The Periodic Table Mendeleev Dmitri Mendeleev 1869 Russian

The Periodic Table

Mendeleev Dmitri Mendeleev (1869, Russian) Organized elements by increasing atomic mass. Elements with similar properties were grouped together. There were some discrepancies.

Mendeleev Dmitri Mendeleev (1869, Russian) Predicted properties of undiscovered elements.

Moseley Henry Moseley (1913, British) Organized elements by increasing atomic number. Resolved discrepancies in Mendeleev’s arrangement. Periodic Law-the physical and chemical properties of the elements are periodic functions of the atomic numbers.

Metalloids Metals Non-Metals
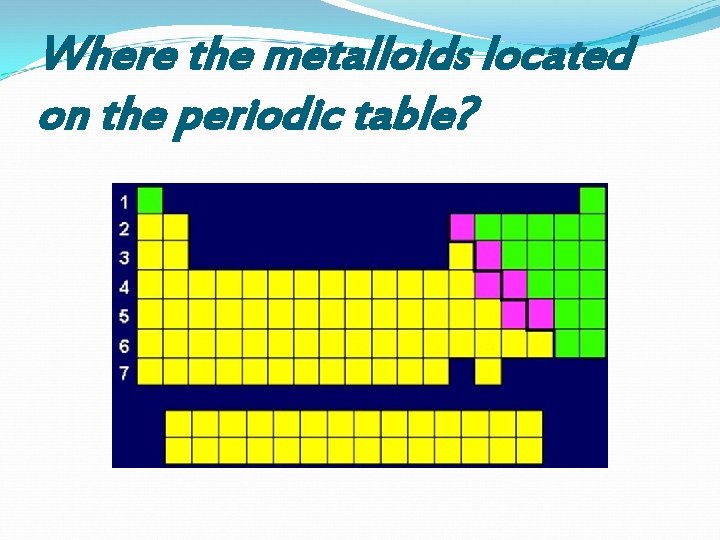
Where the metalloids located on the periodic table?

Properties of Metalloids Metallic appearance Brittle and only fair conductors of electricity Behave most like nonmetals Can form alloys with metals (example brass) Most of their physical and chemical properties are intermediate in nature Located along stairstep B, Si, Ge, As, Sb, Te are the most recognized metalloids
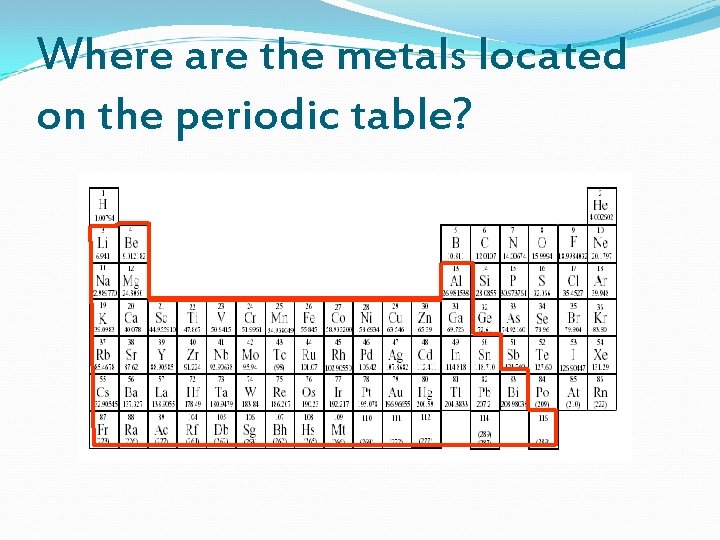
Where are the metals located on the periodic table?

