Metals Nonmetals and Metalloids Coloring in the Periodic



























- Slides: 36

Metals, Nonmetals and Metalloids

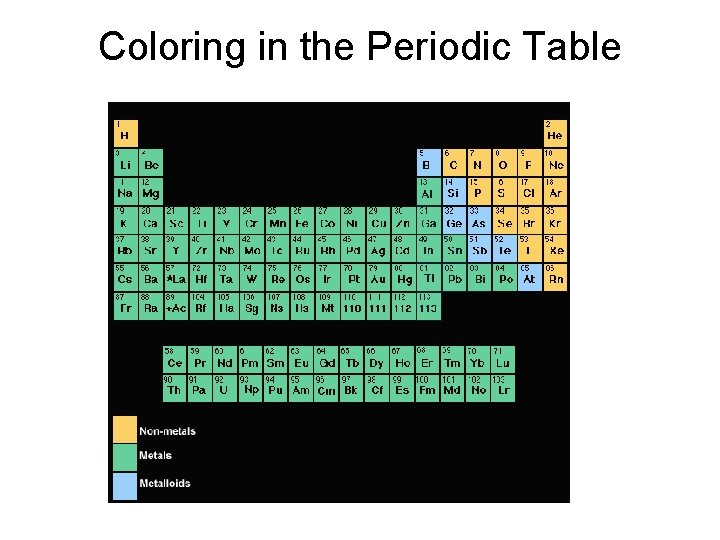
Coloring in the Periodic Table

Notice the difference between the appearance of the metals and nonmetals. Click here for a better view of each of the elements.

• • • Physical Properties of Metals: Luster (shininess) Good conductors of heat and electricity High density (heavy for their size) High melting point Ductile (most metals can be drawn out into thin wires) Malleable (most metals can be hammered into thin sheets) Chemical Properties of Metals: Easily lose electrons Bismuth Corrode easily. Corrosion is a gradual wearing away. (Example: silver tarnishing and iron rusting


Physical Properties of METALS Metals are malleable. metals ability to be shaped or formed as by hammering or pressure; can be beaten into thin sheets Aluminum is malleab

Physical Properties of METALS Metals have shiny luster. (or metallic luster) LUSTER – the way an object’s surface reflects light

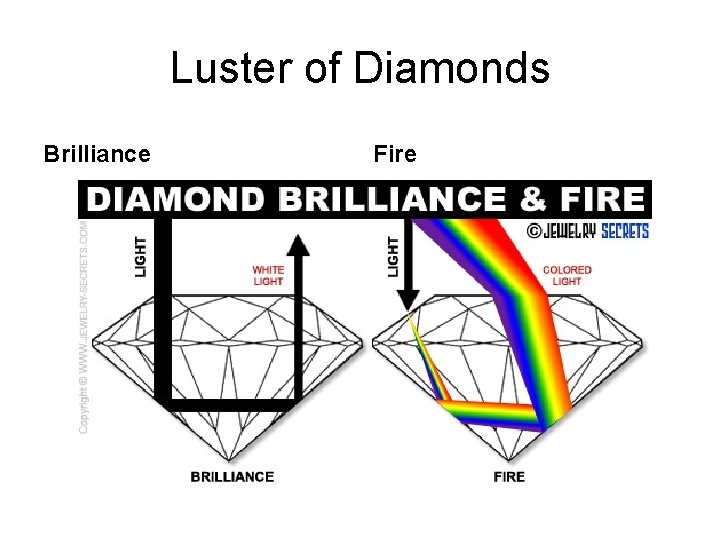
Luster of Diamonds Brilliance Fire

Physical properties of METALS • Metals are SOLIDS. (except mercury) • Metals are HARD. (except Lithium, Potassium, Sodium)


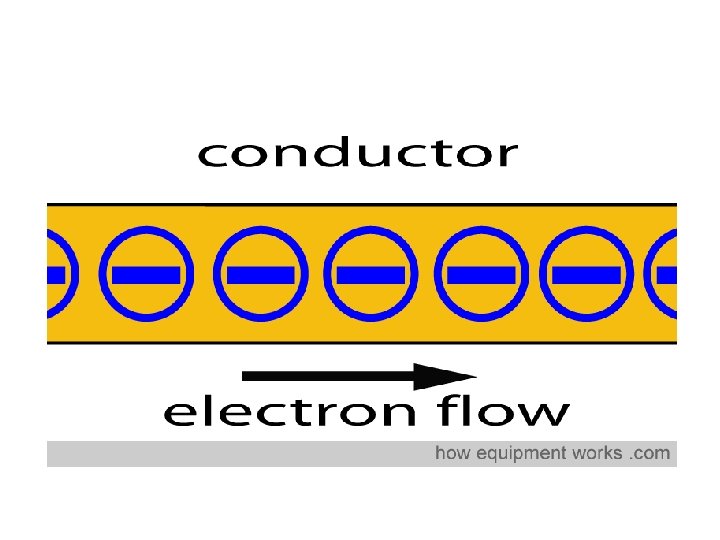
Physical Properties of METALS • Metals are good conductors of electricity. In a conductor, electric current can flow freely. Copper Wiring Copper, silver and gold are good electrical conductors!

