Organic Chemistry II Dr Christopher Cioffi Monday 3202017






![The Diels-Alder Reaction: A [4+2] Cycloaddition The Diels-Alder Reaction: A [4+2] Cycloaddition](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-12.jpg)
![The Diels-Alder Reaction: A [4+2] Cycloaddition The Diels-Alder Reaction: A [4+2] Cycloaddition](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-13.jpg)
![The Diels-Alder Reaction: [4+2] Cycloaddition § The reaction involves 4 -p electrons of the The Diels-Alder Reaction: [4+2] Cycloaddition § The reaction involves 4 -p electrons of the](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-14.jpg)








- Slides: 46

Organic Chemistry II Dr. Christopher Cioffi Monday 3/20/2017 9: 00 AM – 9: 50 AM

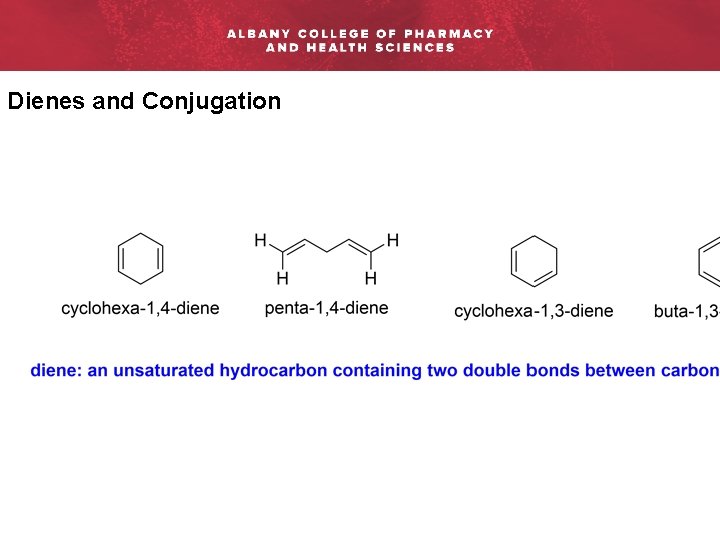
Dienes and Conjugation

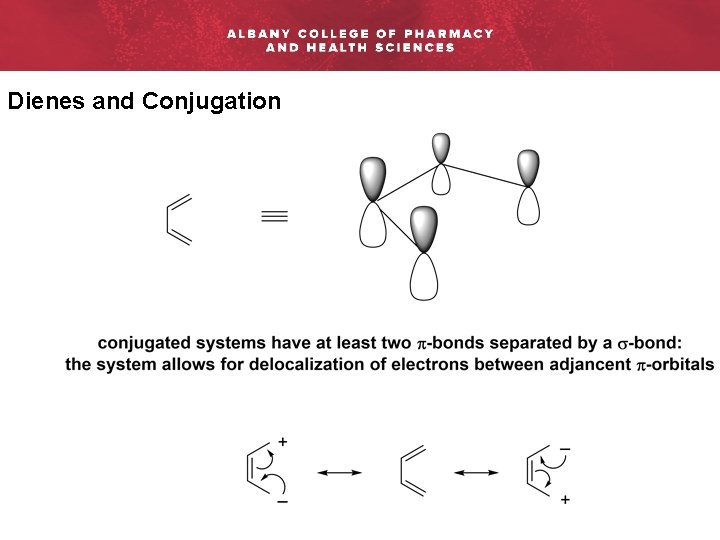
Dienes and Conjugation

Dienes and Conjugation

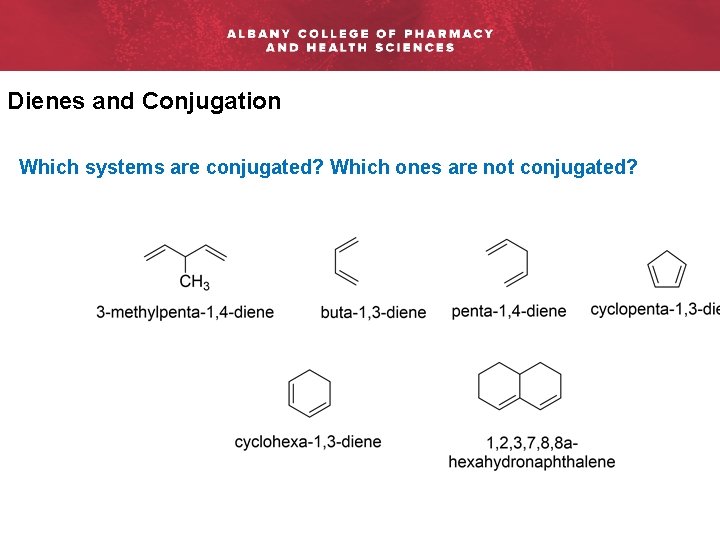
Dienes and Conjugation Which systems are conjugated? Which ones are not conjugated?