Properties of Metals Shiny Conduct heat & electricity Malleable (bendable) Ductile (can be bent into wires) Mostly solids(exception: Mercury) Located in Groups 1 -12 Family names include: Alkali Metals, Alakaline Earth Metals& Transition Metals
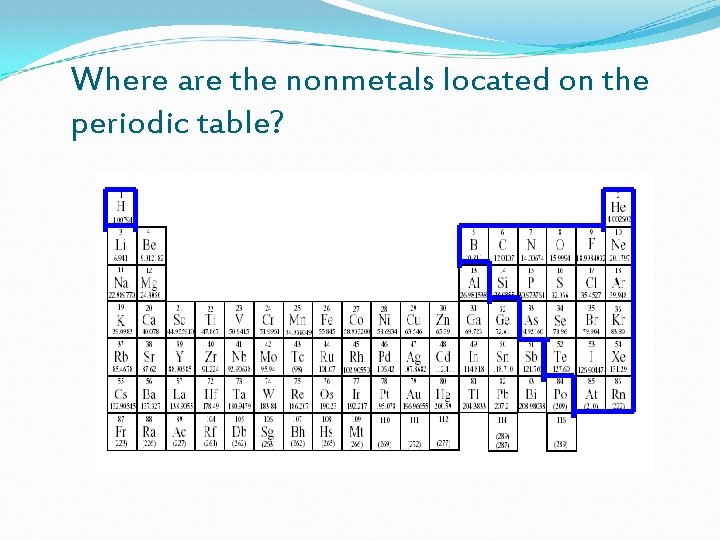
Where are the nonmetals located on the periodic table?

Properties of Non-Metals Brittle Can be solids, liquids, and are mostly Gases Do not conduct heat & electricity Located in Groups 13 -18 Family names include: Halogens & Noble Gases

Families on the Periodic Table Elements on the periodic table can be grouped into families bases on their chemical properties. Each family has a specific name to differentiate it from the other families in the periodic table. Elements in each family react differently with other elements.

ALKALI METALS Group 1 1 valence electron Reacts explosively with water Shiny, silver, soft, can be cut with a butter knife Because of extreme reactivity with moisture, usually stored under kerosene I
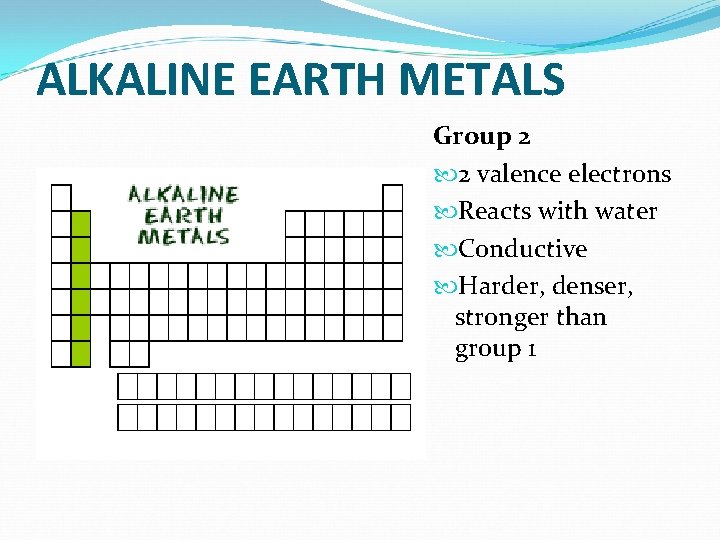
ALKALINE EARTH METALS Group 2 2 valence electrons Reacts with water Conductive Harder, denser, stronger than group 1

