The Transition Metals the Lanthanides and the Antinides





































- Slides: 55

The Transition Metals, the Lanthanides and the Antinides

Transition Elements 3 The Metals in the Middle • Groups 3 -12 are called the transition elements. • All of them are metals. • Across any period from Group 3 through 12, the properties of the elements change less noticeably than they do across a period of representative elements. • Most transition elements are found combined with other elements in ores.

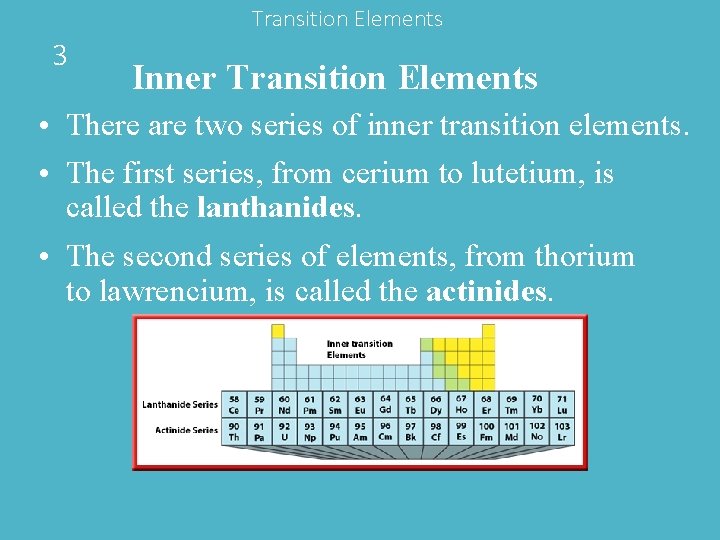
Transition Elements 3 Inner Transition Elements • There are two series of inner transition elements. • The first series, from cerium to lutetium, is called the lanthanides. • The second series of elements, from thorium to lawrencium, is called the actinides.

Transition Elements 3 The Lanthanides • They are soft, malleable, shiny metals with high conductivity. • They are mixed with more common metals to produce alloys, which are a mixture of metal with one other element, usually another metal. • Despite the name rare earth, the lanthanides are not as rare as originally thought. • Cerium makes up 50 percent of an alloy called misch (MIHSH) metal. • Flints in lighters are made from misch metal.

Transition Elements 3 The Actinides • All the actinides are radioactive. • The nuclei of atoms of radioactive elements are unstable and decay to form other elements. • Thorium, protactinium, and uranium are the only actinides that now are found naturally on Earth. • Uranium is found in Earth’s crust because its half-life is long— 4. 5 billion years.

Transition Elements 3 The Actinides • All other actinides are synthetic elements. • Synthetic elements are made in laboratories and nuclear reactors. • Plutonium is used as a fuel in nuclear power plants. • Americium is used in some home smoke detectors. • Californium-252 is used to kill cancer cells.

Synthetic & Radioactive Elements Chemistry

Synthetic Definition? Not of natural origin; prepared or made artificially

Synthetic Elements - Metals • Elements with atomic numbers higher than 92 are sometimes described as synthetic elements because they are not found naturally on Earth. • Instead, elements that follow uranium are made – or synthesized – when nuclear particles are forced to crash into one another. • To make even heavier elements (with atomic numbers above 95), scientists use powerful machines called particle accelerators which move atomic nuclei faster and faster until they reach very high speeds and then crash into each other.

Which Elements are Synthetic? There are currently 28 synthetic elements

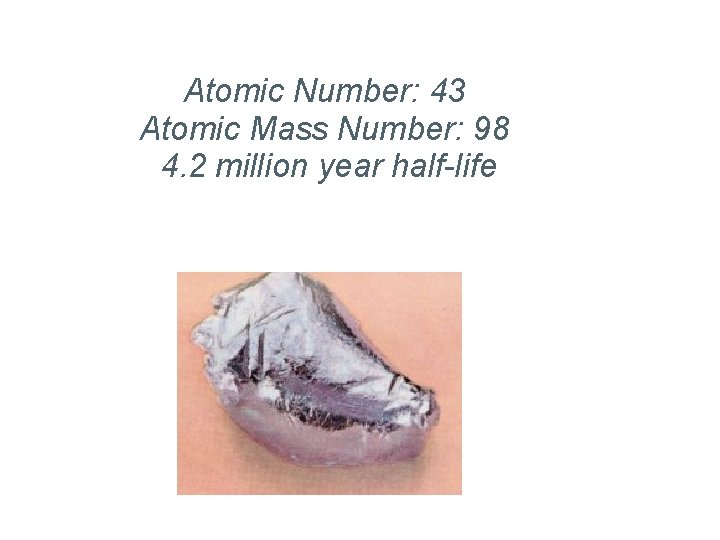
Technetium Atomic Number: 43 Atomic Mass Number: 98 4. 2 million year half-life

