BOHR DIAGRAMS METALS NONMETALS AND METALLOIDS BOHR DIAGRAMS

BOHR DIAGRAMS, METALS, NONMETALS, AND METALLOIDS

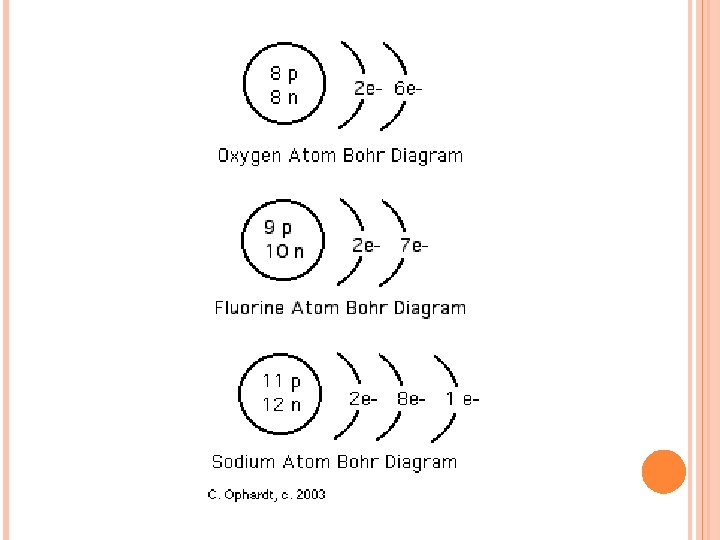
BOHR DIAGRAMS A model invented by Niels Bohr in the 1900’s to show the number and arrangement of electrons in each shell/energy level. Rule: Each shell can only hold a certain number of electrons. 1 st shell 2 electrons 2 nd shell 8 electrons 3 rd shell 8 electrons 4 th shell 18 electrons This rule of 2 -8 -8 -18 applies to ALL atoms!
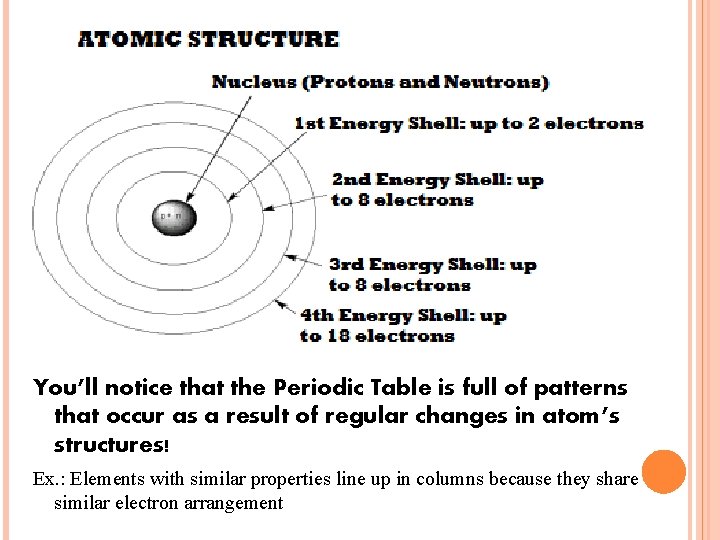
You’ll notice that the Periodic Table is full of patterns that occur as a result of regular changes in atom’s structures! Ex. : Elements with similar properties line up in columns because they share similar electron arrangement
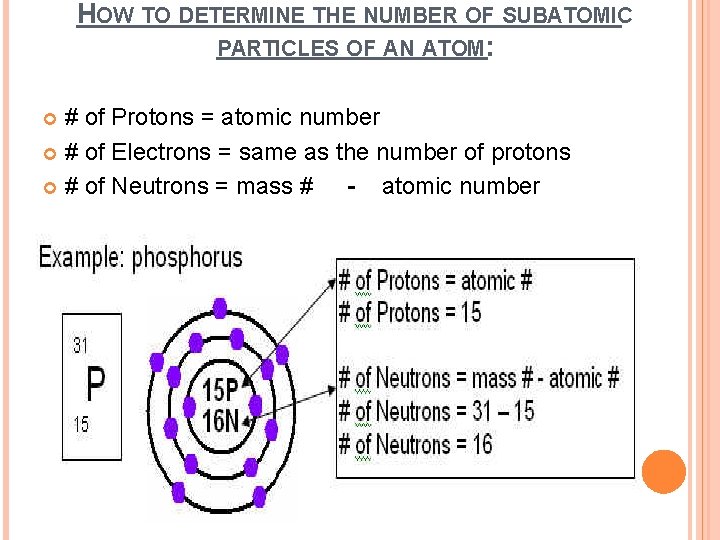
HOW TO DETERMINE THE NUMBER OF SUBATOMIC PARTICLES OF AN ATOM: # of Protons = atomic number # of Electrons = same as the number of protons # of Neutrons = mass # - atomic number