Which systems are conjugated? Which ones are not conjugated?

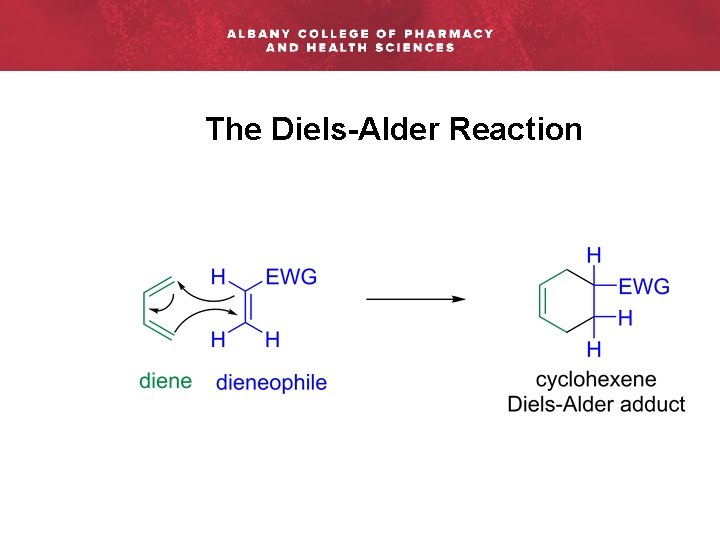
The Diels-Alder Reaction

The Diels-Alder Reaction

The Diels-Alder Reaction: Highlights § Pericyclic reaction § A concerted (one-step) reaction that proceeds via a cyclic transition state § [4+2] Cycloaddition reaction between a conjugated diene an dienophile § Forms six-membered ring systems (cyclohexene products) § Generates molecular complexity efficiently § Accommodates several functional groups, generates a new double-bond, and con stereochemistry and regiochemistry: can establish up to four stereogenic centers

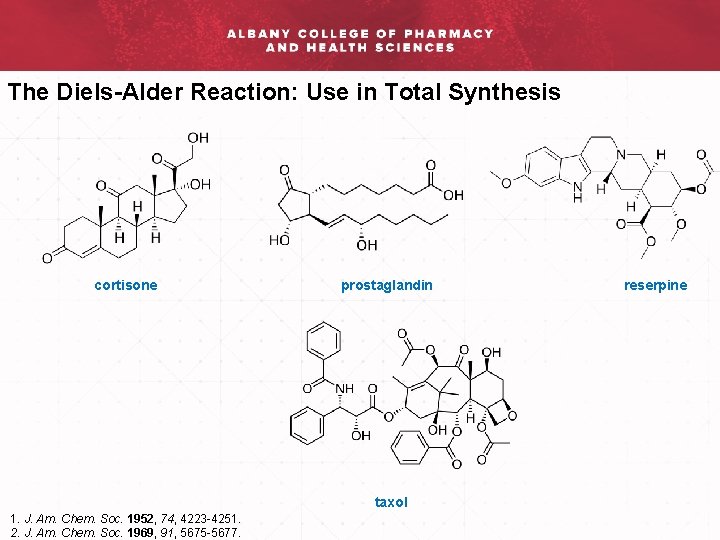
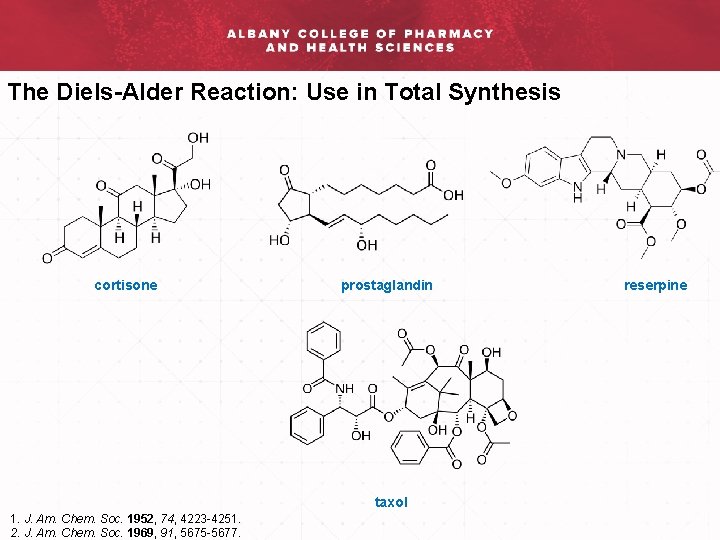
The Diels-Alder Reaction: Use in Total Synthesis cortisone prostaglandin taxol 1. J. Am. Chem. Soc. 1952, 74, 4223 -4251. 2. J. Am. Chem. Soc. 1969, 91, 5675 -5677. reserpine

