Metals Alkali Alkaline Transition Metals in Mixed Group














- Slides: 17

Metals!! Alkali, Alkaline, Transition, Metals in Mixed Group, Lanthanides, and Actinides


Alkali Metals � Group � Alkali 1 on the periodic table metals react with other elements by losing one electron � They are soooo REACTIVE that they are never found alone in nature. � Soft and shiny, you could cut it with a plastic knife.

� Examples: � **Lithium – found in batteries and medicines. � **Sodium and potassium are good for our health. � -Sodium is found in seawater and salt beds. � -Potassium is in spinach, baked potato with skin, plain yogurt and fish


� Group 2/starts with Berylium) � Reacts by losing 2 electrons � Also found combined in nature. � Gray and white � Fairly hard � Good conductors of electricity

� ** Calcium – is found in yogurt, milk, all dairy products, green leafy vegetables. It gives us strong bones. � ** Magnesium – When we mix Mg with aluminum it creates a strong but lightweight material that’s used to make ladders, airplane parts, and car parts.


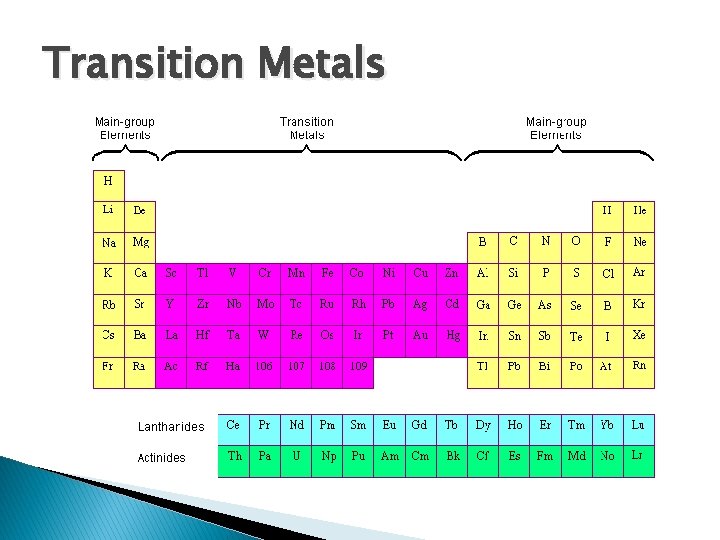
Transition Metals

� � Groups 3 -12. Some famous transition metals are: gold, platinum, silver, iron, mercury, � Hard and shiny � Good conductors of electricity � Form colorful compounds � Not so reactive

� Gold: very precious metal because it’s not reactive � **Iron: iron produces hemoglobin which carries oxygen into our bloodstream.

Metals in Mixed Group � Groups 13 -15 � Some are metals- not all! ( MIXED) � Less reactive � Aluminum, tin, lead

� Aluminum – lightweight metal used in beverage cans and airplane parts � Tin - a thin coating of tin protects steel from corrosion in cans of food. � Lead – it was once used in paints and water pipes. Since it is poisonous, it is no longer anymore. Now, it is used in car batteries and tires.


Lanthanides Actinides

Lanthanides � Soft � Malleable � Shiny � High � They conductivity are mixed with more common metals to make alloys. � Alloys- 2 or more metals mixed together � They are difficult to separate because they all have very similar properties.

Actinides � Only 4 actinides are found in nature. Uranium 238 is used in a power plant to create nuclear energy. The rest of the actinides are not found in nature. They are created in a lab, they produce a great amount of energy, and then they fizzle out. The energy is captured by scientists.