Metals NonMetals Metalloids The periodic table separates elements
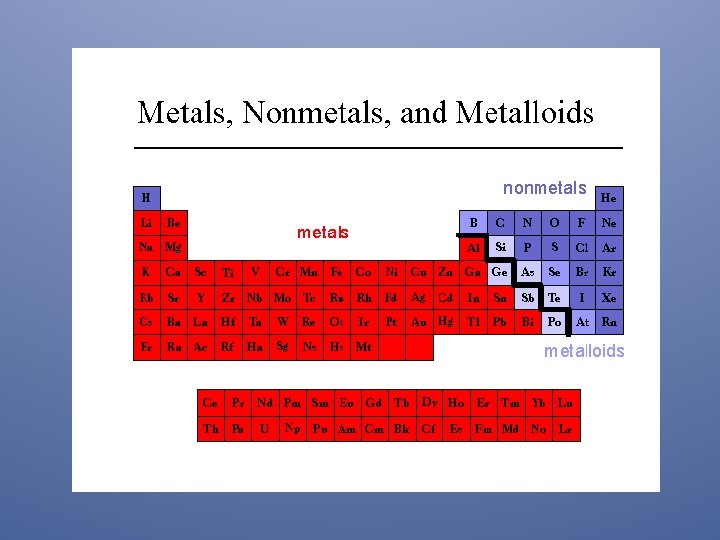

Metals, Non-Metals, Metalloids The periodic table separates elements into three groups: the metals (on the left), nonmetals (on the right), and metalloids (down the stairs).

Metals These metals have properties that you normally associate with the metals you encounter in everyday life: • Are silver-grey in color • Are solids at room temperature (except for Mercury) • Reflect light when polished (luster) • Can be bent or hammered flat (malleable) • Can be drawn into wire (ductile)

Metals • Are strong and resist bending and breaking (tensile strength) • Have a characteristic sound when struck with an object (sonorous) • Have high melting points • Have high boiling points • Are good conductors of heat and electricity • Have high densities (heavy for their size) • Can be attracted to a magnet
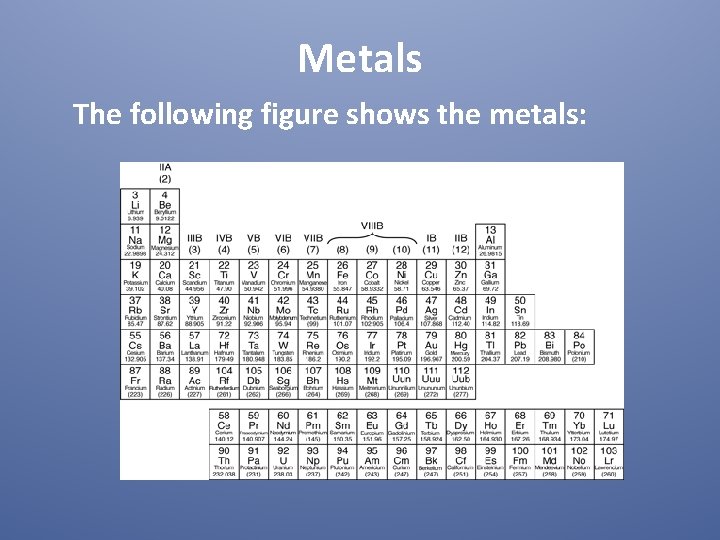
Metals The following figure shows the metals:

Non-metals Except for the elements that border the stairstepped line, the elements to the right of the line are classified as nonmetals (along with hydrogen, H). Nonmetals have properties opposite those of the metals.