The Diels-Alder Reaction: Highlights § Active Pharmaceutical Ingredients (APIs) § Agrochemicals § Flavors and fragrances Funel, J. A. ; Abele, S. ; Industrial applications of the Diels-Alder reaction. Angew. Chem. Int. Ed. Engl. 2013, 52, 3822 -3863.
![The DielsAlder Reaction A 42 Cycloaddition The Diels-Alder Reaction: A [4+2] Cycloaddition](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-12.jpg)
The Diels-Alder Reaction: A [4+2] Cycloaddition
![The DielsAlder Reaction A 42 Cycloaddition The Diels-Alder Reaction: A [4+2] Cycloaddition](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-13.jpg)
The Diels-Alder Reaction: A [4+2] Cycloaddition
![The DielsAlder Reaction 42 Cycloaddition The reaction involves 4 p electrons of the The Diels-Alder Reaction: [4+2] Cycloaddition § The reaction involves 4 -p electrons of the](https://slidetodoc.com/presentation_image_h/c66ee295a2100618e4ed407d212ca6d4/image-14.jpg)
The Diels-Alder Reaction: [4+2] Cycloaddition § The reaction involves 4 -p electrons of the diene and 2 -p electrons of the die § Converts two p-bonds into two s-bonds § Concerted reaction: all bond breaking and bond making happens simultane a single step

The Diels-Alder Reaction: Requirements

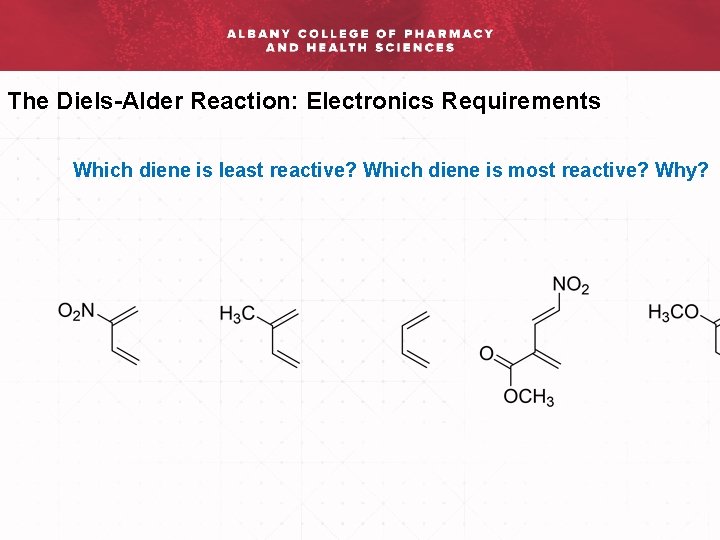
The Diels-Alder Reaction: Electronics Requirements Which diene is least reactive? Which diene is most reactive? Why?

The Diels-Alder Reaction: Electronics Requirements Which diene is least reactive? Which diene is most reactive? Why?

Which dieneophile is least reactive? Which dieneophile is most reactive

Which dieneophile is least reactive? Which dieneophile is most reactive?

