TOPIC Metals vs Nonmetals Classifying the Elements 23








- Slides: 19

TOPIC: Metals vs Nonmetals

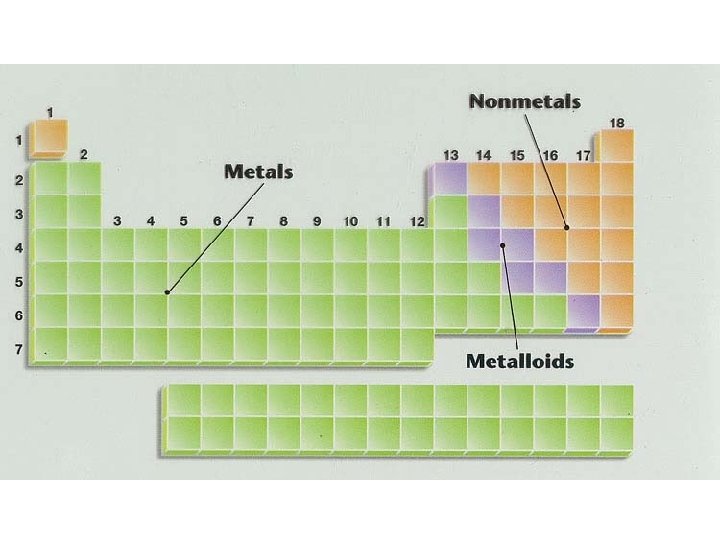
Classifying the Elements • 2/3 (75%) of elements are metals • Remaining elements: non-metals & metalloids (semi-metals) • Metalloids: – some properties of metals & some properties of nonmetals • Staircase: – dividing line between metals & nonmetals – elements to left are metals (except H) – elements to right are non-metals


Most elements are solid at room temperature • Some nonmetals are gases – All the noble gases, of course – Some diatomics (H 2 N 2 O 2 F 2 Cl 2) • There are only 2 Liquids at STP (room temp) – Br 2 (non-metal) and Hg (metal)

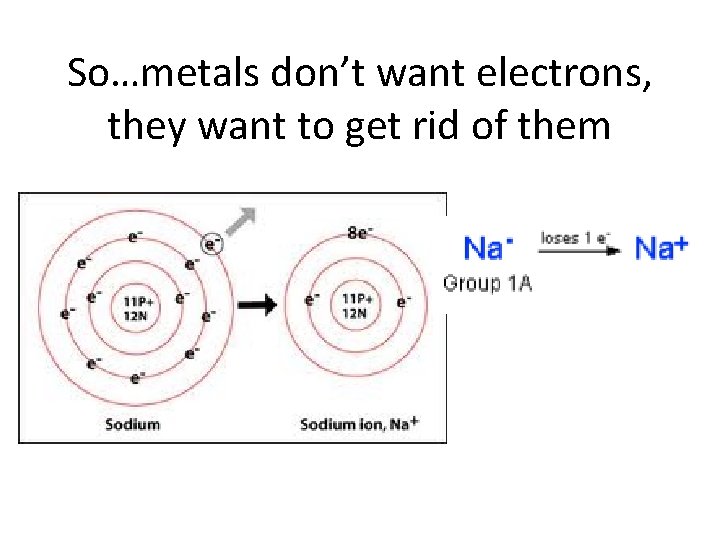
Properties of Metals • • • Malleable – flattened into sheets Ductile – drawn into wires & tubes have Luster Good Conductors of heat & electricity Solid at room temperature (except Hg) Metals lose electrons & form positive ions “Metals are losers” • Most reactive metal is Fr • Most reactive family is Alkali Metals

