The Periodic Table Metals Nonmetals and Metalloids The

The Periodic Table: Metals, Nonmetals, and Metalloids

The Periodic Table • Created in 1869 by both Mendeleev and Meyers • Mendeleev periodic table more widely accepted • Organized by – Atomic mass – Chemical properties 1/20/2022 2

The Periodic Table • Organized in regular, repeating patterns known as periodicity • Elements in the same column or row have similar properties or patterns 1/20/2022 3

The Periodic Table • Group or Family – A vertical column on the periodic table – Similar chemical and physical properties • Period – A horizontal row on the periodic table – Properties change progressively across 1/20/2022 4

Mendeleev’s Periodic Table 1/20/2022 5

Mendeleev’s Periodic Table • Based upon the repeating patterns, Mendeleev left blank spaces on his periodic table • These spaces were eventually filled in with undiscovered elements • Discovered elements had similar properties compared to their group 1/20/2022 6

• Organized in order of increasing atomic mass 1/20/2022 7

Mendeleev’s Periodic Table • Flaws in this periodic table: – As mass increased, the properties did not all line up – Led to confusion of properties of known elements – Was solved with the discovery of the atomic number 1/20/2022 8

Modern Periodic Table • Organized by atomic number and properties • This led to the “periodic law” which states: – The properties of the elements are periodic functions of their atomic number 1/20/2022 9

Modern Periodic Table • Developed by British scientist Henry Mosley • Arranged by increasing atomic number • Determined elements fell into columns with other elements with similar properties – There were no exceptions found 1/20/2022 10
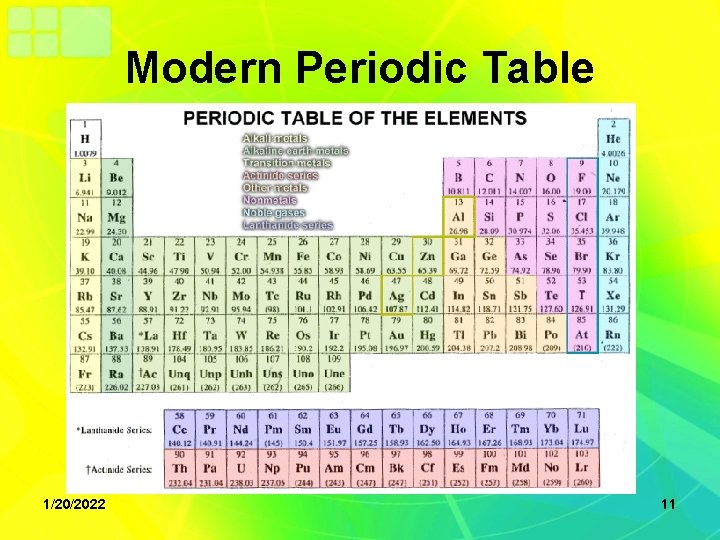
Modern Periodic Table 1/20/2022 11

Periodic Families • There are 10 main families on the periodic table – Metals – Alkali metals – Alkaline earth metals – Transition metals – Inner-transition metals 1/20/2022 12

Periodic Families • There are 10 main families of the periodic table – Other metals – Metalloids – Nonmetals – Halogens – Noble Gases 1/20/2022 13

Metals • • Located below the stair step line Form cations Solids Shiny metallic appearance Malleable Ductile Conductive High melting point and densities 1/20/2022 14

Metalloids • Located on the stair step line • Have properties of both metals and nonmetals • Form both cations and anions • Make good semiconductors • Does NOT include aluminum (other metal) 1/20/2022 15

Nonmetals • • • Located above the stair step line Form anions Solids, liquids and gases Poor conductors Brittle Not malleable or ductile 1/20/2022 16

Alkali Metals • • • Located in Group 1 Low density 1 valence electron Forms a +1 cation Highly reactive in water 1/20/2022 17

Alkaline Earth Metals • • • Located in Group 2 Harder and denser than alkali metals 2 valence electrons Forms a +2 cation Reducing agents (reactive) 1/20/2022 18

Transition Metals • • Located in the “d” block High densities and melting points White lustrous metals (except copper) Form cations 1/20/2022 19

Inner-transition Metals • • • Located in “f” block Also known as the rare earth metals Most are synthetic (man made) Most are radioactive Form cations 1/20/2022 20

Other Metals • Located next to the transitional metals and under the stair step line • Properties vary • Form cations 1/20/2022 21

Halogens • • • Located in group 17 7 valance electrons Forms a -1 anion Highly electronegative Diatomic Very reactive with the alkali metals 1/20/2022 22

Noble Gases • • Located in group 18 8 valance electrons Do not react!!!! Also known as Inert Gases 1/20/2022 23
- Slides: 23