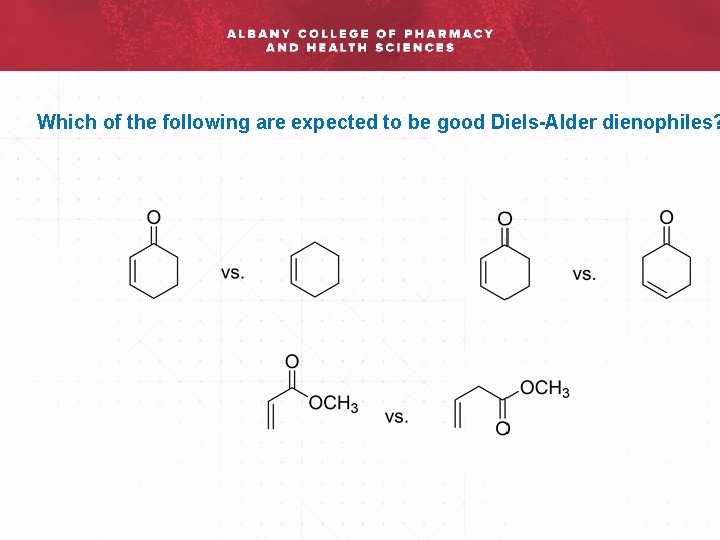
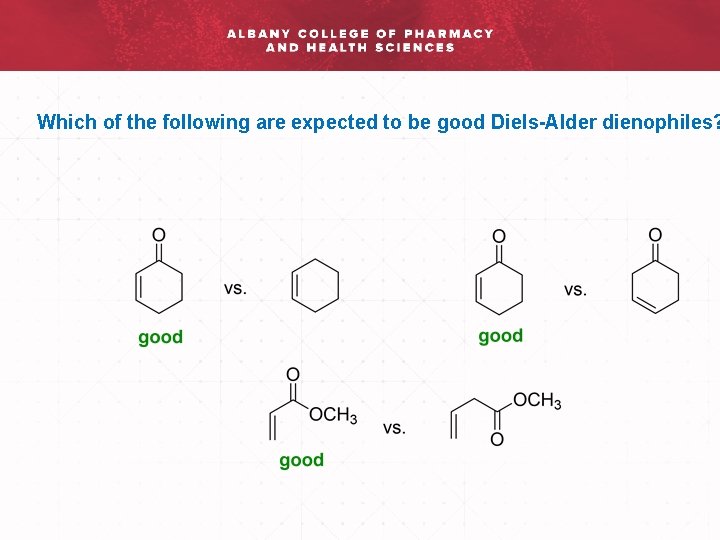
Which of the following are expected to be good Diels-Alder dienophiles?

Which of the following are expected to be good Diels-Alder dienophiles?

Diene Requirement: Conjugation and s-Cis Conformation

Diene Requirement: Conjugation and s-Cis Conformation

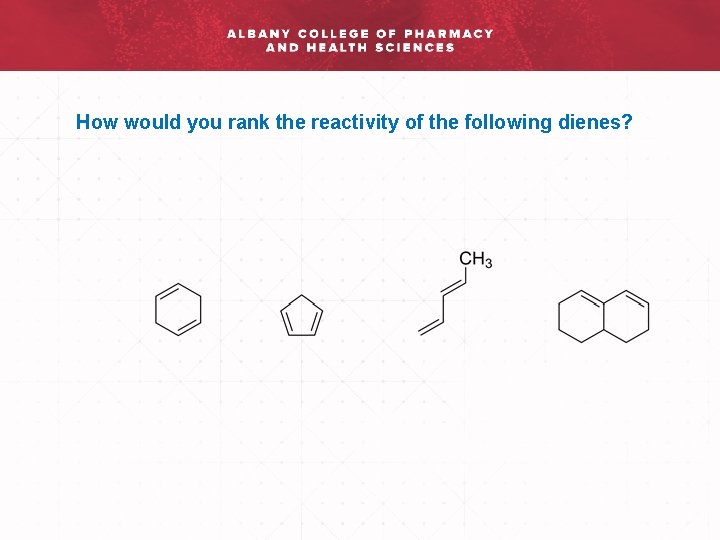
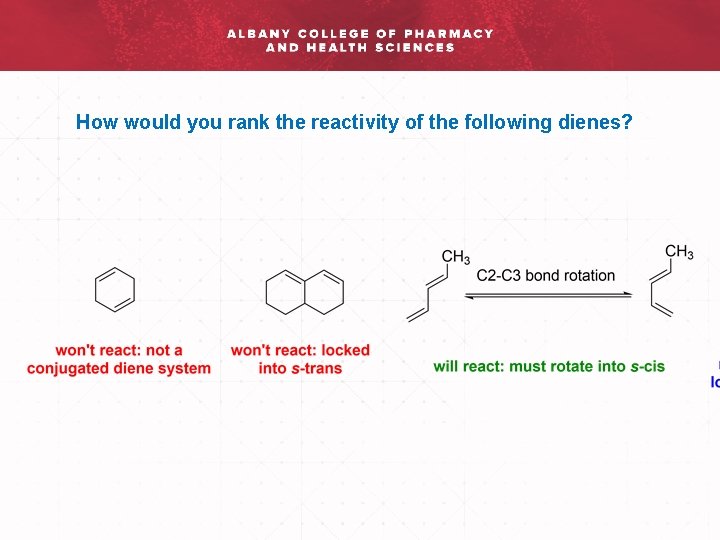
How would you rank the reactivity of the following dienes?

How would you rank the reactivity of the following dienes?

