The Periodic Table GroupsFamily Columns Each column contains
The Periodic Table
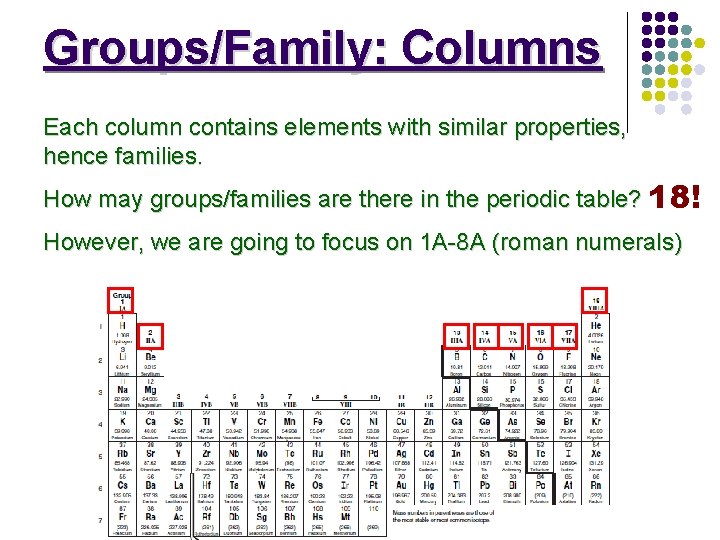
Groups/Family: Columns Each column contains elements with similar properties, hence families. How may groups/families are there in the periodic table? 18! However, we are going to focus on 1 A-8 A (roman numerals)

Periods: Rows The rows are also known as shells, energy levels or “n”. How many shells can an element contain? 7!

Staircase: The metalloids Separates metals and nonmetals Contain the metalloids on either side. * Metalloids have properties of both metals and nonmetals! * *
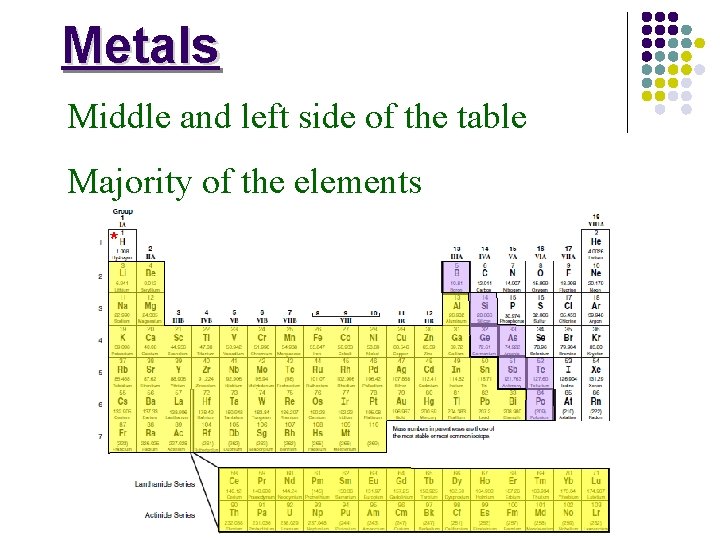
Metals Middle and left side of the table Majority of the elements *
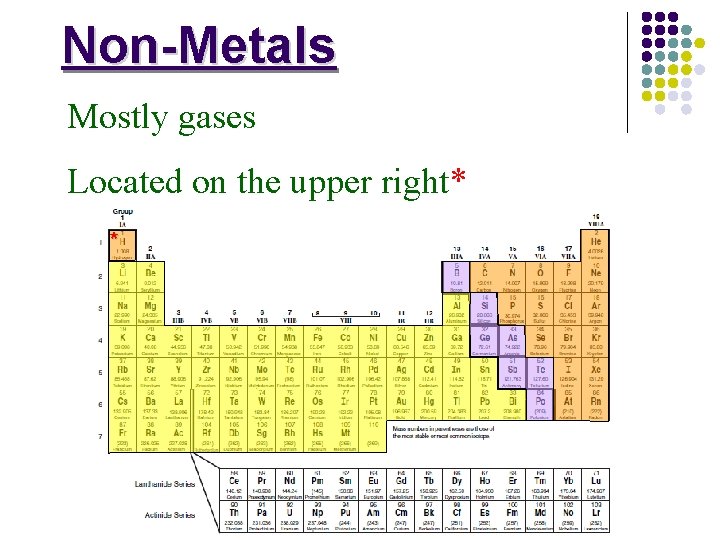
Non-Metals Mostly gases Located on the upper right* *
Alkali Metals: 1 A Highly reactive metals (reacts violently with water! Silver, shiny and easily cut. (1 outer electron)
Alkaline Metals: 2 A A little more dense than the alkali metals Reactive metals (2 outer electrons)
Transition Metals: B’s Most of the “normal” metals you think of: gold, silver, platinum, iron, etc. (1 -3 outer electrons)

