THE ALKALI METALS Boardworks Ltd 2001 Alkali metals

THE ALKALI METALS © Boardworks Ltd 2001
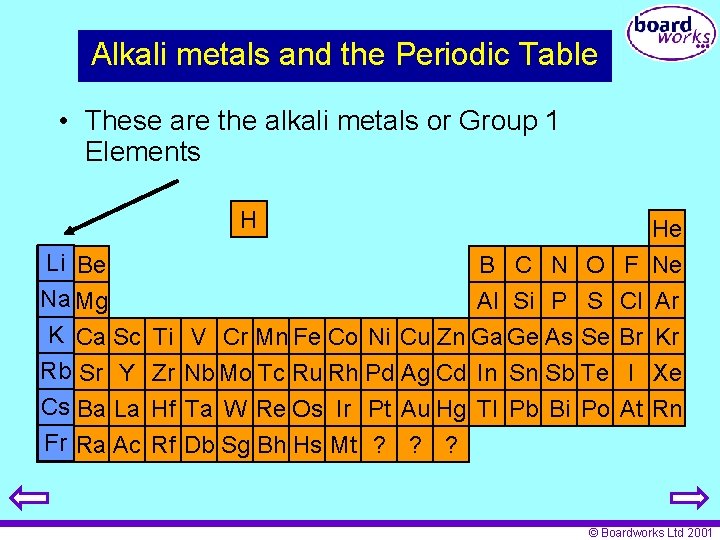
Alkali metals and the Periodic Table • These are the alkali metals or Group 1 Elements H Li Be Na Mg K Ca Sc Rb Sr Y Cs Ba La Fr Ra Ac He B C N O Al Si P S Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te F Cl Br I Ne Ar Kr Xe Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Rf Db Sg Bh Hs Mt ? ? ? © Boardworks Ltd 2001
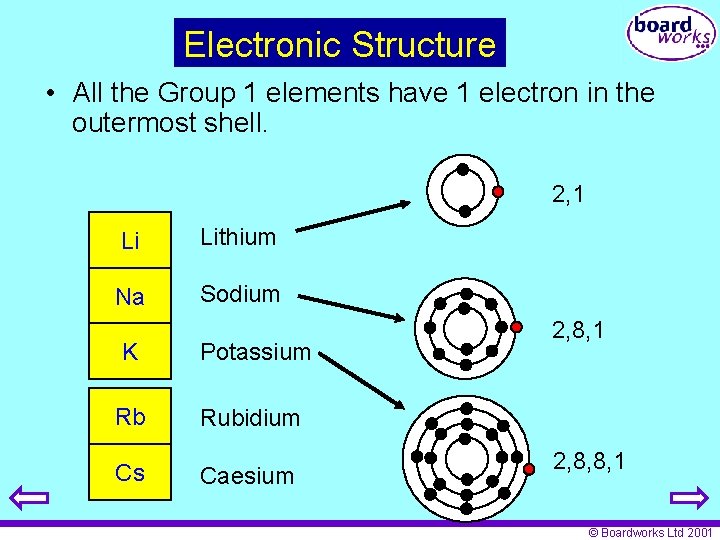
Electronic Structure • All the Group 1 elements have 1 electron in the outermost shell. 2, 1 Li Lithium Na Sodium K Potassium Rb Rubidium Cs Caesium 2, 8, 1 2, 8, 8, 1 © Boardworks Ltd 2001