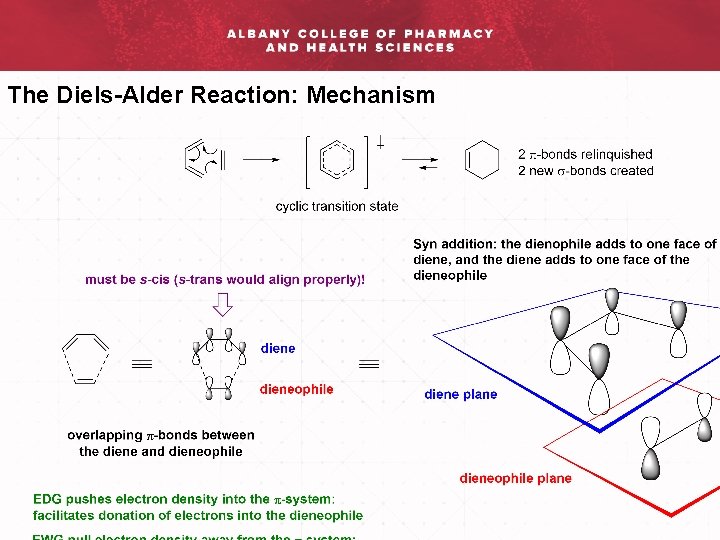
The Diels-Alder Reaction: Mechanism

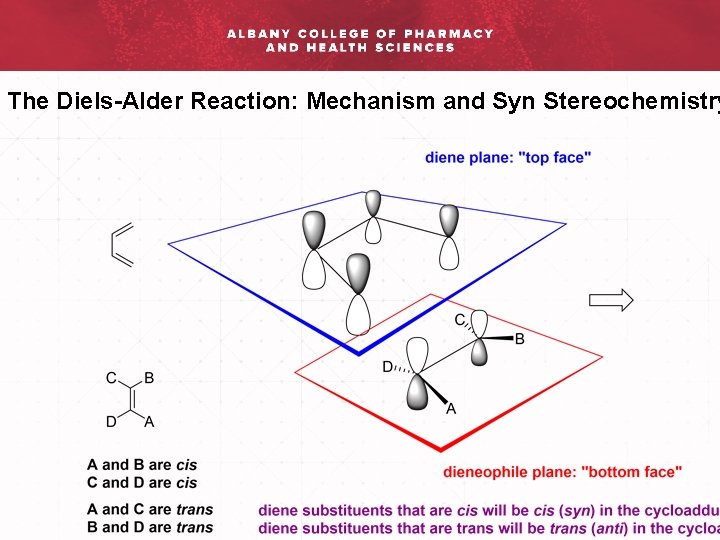
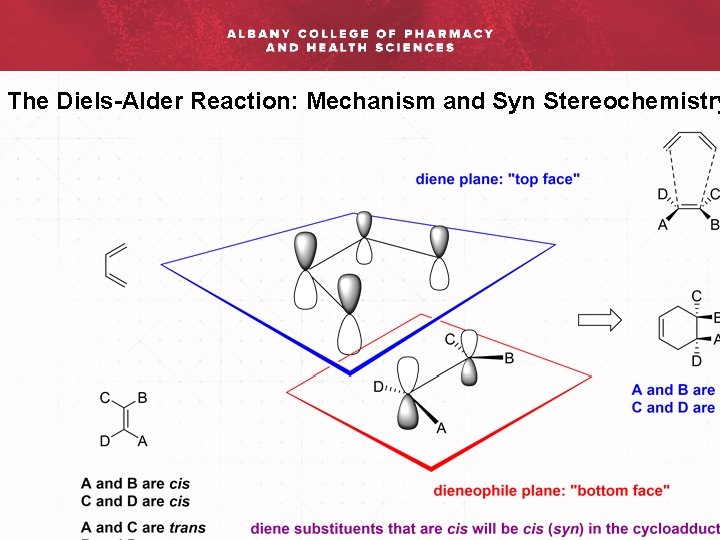
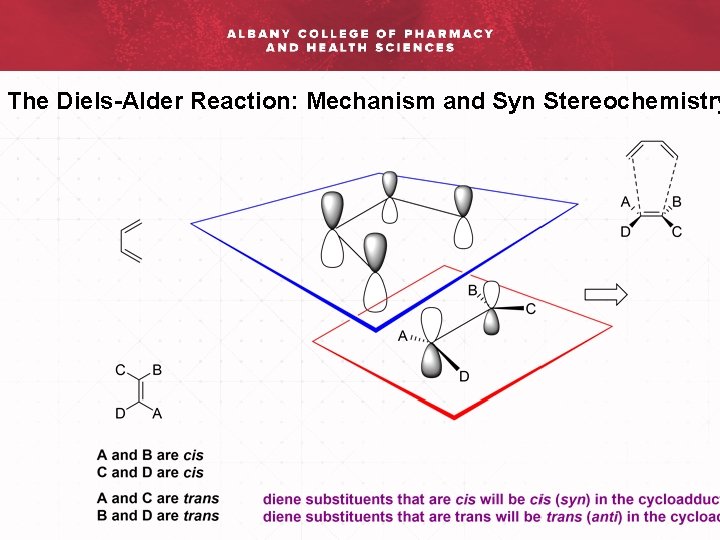
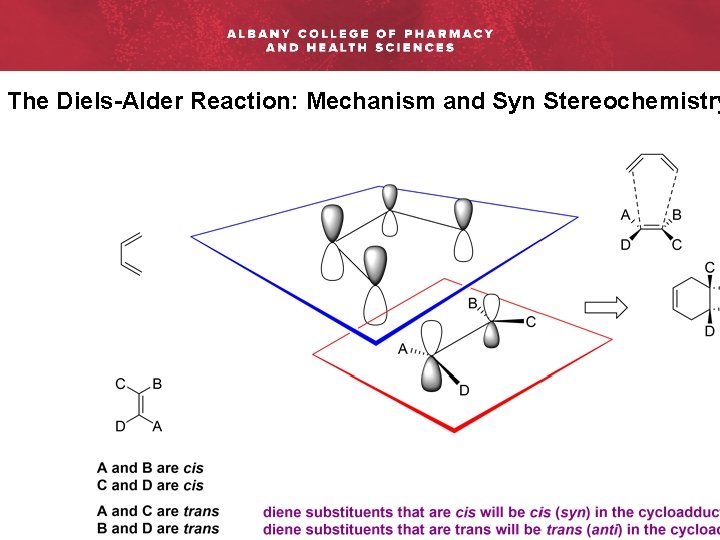