So…metals don’t want electrons, they want to get rid of them

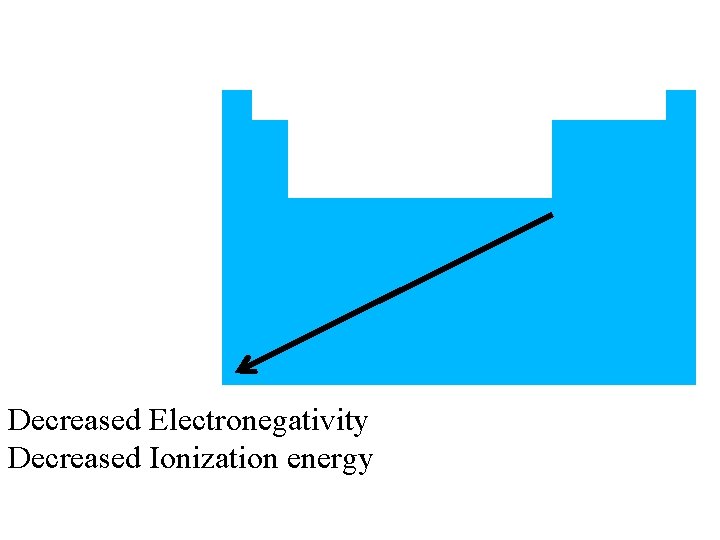
Metals also have… • Low electronegativity (low attraction for electrons) • Low ionization energy (doesn’t take a lot of energy to remove an electron)

Decreased Electronegativity Decreased Ionization energy

Properties of Nonmetals • • • generally gases or solids (except Br 2) solids are Brittle solids are Dull poor conductors of heat & electricity Nonmetals gain electrons & form negative ions “Nonmetals are winners” • Most reactive nonmetal is F – Properties: OPPOSITE of metals

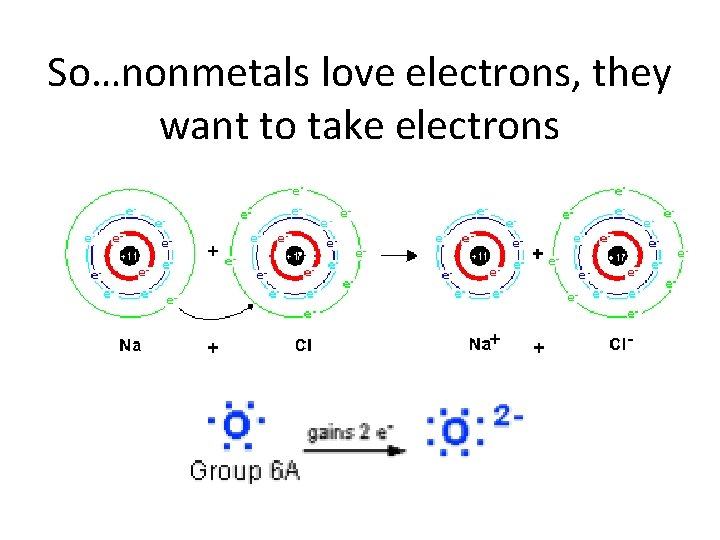
So…nonmetals love electrons, they want to take electrons

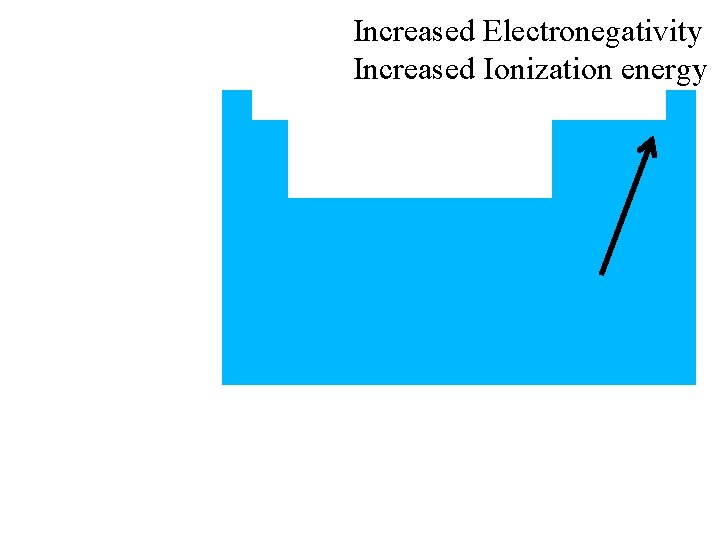
Nonmetals also have… • high electronegativity (high attraction for electrons) • high ionization energy (takes a lot of energy to remove an electron)

