The Periodic Table Metals Nonmetals Semimetals Metals Nonmetals

The Periodic Table
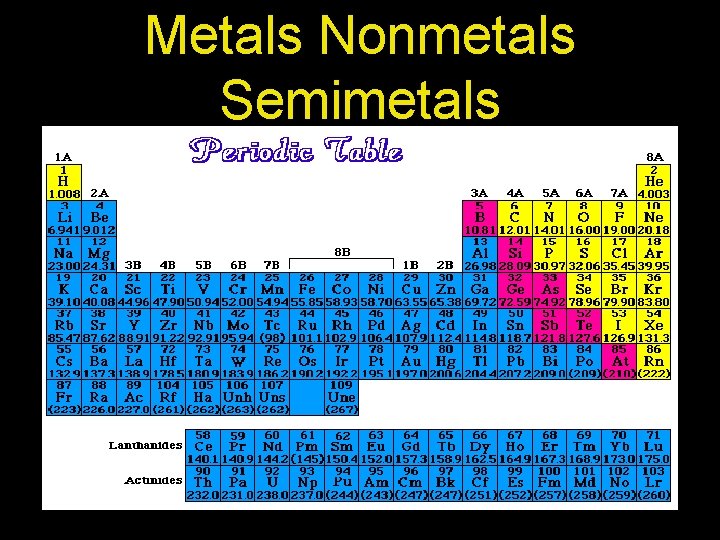
Metals Nonmetals Semimetals

Metals Nonmetals Semimetals • Metals- hard, shiny, conductors, react with acids • Non metals- typically gases, brittle if solid, bad conductors, rarely react with acids

Metals Nonmetals Semimetals • Semi-metals- have both metal and non metal properties, sometimes look like they contradict each other. Ex: Silicon- brittle but shiny and conducts electricity

History of its Development • Johann Dobereiner-1817, German

History of its Development • Johann Dobereiner-1817, German • Discovered that Ca, Ba, Sr had similar properties so he grouped them into a “Triad” in order of mass

Dobereiner • Triad 1 Ca: Sr: Ba • Triad 2 Cl: Br: I • Triad 3 S: Se: Te • *He noticed that the middle element’s mass was very close to the avg. of all 3.

John Newlands, 1863 -65 • 62 known elements

John Newlands, 1863 -65 • 62 known elements • Arranged them in order of increasing atomic mass and noticed a repetition of th properties every 8 element.

John Newlands, 1863 -65 • His P. T. is grouped into a period of 7 elements stacked into rows. • He called it the Law of Octaves

John Newlands, 1863 -65 • Li Be B C N O F • Na Mg Al Si P S Cl

Dmitri Mendeleev, 1869 • Russian Teacher

Dmitri Mendeleev, 1869 • Russian Teacher • Suggested that properties of elements were dependent upon atomic mass (wrong).

Dmitri Mendeleev, 1869 • Also noticed that some elements appear to be in wrong row because they did not have similar properties: Te & I, Co & Ni, Ar & K.

Dmitri Mendeleev, 1869 • He arranged some elements out of their atomic mass order so the properties would align vertically.

Dmitri Mendeleev, 1869 • He left gaps in his table when properties didn’t align, predicting that a yet to be discovered element belonged there. Ex: Ge, discovered in 1886.

Henry Moseley 1913 • Explored the nucleus with Xrays. ,

Henry Moseley 1913 • Explored the nucleus with Xrays. , • Discovered that the “atomic number” is equal to the number of protons.

Henry Moseley 1913 • He ordered the elements by atomic number (not mass or properties) and found that the properties still matched up vertically. • This was the Modern P. T. • Periodic Law: properties of elements are determined by their atomic number.

Families of the Periodic Table • Family= group, vertical row • Group 1 A Alkali Metals –Li Na K Rb Cs Fr

Families of the Periodic Table • Alkali Metals Properties-shiny, malleable, very reactive to water, Reactivity increases down the group

Families of the Periodic Table • Alkaline Earth Metals- Be Mg Ca Sr Ba Ra are shiny, malleable, moderately reactive, slight radioactivity in Ra and Ba

Families of the Periodic Table • Boron Family-B to Tl, have both metallic and nonmetallic properties • Carbon Family- C to Pb, metals and nonmetals, Pb & Sn can be toxic

Families of the Periodic Table • Nitrogen Family-N to Bi; nonmetals and metals As, Sb, Bi poisionous • Chalcogens- O to Po, all nonmetals e/c Po, Te

Families of the Periodic Table • Halogens- F to At, gas, liquid and solid , most reactive nonmetals • Transition Metals- Groups 3 to 12 elements, very versatile


Families of the Periodic Table • Lanthanides & Actinides elements 57 to 70; 89 to 102 - contain synthetic elements, most are radioactive

Families of the Periodic Table • This family goes through transmutation more readily than other families

Families of the Periodic Table • Transmutation- the process of spontaneously changing into another element

Trends in the Periodic Table • Called “Periodicity” • Atomic Radius- increases down the PT and increases as you go left. • Biggest Atom? • Smallest Atom?

Trends in the Periodic Table Ionization Energy-energy needed to remove a valence electron Increases as you go up and right--opposite of atomic size

Trends in the Periodic Table Electron Affinity- ability to attract an electron from another atom Increases as you go up and right-same as I. E.

Trends in the Periodic Table Electronegativity- rating scale indicating an element’s ability to attract electrons Trend: same as I. E.

Trends in the Periodic Table
- Slides: 34