Metals Nonmetals and Metalloids Tuesday September 16 th

Metals, Nonmetals, and Metalloids Tuesday, September 16 th, 2014

Do Now � Take out the form that you got signed last night. I will ask to collect them after the Do Now � What is a metal? � What is a nonmetal? � What is a metalloid?

Objectives � SWBAT identify the characteristics and properties of metals, non-metals, and metalloids by completing a Venn Diagram Essential Question � How can you use the Periodic Table to predict the properties of an element?
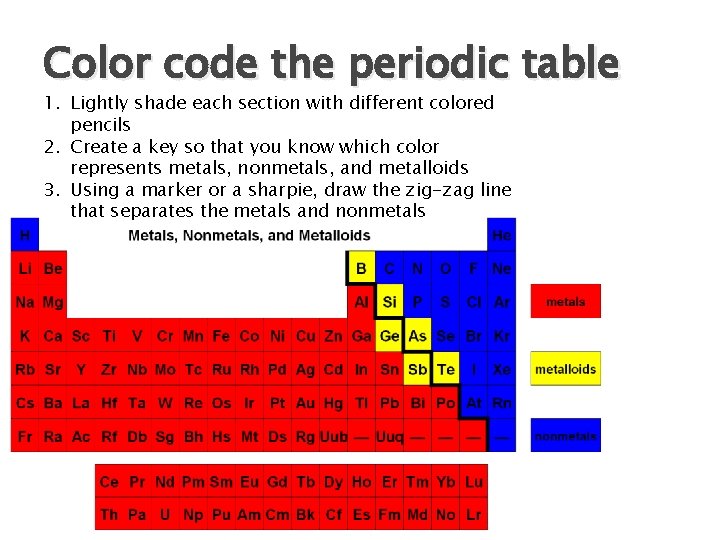
Color code the periodic table 1. Lightly shade each section with different colored pencils 2. Create a key so that you know which color represents metals, nonmetals, and metalloids 3. Using a marker or a sharpie, draw the zig-zag line that separates the metals and nonmetals

Metals � Most malleable and _______ ductile metals are _____ hammered into thin sheets ◦ They can be ________ wire ◦ They can be drawn into a thin ____ solid at room temperature � Metals are ______ liquid is _____ mercury ◦ The only metal that is a _______ energy � Metals have a low ionization ________ lose electrons in ◦ This means that they usually ____ chemical reactions metallic � Metals have _____ properties shiny ◦ They are _______ conductors of heat and electricity ◦ They are good ______

Non-Metals vary in appearance ______ heat and ______ electricity � Poor conductors of _____ insulators but good ______ melting point � Low ________ high ionization energy � Have a ______ gain electrons in chemical ◦ This means that they ______ � Nonmetals reactions � Solid brittle nonmetals are ____

Metalloids intermediate between are _______ metals and nonmetals � Metalloids are useful ________ semiconductors increases the conductivity ◦ As the temperature ______, also _____ increases ionization energy � Can have a high or low ________ � Properties
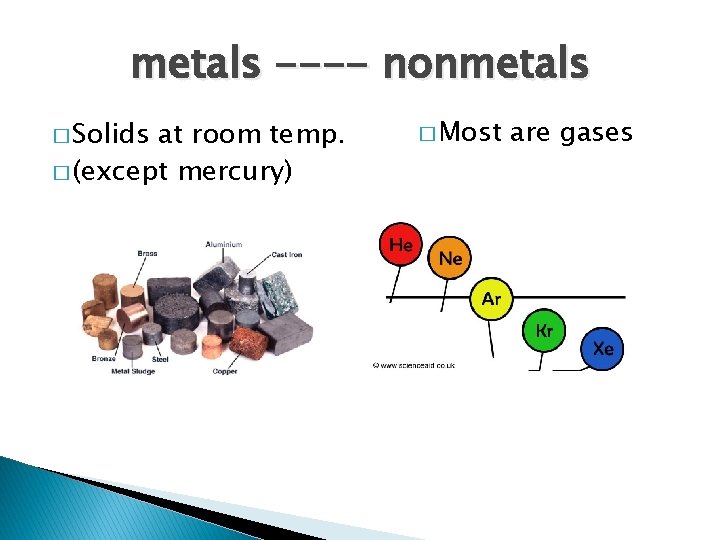
metals ---- nonmetals � Solids at room temp. � (except mercury) � Most are gases