Conductors electrons free to roam


METALS are the best conductors of heat. Electrons in metals move more freely, allowing heat energy to travel across the metal. Think about whenever you’ve left a spoon in a hot drink! Best conductors: silver and copper

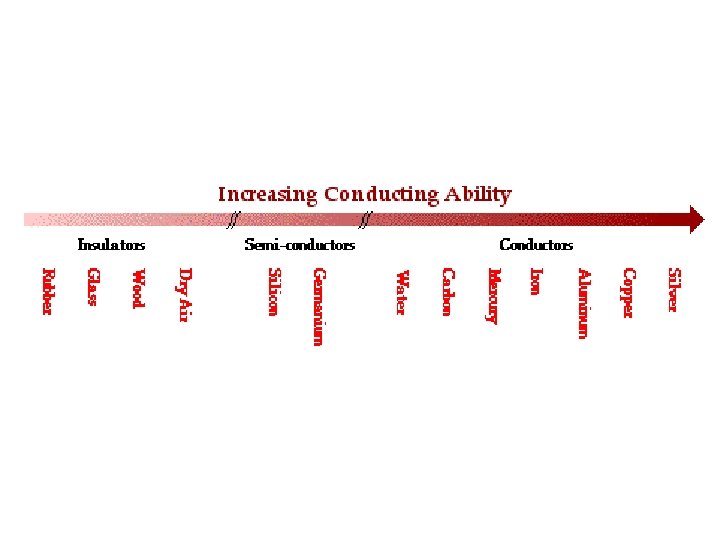
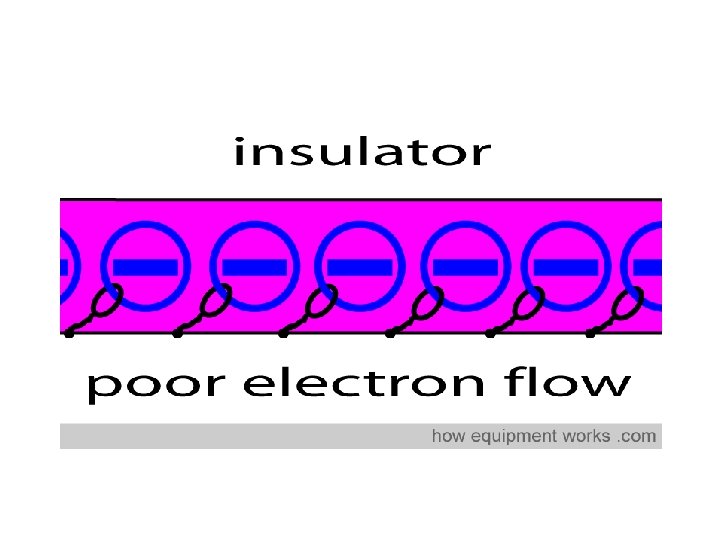
Insulators • Do not allow heat and electricity to travel through Examples: rubber plastic



Insulators – electrons don’t transfer energy

Physical Properties of METALS Metals are ductile. Ductility or ductile – can be drawn into a wire


Physical Properties of Metals: • • • Luster (shininess) Good conductors of heat and electricity High density (heavy for their size) High melting point Ductile Malleable Chemical Properties of Metals: • Easily lose electrons • Corrode easily. Corrosion is a gradual wearing away. (Example: silver tarnishing and iron rusting

