BOHR DIAGRAMS BOHR DIAGRAMS Show the electrons are

BOHR DIAGRAMS

BOHR DIAGRAMS Show the electrons are arranged. You need to know the number of protons, neutrons and electrons. Atomic number = protons = electrons How many protons does an atom of Boron have? � 5 protons How many electrons does an atom of Silver have? � 47 electrons

To find the number of neutrons: neutrons = Atomic mass – Atomic number Big Number Small Number The number of neutrons must be a whole number. How many neutrons does an atom of Boron have? � 11 – 5 = 6 neutrons How many neutrons does an atom of Silver have? � 108 – 47 = 61 neutrons

DRAWING BOHR DIAGRAMS Put the number of neutrons and protons in the nucleus (middle) Put electrons around the nucleus in orbits, also called energy levels �The first orbit can only hold 2 electrons �The second orbit can hold 8 electrons �The third orbit can hold 8 electrons
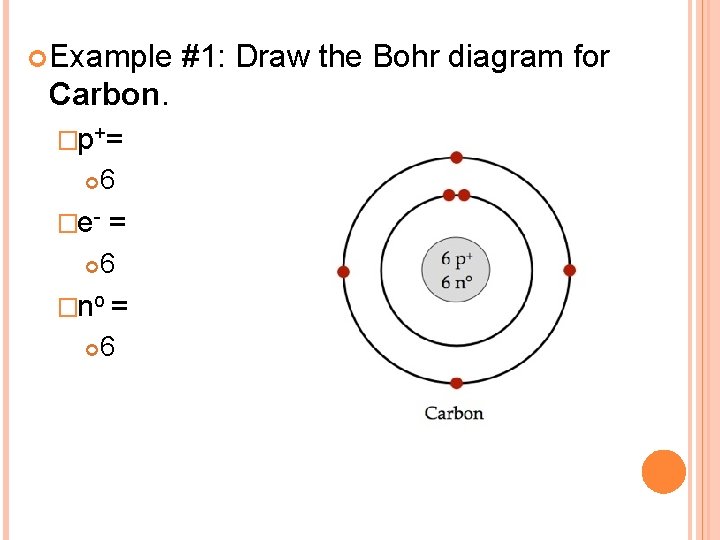
Example #1: Draw the Bohr diagram for Carbon. �p+= 6 �e- = 6 �no = 6
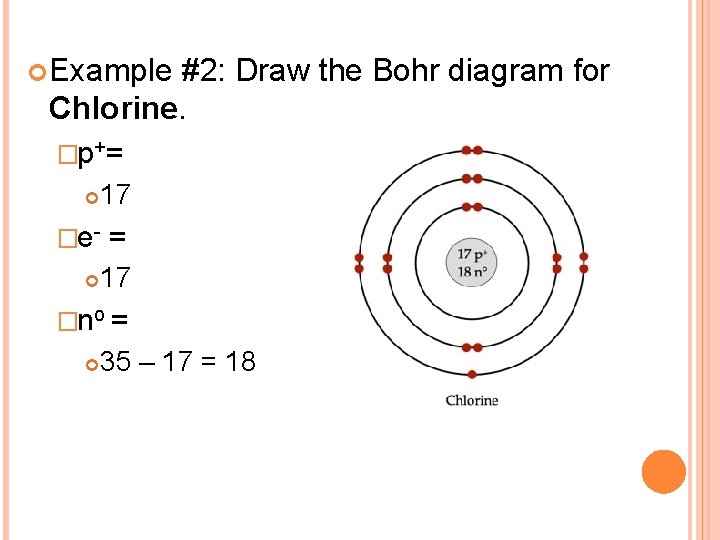
Example #2: Draw the Bohr diagram for Chlorine. �p+= 17 �e- = 17 �no = 35 – 17 = 18

VALENCE ELECTRONS Valence Electrons are the electrons located in the outermost energy level of the atom. Valence electrons have the strongest influence on the properties of an atom. Noble gases are unreactive because they have a full valence shell, so they do not gain, lose or share electrons with other atoms.
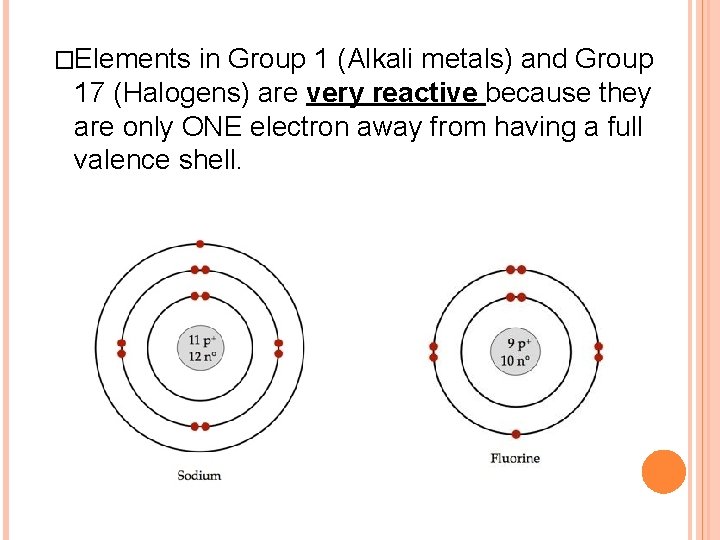
�Elements in Group 1 (Alkali metals) and Group 17 (Halogens) are very reactive because they are only ONE electron away from having a full valence shell.

- Slides: 9