metals ----- nonmetals � higher melting points � lower melting points

metals ---- nonmetals � malleable � brittle (break easily) � Softer than metals (can be pounded & shaped) � harder

metals ---- nonmetals � Ductile (can be drawn into wire) � Not ductile

metals ---- nonmetals � lustrous (shiny) � dull
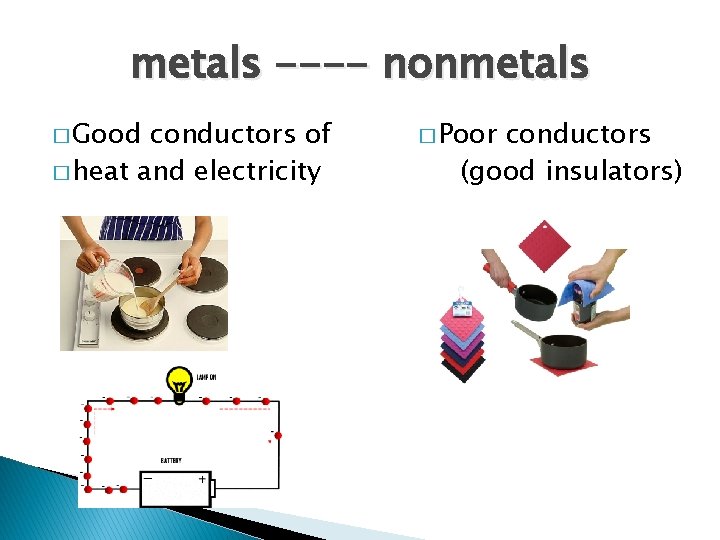
metals ---- nonmetals � Good conductors of � heat and electricity � Poor conductors (good insulators)

metals ---- nonmetals � Includes the most � reactive group (1) (alkali metals) Brainiac Video (no commercials) Backup copy on teachertube--with commercials � Includes the least reactive group (18) (noble gases) *most stable

metals ---- nonmetals � Tend to lose electrons (form positive ions +) � Tend to gain electrons (form negative ions -)

Metalloids � Have some of the properties of metals and some of the properties of nonmetals � Include semiconductors (Used in electronics)
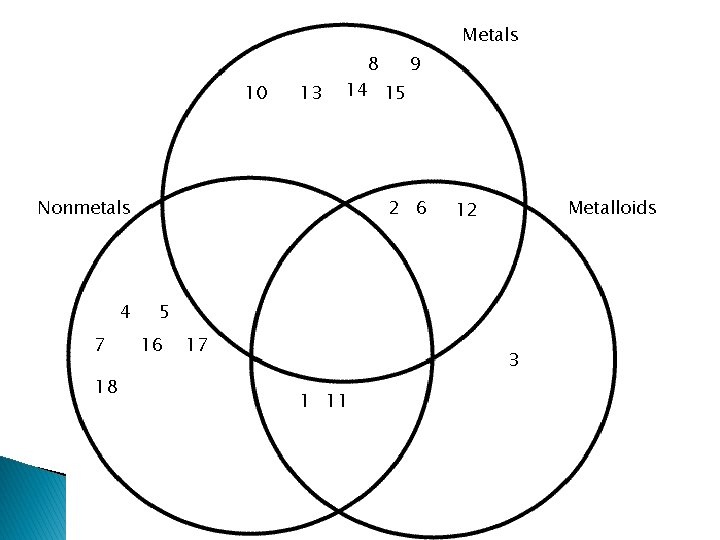
Metals 10 13 8 9 14 15 2 6 Nonmetals 4 7 18 Metalloids 12 5 16 17 3 1 11

Answer the Essential Question On a sheet of lined paper or on the bottom of your guided notes � How can you use the Periodic Table to predict the properties of an element?

Exit Ticket � On the slip of paper � Have a great day
- Slides: 19