Some other Synthetic Elements are. . . Promethium (61) Curium (96) Hassium (108)

Uses of Plutonium • explosive in nuclear weapons • fuel in nuclear power plants • nuclear batteries

Why Make Synthetic Elements? • Many have no practical use • Made to study properties

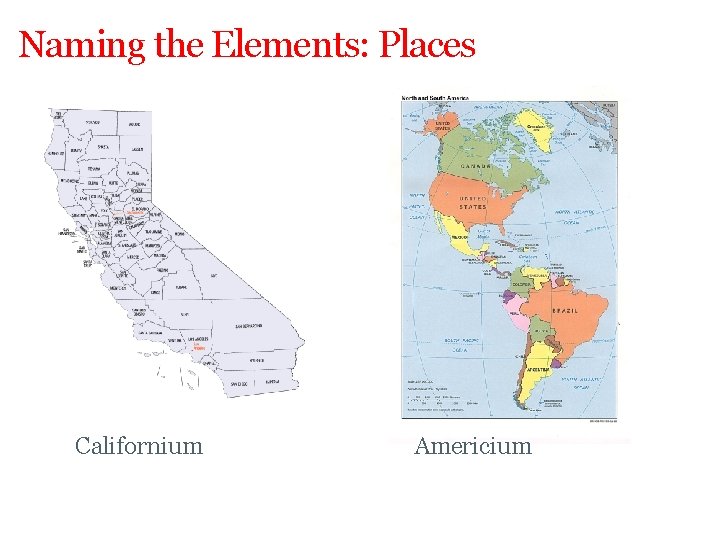
Naming the Elements: Places Californium Americium

Naming the Elements: Famous Scientists Einsteinium Curium Seaborgium Element names are often controversial: • • International Union of Pure and Applied Chemistry tried to tell team that no elements can be named after living scientists Also can be controversy over who found the element because whoever found it gets to name it

Synthetic Elements are made in a Proton Accelerator




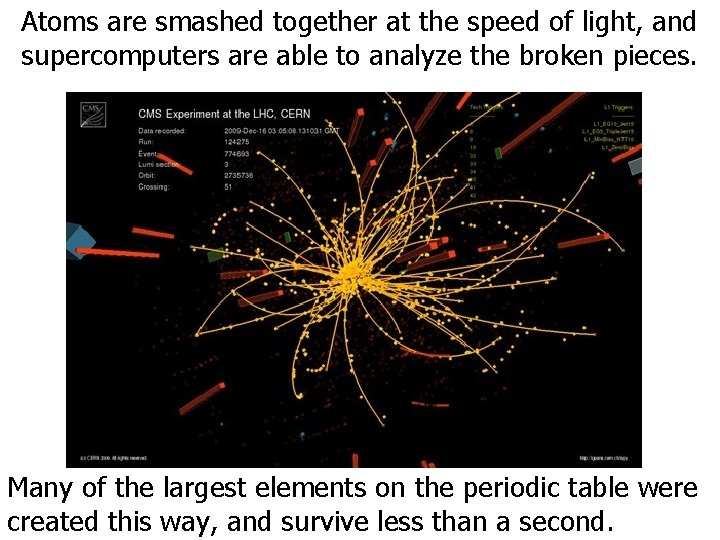
Atoms are smashed together at the speed of light, and supercomputers are able to analyze the broken pieces. Many of the largest elements on the periodic table were created this way, and survive less than a second.