Trends in Density • Lithium, sodium and potassium are all less dense than water so will float. • Densities follow a general, although not perfect, trend. Element Lithium Sodium Potassium Rubidium Caesium Symbol Li Na K Rb Cs Density 0. 53 0. 97 0. 86 1. 53 1. 88 © Boardworks Ltd 2001
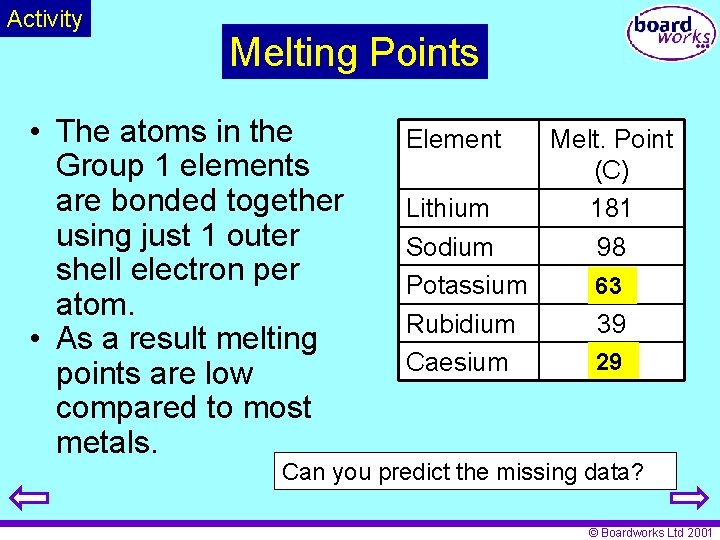
Activity Melting Points • The atoms in the Group 1 elements are bonded together using just 1 outer shell electron per atom. • As a result melting points are low compared to most metals. Element Lithium Sodium Potassium Rubidium Caesium Melt. Point (C) 181 98 63 39 29 Can you predict the missing data? © Boardworks Ltd 2001

Trends in Chemical Reactivity Li Na K Rb Cs Reactivity Increases • Reactivity increases down the group. • Reactions all involve loss of the outermost electron which changes the metal atom into an metal 1+ ion. • So, losing this electron seems to get easier as we go down the group. © Boardworks Ltd 2001
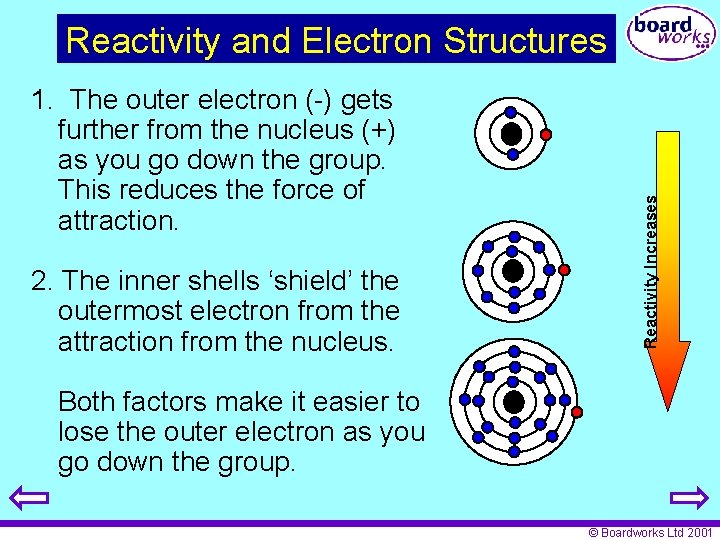
1. The outer electron (-) gets further from the nucleus (+) as you go down the group. This reduces the force of attraction. 2. The inner shells ‘shield’ the outermost electron from the attraction from the nucleus. Reactivity Increases Reactivity and Electron Structures Both factors make it easier to lose the outer electron as you go down the group. © Boardworks Ltd 2001
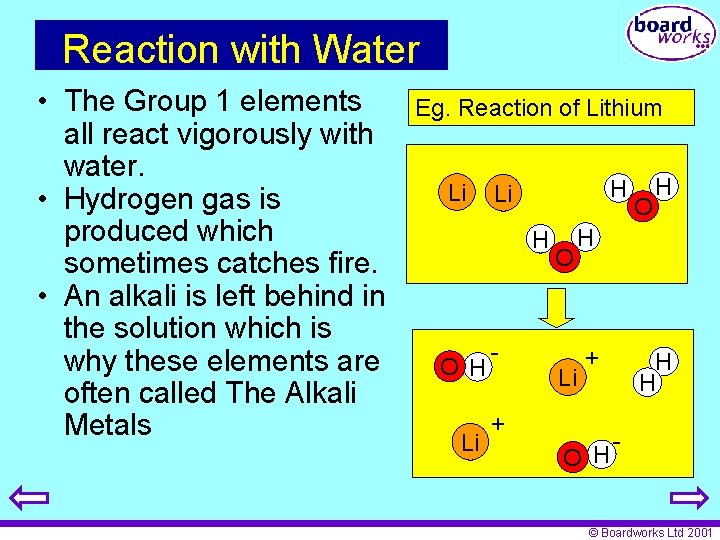
Reaction with Water • The Group 1 elements all react vigorously with water. • Hydrogen gas is produced which sometimes catches fire. • An alkali is left behind in the solution which is why these elements are often called The Alkali Metals Eg. Reaction of Lithium Li H O H Li - O O H H Li + + O H H H - © Boardworks Ltd 2001