Increased Electronegativity Increased Ionization energy

Properties of Metalloids 7 metalloids: – 5 on right of staircase: B, Si, As, Te, At – 2 on left of staircase: Ge, Sb Each metalloid has some metallic and some nonmetallic properties – Example: Si Metalloids are elements touching the stairs • shiny like metal but brittle like nonmetal (except Al) and have some properties of metals and some of nonmetals

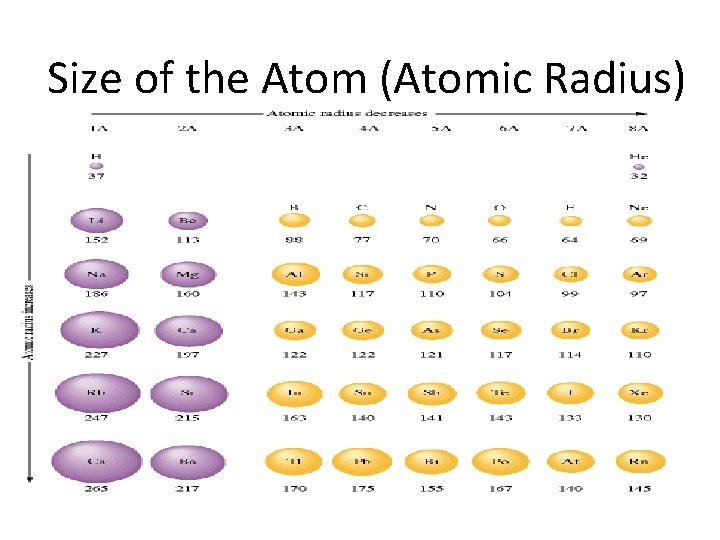
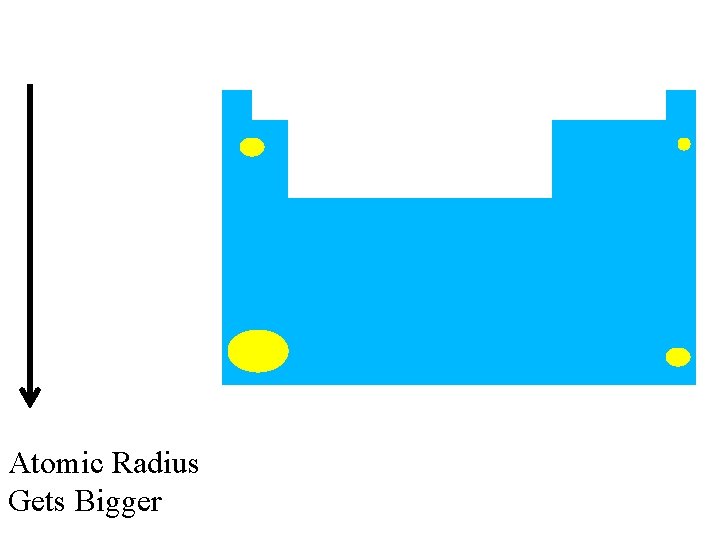
Size of the Atom (Atomic Radius)