HOW TO DRAW BOHR DIAGRAMS Step 1: Determine the # of protons, neurtons, and electrons Eg. P (Phosphorus) Protons = 15 Electrons = 15 Neutrons = atomic mass – atomic # = 31 - 15 = 16 Step 2: Draw the nucleus as a circle:
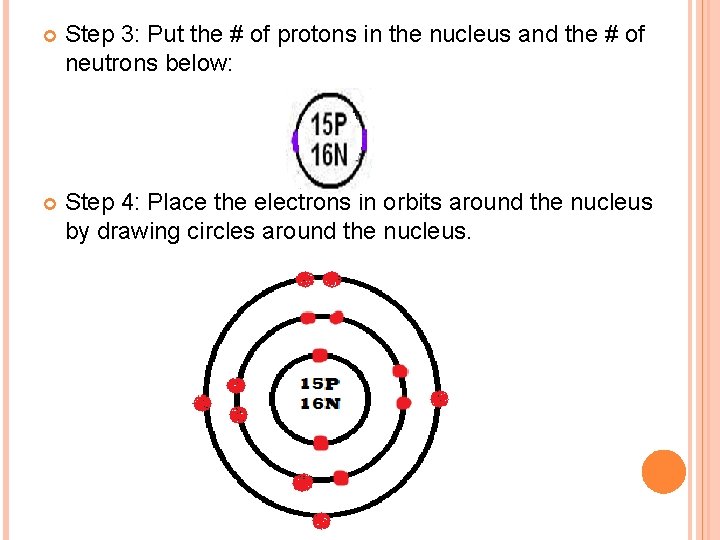
Step 3: Put the # of protons in the nucleus and the # of neutrons below: 12 p 12 n Step 4: Place the electrons in orbits around the nucleus by drawing circles around the nucleus. 12 p 12 n

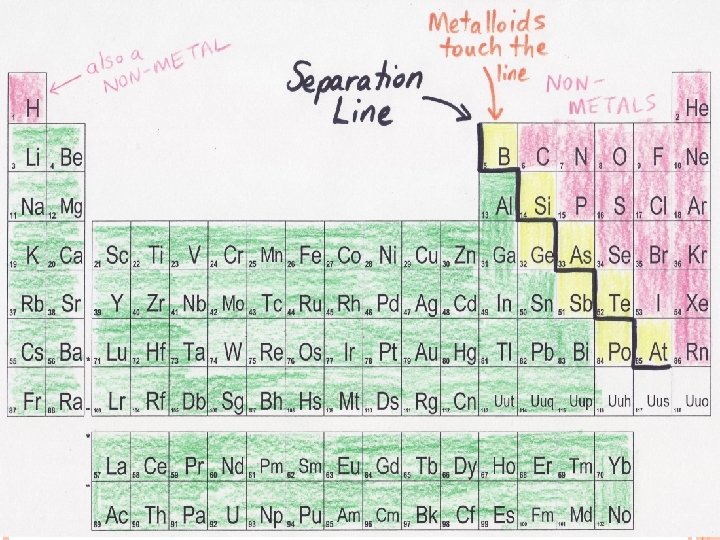
METALS, NON-METALS, AND METALLOIDS Mendeleev organized the Periodic Table by properties, which made some interesting patterns He found 3 groups: � Metals � Non-metals � Metalloids The metalloids share some properties with both metals and non-metals!
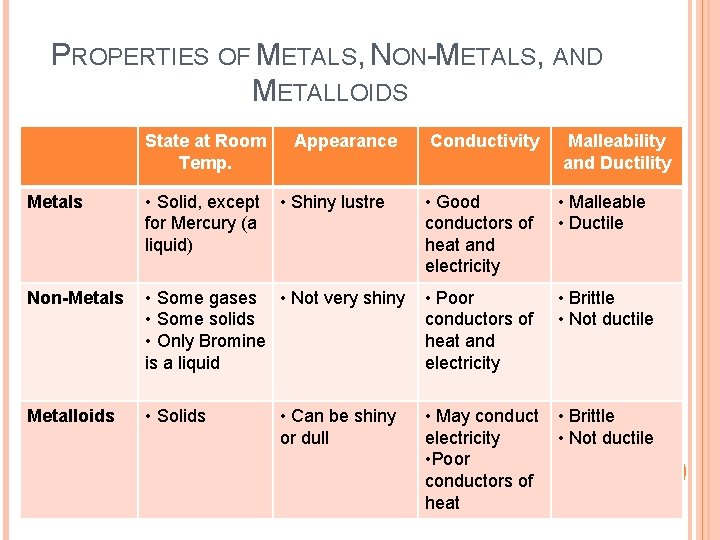
PROPERTIES OF METALS, NON-METALS, AND METALLOIDS State at Room Temp. Appearance • Shiny lustre Conductivity Malleability and Ductility • Good conductors of heat and electricity • Malleable • Ductile Metals • Solid, except for Mercury (a liquid) Non-Metals • Some gases • Not very shiny • Some solids • Only Bromine is a liquid • Poor conductors of heat and electricity • Brittle • Not ductile Metalloids • Solids • May conduct electricity • Poor conductors of heat • Brittle • Not ductile • Can be shiny or dull
- Slides: 10