The Transition Metals General Properties of Transition Metals













- Slides: 15

The Transition Metals

General Properties of Transition Metals Do Now: What do you know already about the transition metals?

What do we know about the d-block? Transition metals • The outer e- are in the d sub-shell. • “Transition metals are d-block elements that form one or more stable ions with incompletely filled d-orbitals” • Why are Sc and Zn d-block elements, but NOT transition elements? • Sc 1 s 22 p 63 s 23 p 63 d 14 s 2 Zn 1 s 22 p 63 s 23 p 63 d 104 s 2

What are Transition Metals? Transition metals form at least one stable ion with a partially filled d sub-shell. • Top row transition metals: Ti – Cu • According to the definition. . . • Sc is not a transition metal (Sc and Sc 3+) • Zn is not a transition metal (Zn and Zn 2+)

Electronic Configurations • Task: Complete the electronic configurations for the following atoms and ions (Use shorthand notation e. g. , [Ar]. . . ) Remem ber! 4 s fills a nd emp before 3 ties d Fe [Ar] 4 s 2 3 d 6 Cu [Ar] 4 s 1 3 d 10 Fe 3+ [Ar] 3 d 5 Cu+ [Ar] 3 d 10 Sc [Ar] 4 s 2 3 d 1 Cu 2+ [Ar] 3 d 9 Sc 3+ [Ar] Zn [Ar] 4 s 2 3 d 10 V [Ar] 4 s 2 3 d 3 Zn 2+ [Ar] 3 d 10 V 2+ [Ar] 3 d 3 Cr [Ar] 4 s 1 3 d 5

Characteristics of TM • They are all metals, so your task is to list their characteristics… • Hard TM tend to be stro nger , more den Strong se and le ss r eactive th Shiny an the m etals in Groups High mpt and bpt 1 and 2. Good conductors of heat and electricity Act as catalysts Form coloured ions and compounds Form ions with different oxidation states Form ions with incompletely-filled d-orbitals • •

Uses of TM • Their physical properties together with their fairly low chemical reactivity make the TM very useful • Fe (and alloy steel) for vehicles and to reinforce concrete • Cu for water pipes • Ti for jet engine parts that need to withstand high temps

1) They form coloured ions • The majority of TM complexes are coloured • e. g. , Cu 2+ (aq) is blue - Cu. SO 4

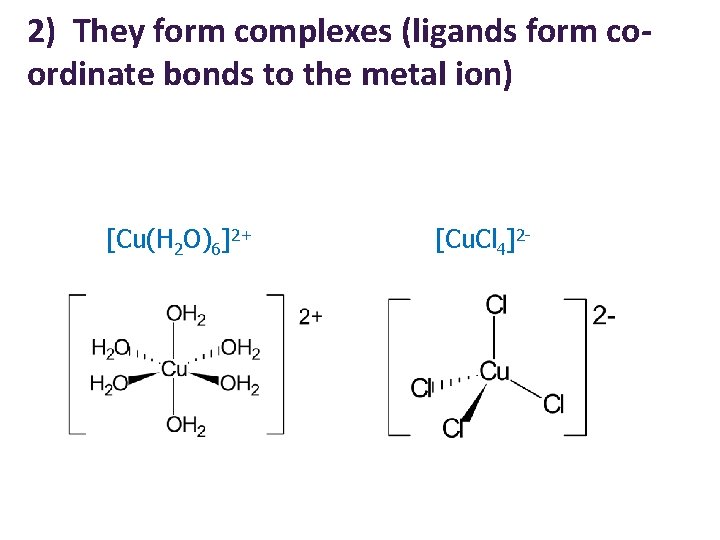
2) They form complexes (ligands form coordinate bonds to the metal ion) [Cu(H 2 O)6]2+ [Cu. Cl 4]2 -

3) They exhibit variable oxidation states • Often have more than one oxidation state in their compounds, e. g. , Cu(I) and Cu(II) • So they can take part in many redox reactions

4) They show catalytic activity Ni Margarine production V 2 O 5 Contact Process making SO 3 for H 2 SO 4 Fe Haber process to make NH 3 Pt, Pd Catalytic converters Mn. O 2 Decomposition of H 2 O 2

Oxidation States What is the oxidation of the TM in the following compounds? 1. 2. 3. 4. 5. 6. 7. 8. Ti. O 3 Cu. SO 4 KMn. O 4 Co. SO 4 V 2 O 5 Mn. Cl 2 Ti. Cl 4 K 2 Cr. O 4

Oxidation States What is the oxidation of the TM in the following compounds? 1. 2. 3. 4. 5. 6. 7. 8. Ti 2 O 3 Cu. SO 4 KMn. O 4 Co. SO 4 V 2 O 5 Mn. Cl 2 Ti. Cl 4 K 2 Cr. O 4 +3 +2 +7 +2 +5 +2 +4 +6

Quick Check: True or False 1. 2. 3. 4. 5. TM are very useful because they are generally more reactive than Group 1 and Group 2 metals. The electronic configuration of Cr is [Ar] 4 s 2 3 d 4 Zn is not a TM The oxidation state of vanadium is VCl 3 is +3 The 4 chemical properties of TM are: Catalysis, formation of coloured ion, formation of complex ions, formation of hydroxides in water

Quick Check: True or False 1. 2. 3. 4. 5. TM are very useful because they are generally more reactive than Group 1 and Group 2 metals. The electronic configuration of Cr is [Ar] 4 s 2 3 d 4 Zn is not a TM The oxidation state of vanadium is VCl 3 is +3 The 4 chemical properties of TM are: Catalysis, formation of coloured ion, formation of complex ions, formation of hydroxides in water