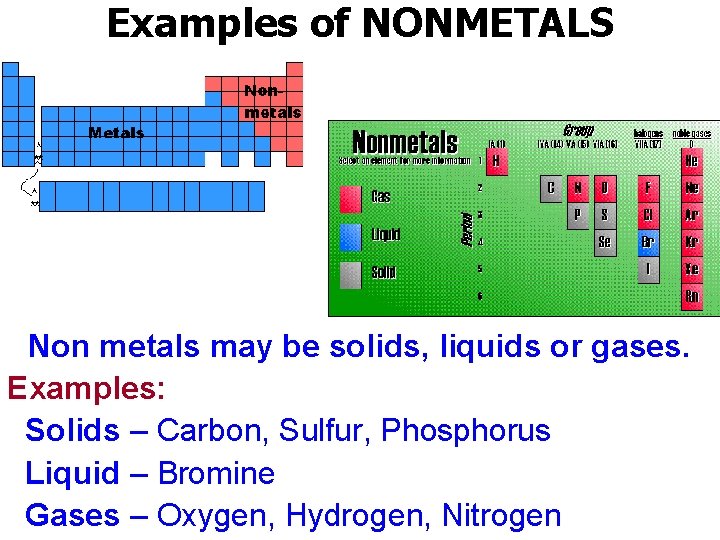
Examples of NONMETALS Non metals may be solids, liquids or gases. Examples: Solids – Carbon, Sulfur, Phosphorus Liquid – Bromine Gases – Oxygen, Hydrogen, Nitrogen

Physical Properties of NONMETALS Nonmetals have a dull luster. (They are not shiny!) Ex. Phosphorus

Physical Properties of NONMETALS Nonmetals are insulators. They do not conduct electricity or heat well. The atoms in nonmetals do not have loose electrons. What would you rather stir a hot pot with —a wooden spoon or a metal spoon?

Physical Properties of NONMETALS Nonmetals are soft (except for diamonds) and brittle. Example: Sulfur


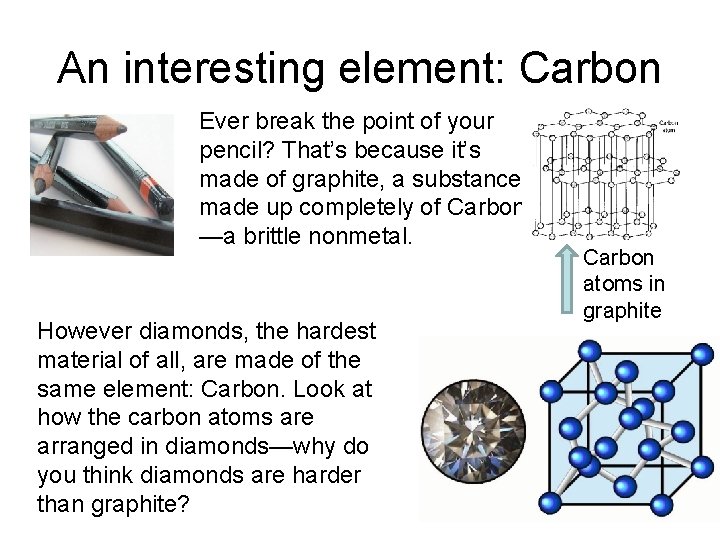
An interesting element: Carbon Ever break the point of your pencil? That’s because it’s made of graphite, a substance made up completely of Carbon —a brittle nonmetal. However diamonds, the hardest material of all, are made of the same element: Carbon. Look at how the carbon atoms are arranged in diamonds—why do you think diamonds are harder than graphite? Carbon atoms in graphite

Non-metals • are not able to conduct electricity or heat very well • cannot be rolled into wires (ductile) or pounded into sheets (malleable) • have no metallic luster • do not reflect light

METALLOIDS The elements contained in the classification of Metalloids:

METALLOIDS • Physical properties of both metals and nonmetals. • Some are shiny, some are dull, they are somewhat malleable and ductile, and can conduct heat and electricity at a lesser level than metals. SILICON BORON ARSENIC

METALLOIDS • Some metalloids are useful semiconductors, which are used in electronics (radio, computers, telephones, etc. ) • They are useful because they conduct just the right amount of electricity or heat.

Metalloids • Metalloids have properties of both metals and non-metals. • Boron (B) • Silicon (Si) • Germanium (Ge) • Arsenic (As) • Antimony (Sb) • Tellurium (Te) • Polonium (Po) Tellurium is one of the metalloids or semimetals.

Where do we find METALS? Some metals like gold, silver, and platinum are found as pure substances in the earth’s crust because they are least reactive. Most metals are reactive and are found as oxides (react with oxygen), carbonates (react with carbon), sulfides (react with sulfur). Minerals : are elements or compounds which occur naturally inside the earth’s crust. Ore : is a mineral from which metals can be extracted profitably.

Video on Physical Properties of Metals • https: //www. youtube. com/watch? v=5 R 08 N 3 u 5 Z_Y • https: //www. youtube. com/watch? v=Oz 8 Gp DVz 5 ag

How coins are made • http: //www. youtube. com/watch? v=Dk. HFN n. OK 3 Bg

How aluminum foil is made • http: //www. youtube. com/watch? v=f 4 OTj 9 y NOak