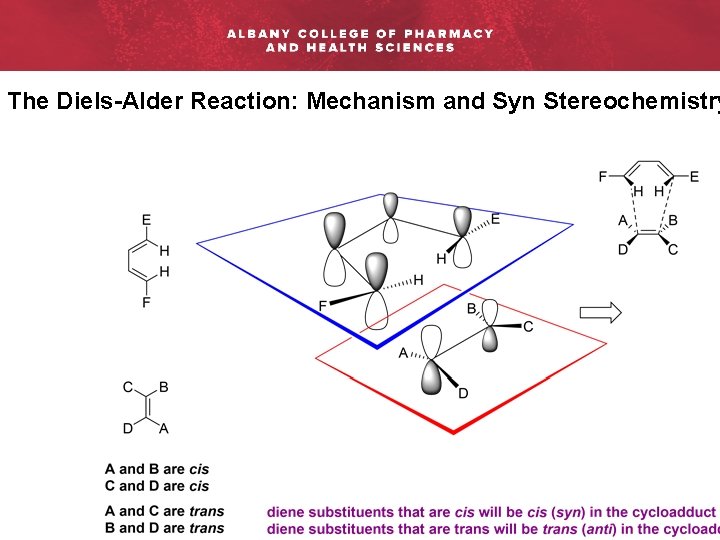
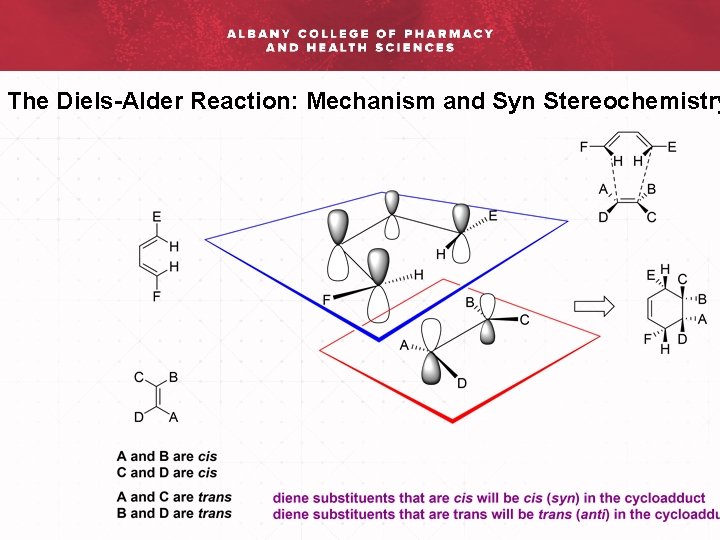
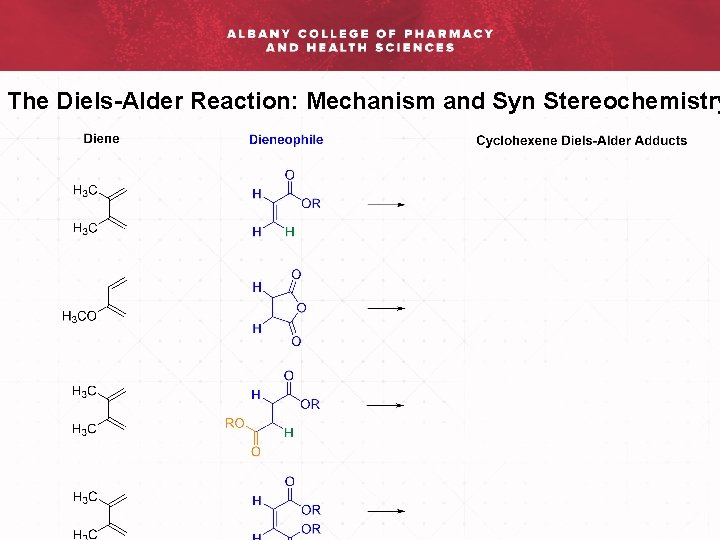
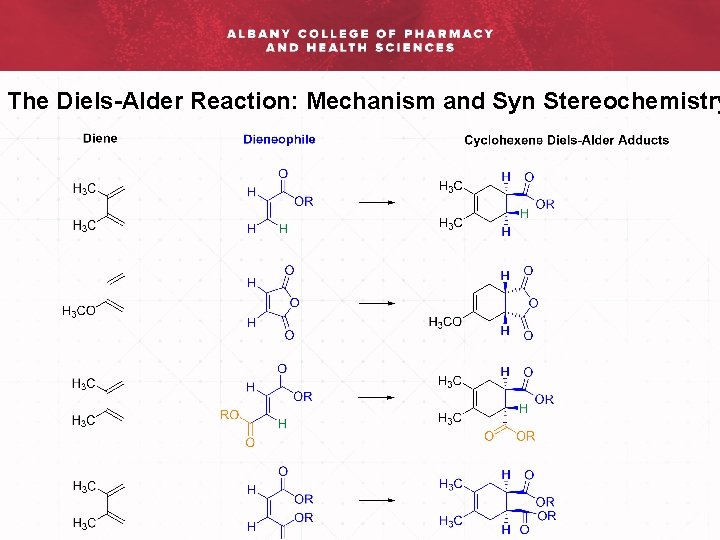
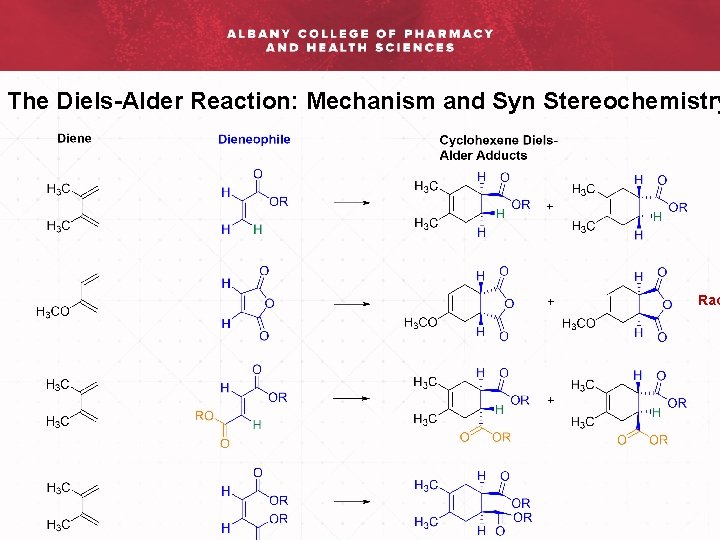
The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry

The Diels-Alder Reaction: Mechanism and Syn Stereochemistry Rac

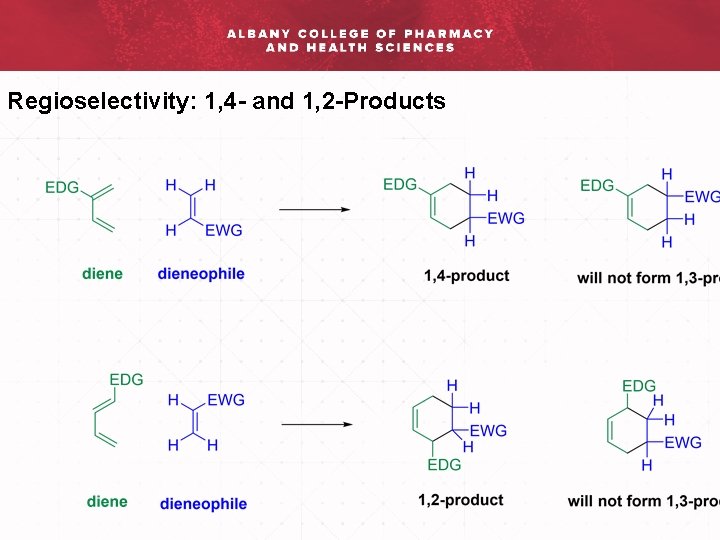
Regioselectivity: 1, 4 - and 1, 2 -Products