Halogens (Halides) Group 17 7 valence electrons Non-metals Corrosive Used for salts (called halides when bonded to a group 1 metal) Solids (I and At) Liquid (Br) Gases (Cl, F)
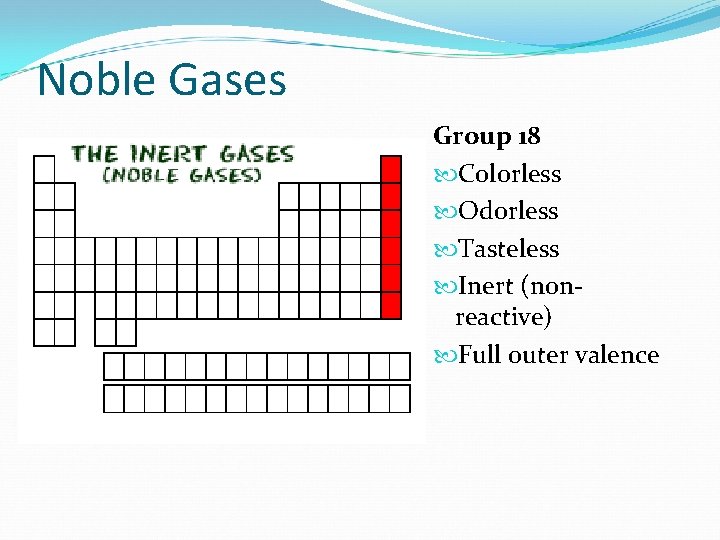
Noble Gases Group 18 Colorless Odorless Tasteless Inert (nonreactive) Full outer valence
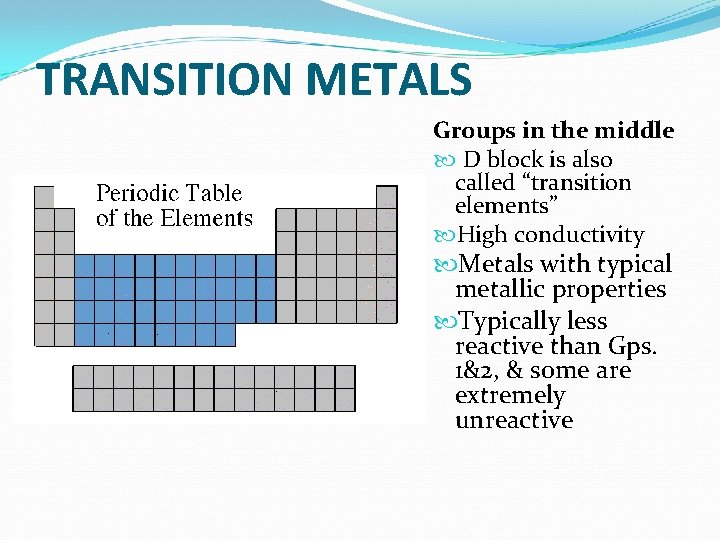
TRANSITION METALS Groups in the middle D block is also called “transition elements” High conductivity Metals with typical metallic properties Typically less reactive than Gps. 1&2, & some are extremely unreactive
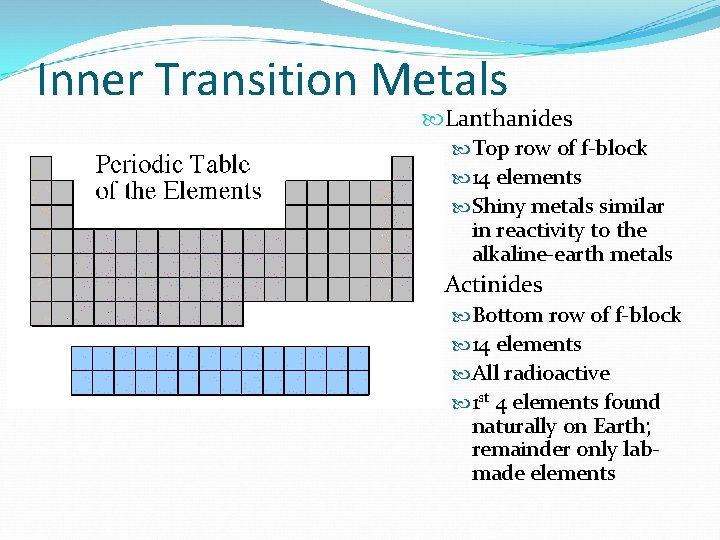
Inner Transition Metals Lanthanides Top row of f-block 14 elements Shiny metals similar in reactivity to the alkaline-earth metals Actinides Bottom row of f-block 14 elements All radioactive 1 st 4 elements found naturally on Earth; remainder only labmade elements
- Slides: 18