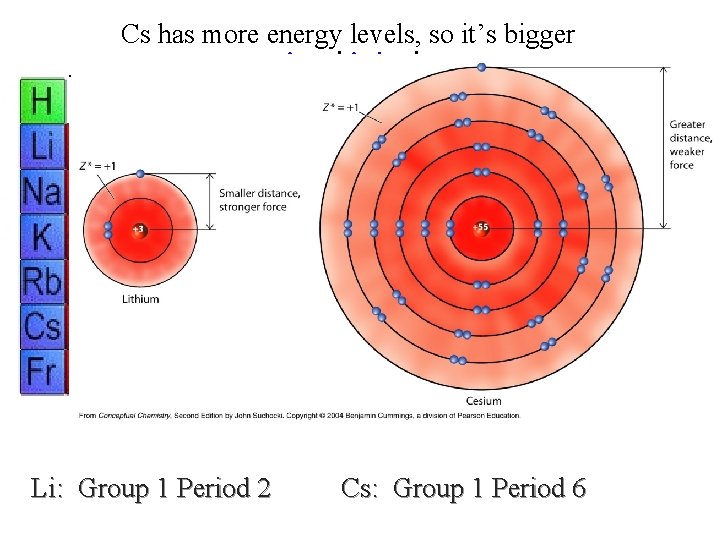
Cs has more energy levels, so it’s bigger previous | index | next Li: Group 1 Period 2 Cs: Group 1 Period 6

Atomic Radius Gets Bigger

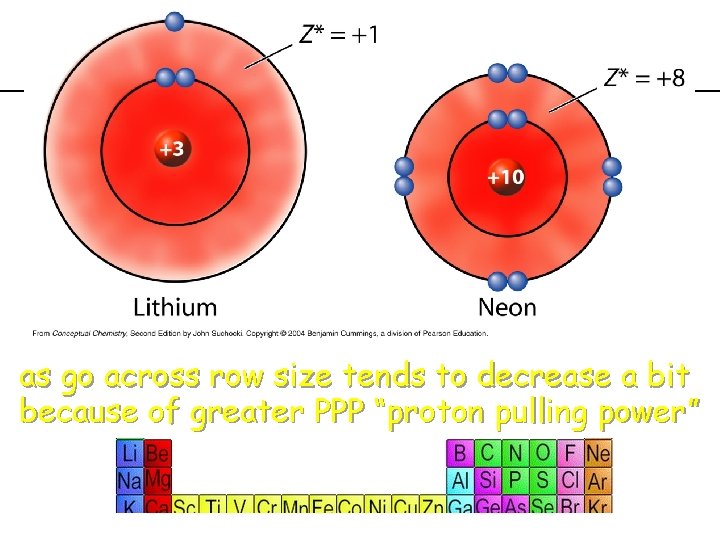
previous | index | next How about as you go across? !

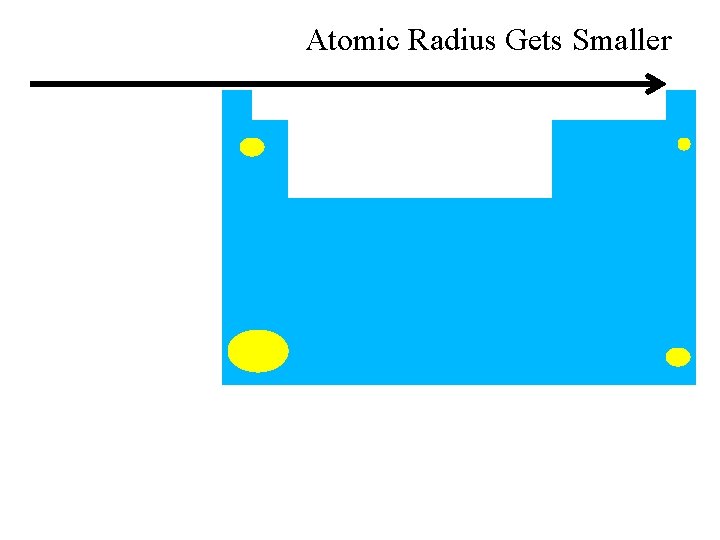
previous | index | next across row size tends to decrease as go a bit because of greater PPP “proton pulling power”

Atomic Radius Gets Smaller