Radioactive Elements Unstable elements whose nucleus breaks down and gives off particles, radiation, and energy

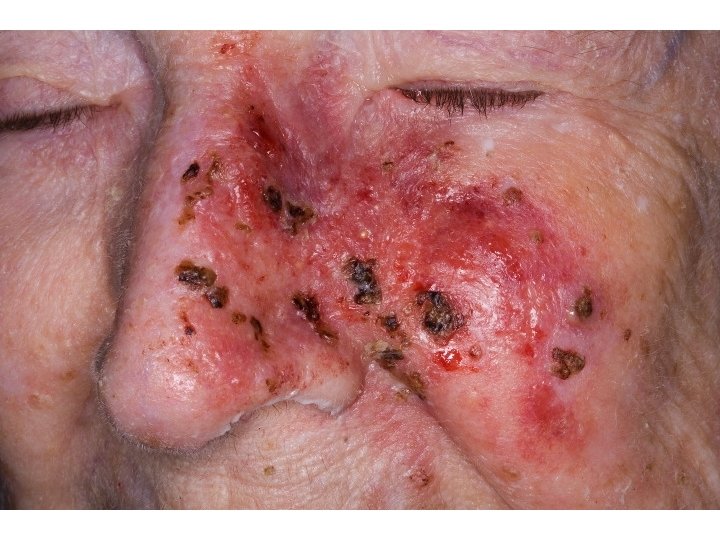
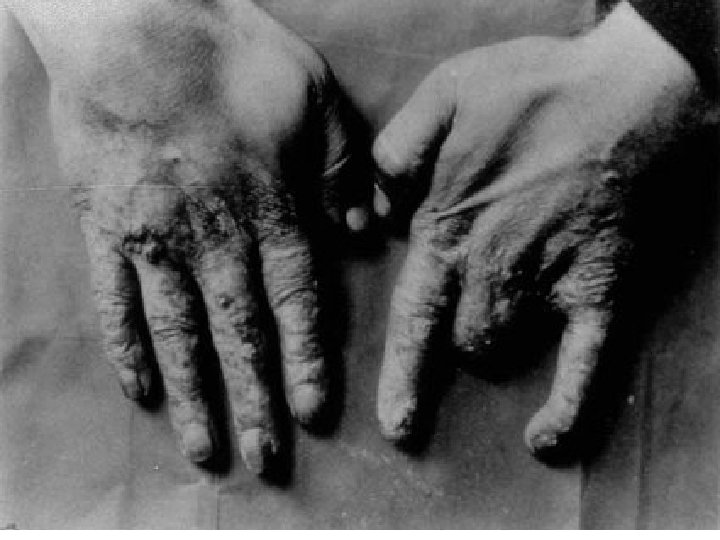
Radiation is invisible energy or particles released by radioactive elements that can damage biological tissue.

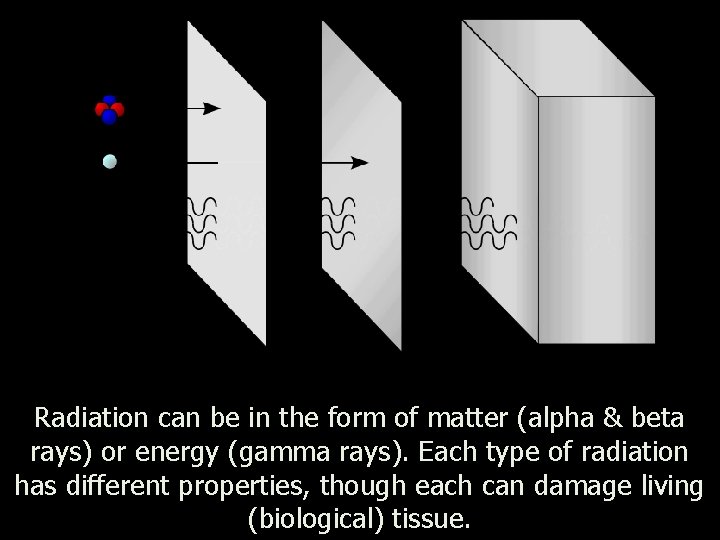
Radiation can be in the form of matter (alpha & beta rays) or energy (gamma rays). Each type of radiation has different properties, though each can damage living (biological) tissue.

What happens when living things are exposed to large amounts of radiation?

Types of Radiation Exposure Acute Short-term exposure – effect on biological tissue depends on the intensity of the radiation Chronic Long-term exposure of a small amount of radiation

Acute Effects Occur after short-term exposure Ex. Bombs, medical doses, industrial accidents, etc Effects include: Cell death Lethal cell DNA mutations Nonlethal cell DNA mutations (cancer) Mutations to reproductive cells resulting in birth defects


Chronic Exposure Long-term exposure, often the result of living or working in an area with low-levels of radiation Ex. Laying out in the sun everyday. Effects include: Lethal and non-Lethal DNA mutation (cancer) Mutations to reproductive cells resulting in birth defects


Why are some elements radioactive?

