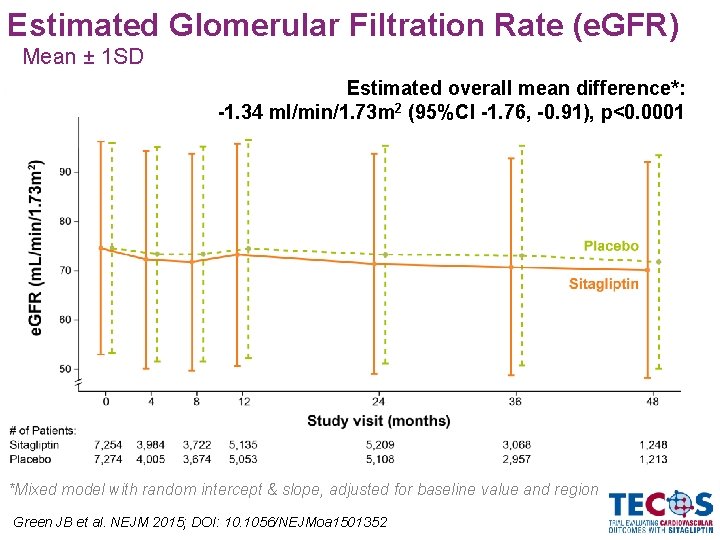
Estimated Glomerular Filtration Rate (e. GFR) Mean ± 1 SD Estimated overall mean difference*: -1. 34 ml/min/1. 73 m 2 (95%CI -1. 76, -0. 91), p<0. 0001 *Mixed model with random intercept & slope, adjusted for baseline value and region Green JB et al. NEJM 2015; DOI: 10. 1056/NEJMoa 1501352

Type 2 diabetes: take home message 4 l In summary, DPP-4 i are effective and well tolerated treatment option for patients with type 2 diabetes mellitus, including those with any degree of renal impairment, when they are used in accordance with the product labelling. l DPP-4 i are a suitable treatment option for patients with advanced type 2 diabetes and impaired renal function who require insulin therapy and present a serious therapeutic challenge in clinical practice. l Newer drugs that may improve the currently very limited treatment options for patients with type 2 diabetes and renal impairment include the dipeptidyl peptidase-4 inhibitors.

Thank you for your attention
- Slides: 63