Reaction of Lithium with Water • Lithium fizzes quickly in water forming lithium hydroxide and hydrogen Lithium + water g Lithium hydroxide + hydrogen 2 Li(s) + 2 H 2 O(l) 2 Li. OH(aq) + H 2(g) • The solution that remains is strongly alkaline. © Boardworks Ltd 2001

Reaction of Sodium with Water • Sodium fizzes very quickly in water catching fire if not allowed to move around. Sodium + water g Sodium hydroxide + hydrogen 2 Na(s) + 2 H 2 O(l) 2 Na. OH(aq) + H 2(g) Sodium on water Enlarged sodium on fire in water © Boardworks Ltd 2001

Activity Potassium with Water • Lithium fizzes. Sodium reacts more vigorously. • What will potassium do? What will the word equation and chemical equations be for the reaction of potassium with water? Potassium + water Potassium hydroxide + hydrogen 2 K(s) + 2 H 2 O(l) 2 KOH(aq) + H 2(g) © Boardworks Ltd 2001

Activity The Group 1 Metals and oxygen: • The Group 1 elements burn in air to form metal oxides. Don’t try to put them out with water! Lithium + oxygen Lithium Oxide 4 Li (s) + O 2(g) 2 Li 2 O (s) • What will the word equation and chemical equations be for the reaction of sodium with air? Sodium + oxygen 4 Na(s) + O 2 (g) sodium oxide 2 Na 2 O (s) © Boardworks Ltd 2001
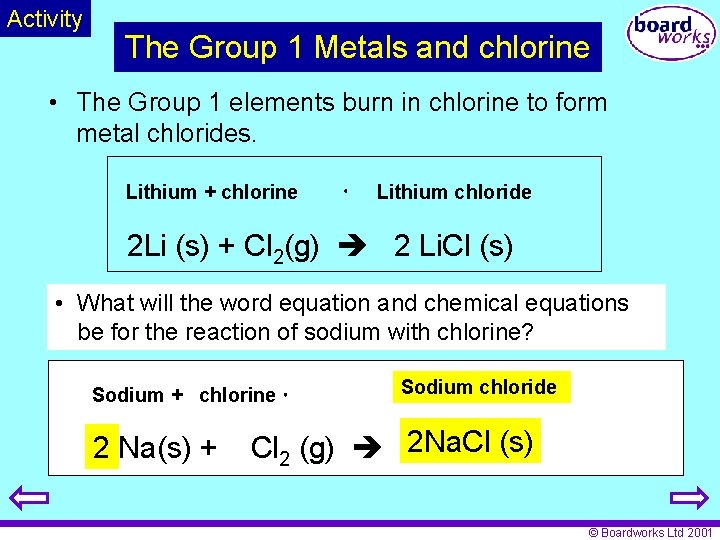
Activity The Group 1 Metals and chlorine • The Group 1 elements burn in chlorine to form metal chlorides. Lithium + chlorine Lithium chloride 2 Li (s) + Cl 2(g) 2 Li. Cl (s) • What will the word equation and chemical equations be for the reaction of sodium with chlorine? Sodium + chlorine 2 Na(s) + Sodium chloride Cl 2 (g) 2 Na. Cl (s) © Boardworks Ltd 2001