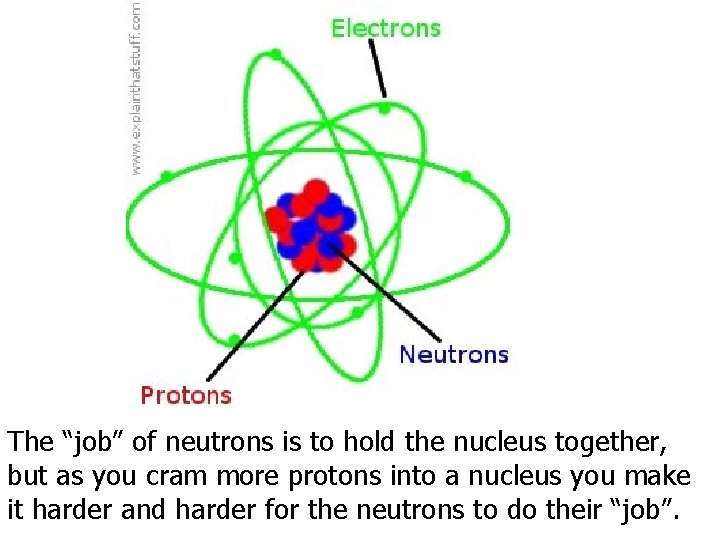
The “job” of neutrons is to hold the nucleus together, but as you cram more protons into a nucleus you make it harder and harder for the neutrons to do their “job”.

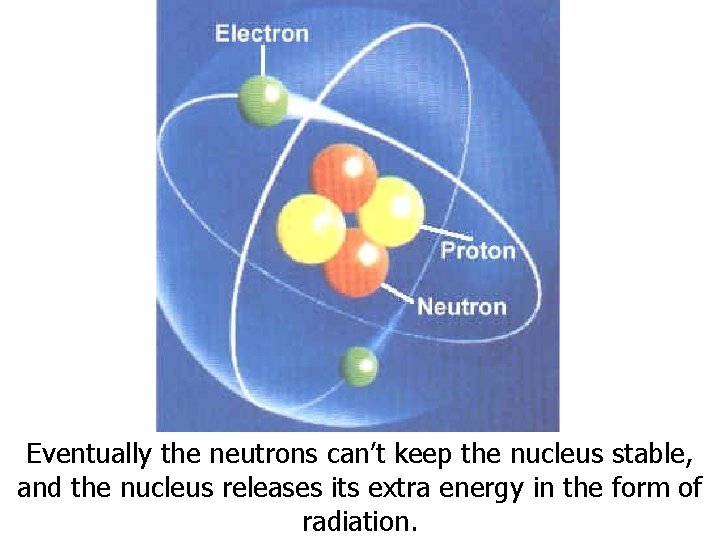
Eventually the neutrons can’t keep the nucleus stable, and the nucleus releases its extra energy in the form of radiation.

So is radiation all bad? ? No, it has a lot of uses!

Seeing in metal stuff


Radiation is either electromagnetic waves or particles of various energy. Irradiation is the process by which something is exposed to radiation. Radiation occurs when an object emits electromagnetic energy. Irradiation occurs when that energy is absorbed by another object.

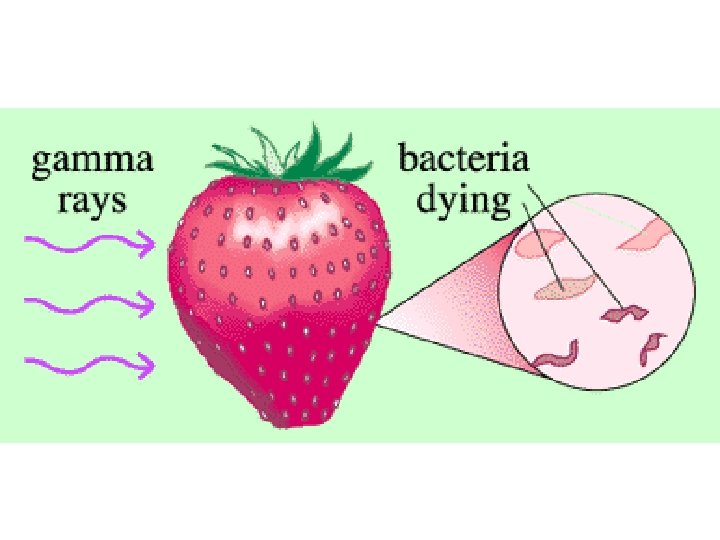
Irradiation This treatment is used to improve food safety by extending product shelf-life (preservation), reducing the risk of foodborne illness, delaying or eliminating sprouting or ripening, by sterilization of foods, and as a means of controlling insects and invasive pests