Nonmetals • Exist as solids, liquids, or gases at room temperature • Do not reflect light well (no luster) • Are brittle (break easily) • Cannot be hammered flat (not malleable) • Cannot be drawn into wire (not ductile) • Are soft and bend or break easily (low tensile strength)

Nonmetals • Do not have a characteristic sound when struck with an object (not sonorous) • Have low melting points • Have low boiling points • Are poor conductors of heat and electricity • Have low densities (light for their size) • Are not attracted to a magnet
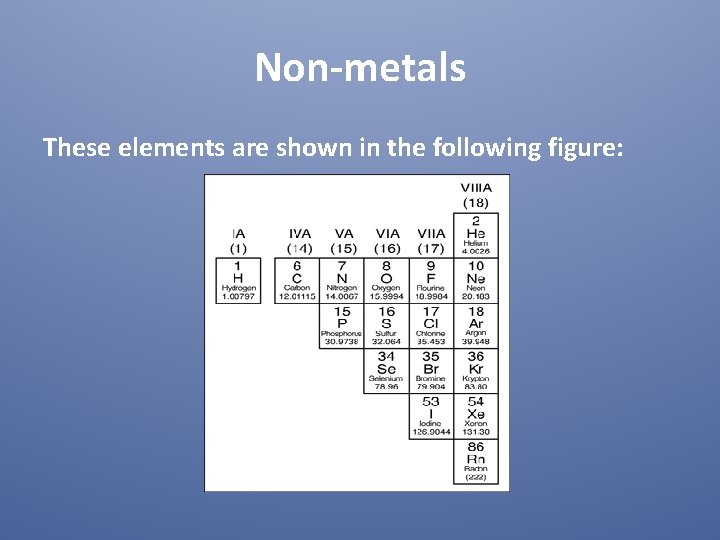
Non-metals These elements are shown in the following figure:

Metalloids The elements that border the stair-stepped line are classified as metalloids. • Are generally solids • Can be shinny or dull (luster) • May or may not be drawn into wire (ductile) • May or may not be hammered flat (malleable) • May or may not be brittle (break easily) • Conduct heat and electricity better than nonmetals, but not as well as metals

Metalloids • • Tend to make good semiconductors Have boiling points that are widely varied Have melting points that are widely varied Have densities that are widely varied
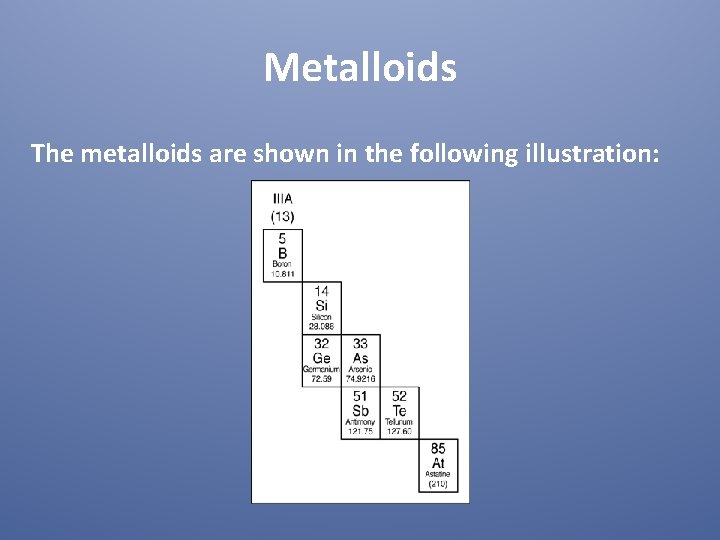
Metalloids The metalloids are shown in the following illustration:

Vocabulary • Property - A characteristic or feature of matter; every type of matter has its own set of properties such as color or density

Vocabulary • Density - A measure of the quantity of some physical property (usually mass) per unit length, area, or volume (usually volume)

Vocabulary • Sonorous - producing or capable of producing sound

Vocabulary • Luster – shiny or dull. Most metals are shiny and nonmetals are not.

Vocabulary • Malleability - how easily something can be molded or beaten into a different shape. Although not all metals are soft enough to change shape without heat, they are still considered malleable.

Vocabulary • Conductivity – how well it allows electricity to move through. Metals are conductive and nonmetals are not.

Vocabulary • Ductile – can be drawn into a wire

Vocabulary • Magnetism – attracted or not attracted to a magnet

Vocabulary • Brittleness – how easily it breaks. Nonmetals are brittle.

Vocabulary • Tensile Strength - the maximum stress that a material can withstand while being stretched or pulled before necking, which is when the specimen's cross section starts to significantly stretch
- Slides: 22