Uses of the Group 1 Metals • The metals themselves are too reactive to have many uses although sodium vapour gives street lights their yellow glow. • Lithium metal is used to improve the strength of aircraft alloys and is also used in some electrical batteries. • Common sodium compounds include “salt”, (sodium chloride), “bicarbonate” (sodium hydrogen carbonate), washing soda (sodium carbonate) and caustic soda (sodium hydroxide. ) • Potassium compounds are used in “NPK fertilisers”, in weedkillers, explosives and many other chemicals. Sodium light potassium © Boardworks Ltd 2001

Activity Using Sodium to Transfer Heat • The activity • Following an accident at a nuclear power station three groups are represented at a public meeting. • The debate centres around an accident involving a spill of molten sodium metal which was being used to cool the reactor. • It needs to include: – The benefits of using sodium to transfer heat and the fact that risks resulting from chemical reactivity are containable. – The chemical reactivity of sodium and the fact that if containment did fail the whole power station could blow up. – Whethere are other safer metals that could be used in place of sodium. • 3 groups prepare presentations. • Others students devise questions to follow the presentations. © Boardworks Ltd 2001

Activity The Incident • Most power stations burn a fossil fuel and use water to transfer heat from the burners to the turbine area. • An alternative to water is sodium. Although solid it melts fairly easily and is a better conductor of heat than water. • This has prompted its use as a coolant to absorb and transfer the heat produced in nuclear power stations. • To Japan, a country with no fossil fuels, nuclear power is particularly attractive. However, in 1996 Japan’s nuclear industry suffered a setback when a split in a stainless steel pipe spewed 3 tonnes of molten sodium over the reactor floor. • Nuclear representatives say there was no radiation leak and opponents to nuclear power were whipping up public concern. • Anti-nuclear protestors say that had it leaked underneath the floor the entire nuclear station would have been at risk. • The government agreed to make plans about how to deal with a nuclear accident just in case one did ever happen. (It’s true!) © Boardworks Ltd 2001

Activity Nuclear Industry Representatives group • Make a case as to why Japan must have nuclear power. • Spell out the very low accident rate in the industry. • Explain why a liquid that can absorb heat better is a good thing (safer? ) for a nuclear reactor. • Spell out the fact that you understand the common reactions of sodium and had already set in place systems to prevent these reactions being a danger. • Make clear that there is no totally safe way of generating energy and that use of fossil fuels also entails accidents and guaranteed pollution. © Boardworks Ltd 2001

Activity Anti-Nuclear Protest group • Explain that risk assessment must take account both of the chances of an accident and the impact of that accident. Leaked long lasting radioactive material or even melt-down and nuclear explosion! • Challenge the use of sodium (rather than larger volumes of water) as representing a needless risk. • Spell out in detail the possibility of explosive reactions involving sodium and potentially devastating outcomes. • Other fuels are available even if they have to be imported. © Boardworks Ltd 2001

Activity Dr Ivan Idea Syndicate • One of the main reasons for using sodium is that it melts easily and, as a metal, it conducts heat well. • Check out the melting point, reactivity and toxicity of other metals and consider the feasibility of using them. • Are there particular risks you would need to guard against? Can you suggest ways to minimise these? • Or - should you just go back to using water for heat transfer? © Boardworks Ltd 2001

Activity Dr Ivan Idea Syndicate 2 Some data on the metals that melt below 500 o. C © Boardworks Ltd 2001

How many electrons do the alkali metals have in their outer shell? A. B. C. D. 1 2 4 7 © Boardworks Ltd 2001

What charge ions are formed by the alkali metals? A. B. C. D. 4 3 2 1 © Boardworks Ltd 2001

Which answer places the alkali metals in order of increasing reactivity? A. B. C. D. Na, Li, K, K, Na, Li Li, Na, K Li, K, Na © Boardworks Ltd 2001

When alkali metals react with water we get: A. B. C. D. Hydrogen + a metal oxide Oxygen + a metal oxide Hydrogen + a metal hydroxide Oxygen +a metal hydroxide © Boardworks Ltd 2001

What will the melting point of caesium be? A. -10 o. C B. 0 o. C C. 25 o. C D. 38 o. C © Boardworks Ltd 2001
- Slides: 25