Nuclear Power

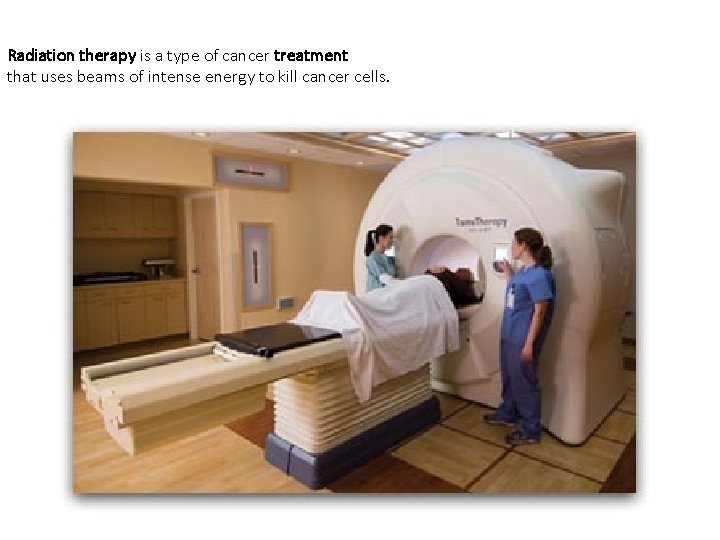
Radiation therapy is a type of cancer treatment that uses beams of intense energy to kill cancer cells.

Nuclear Bombs

Examining internal structures





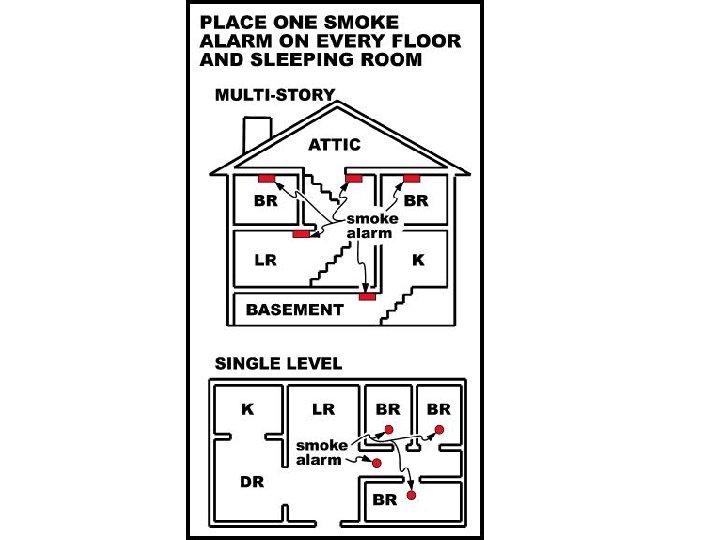
Smoke Detectors If smoke blocks the path of radiation between two points, it goes off



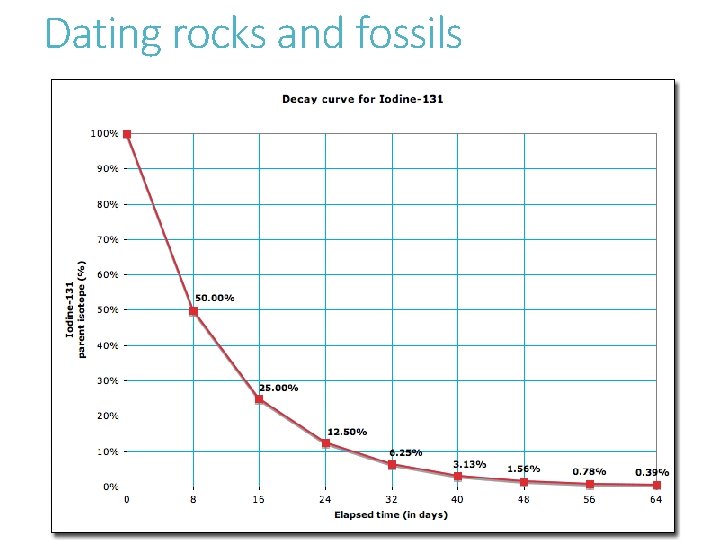
Dating rocks and fossils

The End!