Halogens: 7 A Gases: very reactive Most are diatomic (2 atoms) and form salts (7 outer electrons)
Noble Gases: 8 A Inert: non reactive, colorless, odorless gases Full outer shell of electrons (8 outer electrons) (the octet rule)
Lanthanides Transition metals taken out to save space Shiny and reactive
Actinide All radioactive! 95 - 103 do not exist in nature

Color Code: l l l Color the square for Hydrogen pink. Lightly color all metals yellow. Place black dots in the squares of all alkali metals. Draw a horizontal line across each box in the group of alkaline earth metals. Draw a diagonal line across each box of all transition metals. Color the metalloids purple. Color the nonmetals orange. Draw vertical lines through all the boxes of the halogens. Draw checkerboard lines through all the boxes of the noble gases. Color all the lanthanides red. Color all the actinides green.
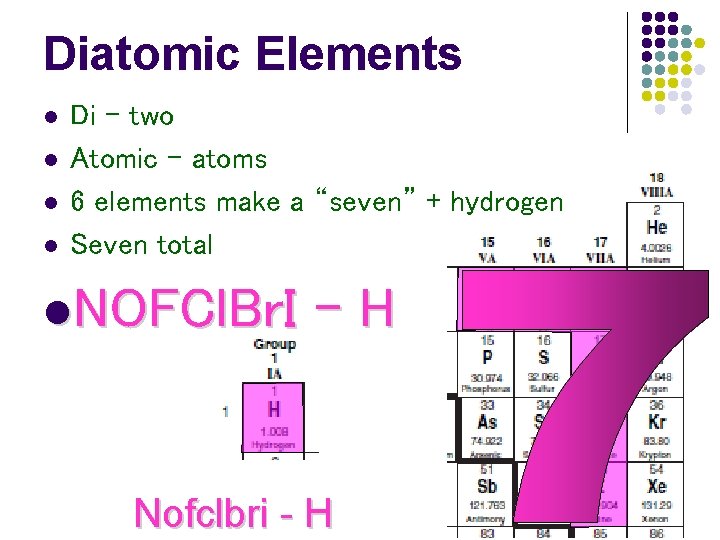
Diatomic Elements l l Di – two Atomic – atoms 6 elements make a “seven” + hydrogen Seven total l. NOFCl. Br. I -H Nofclbri - H

Gases! l l Gases at room temperature Most are noble (8 A)

Liquids! l l Only two elements are liquid at room temp. Bromine and Mercury

Blocks! l The table can also divided into Blocks, which are like neighborhoods l There are 4 blocks: s, p, d, f l Blocks are also known as sub-shells
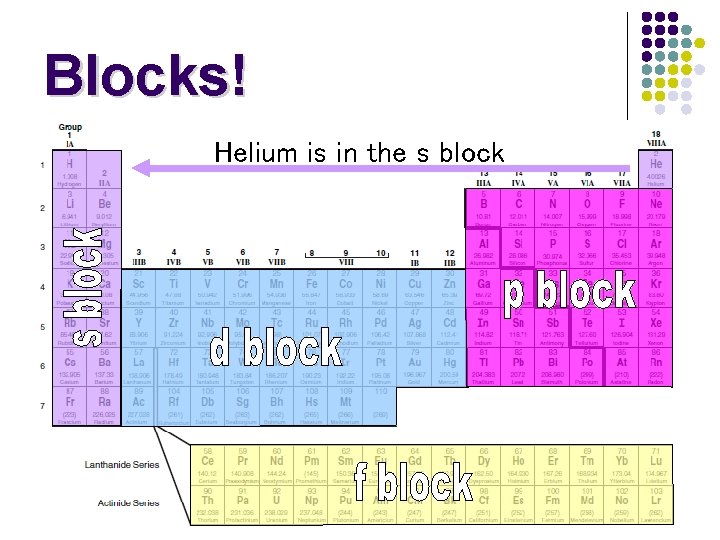
Blocks! Helium is in the s block

Groups: additional l Although there are 18 groups we are only going to focus on the “A” Groups: 1 A – 8 A; leaves out the transition metals and the f block l Groups are also named by the first element in the group. l Ex: carbon group, oxygen group, fluorine group
- Slides: 20