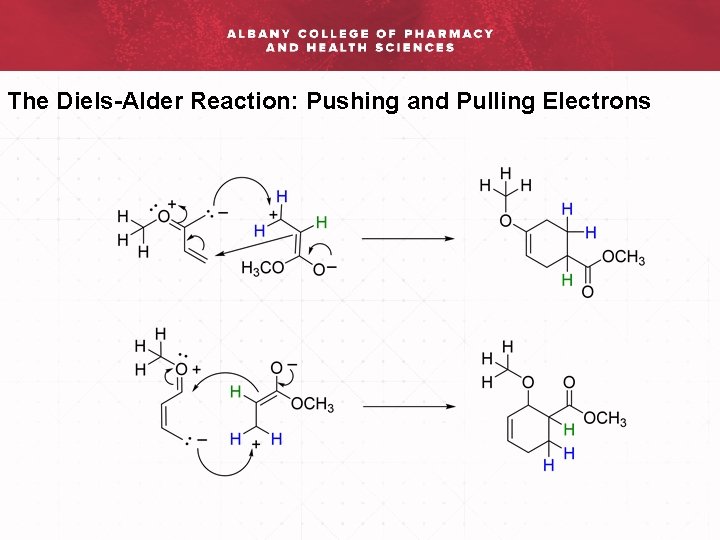
The Diels-Alder Reaction: Pushing and Pulling Electrons via Re


The Diels-Alder Reaction: Pushing and Pulling Electrons

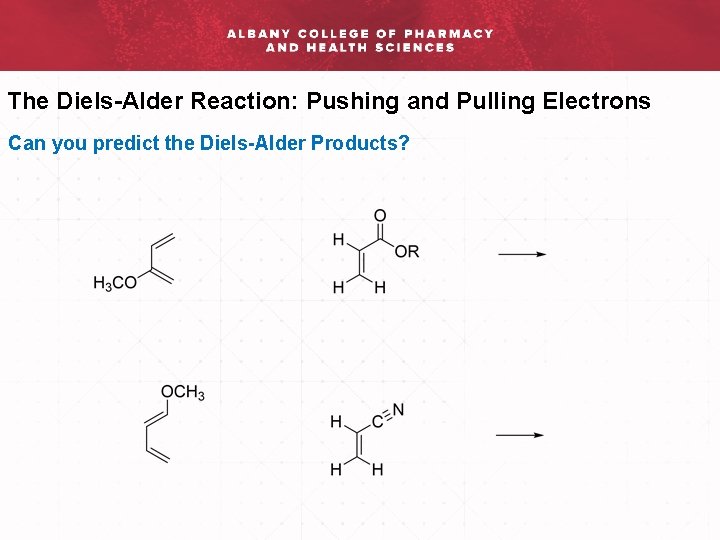
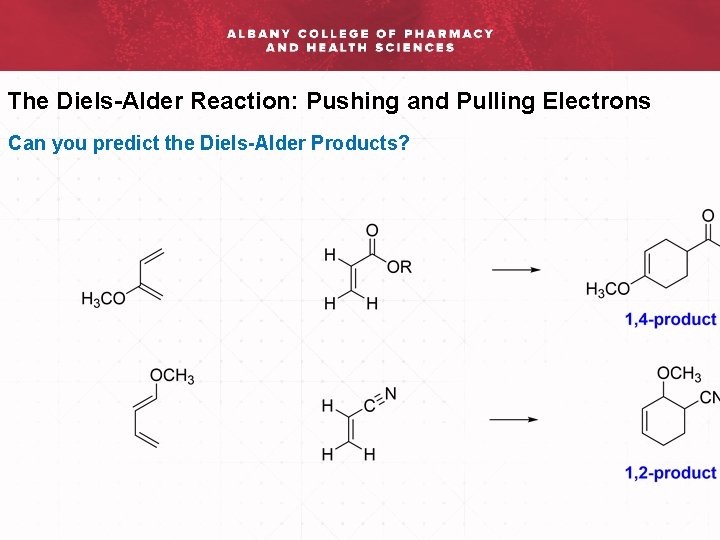
The Diels-Alder Reaction: Pushing and Pulling Electrons Can you predict the Diels-Alder Products?

The Diels-Alder Reaction: Pushing and Pulling Electrons Can you predict the Diels-Alder Products?

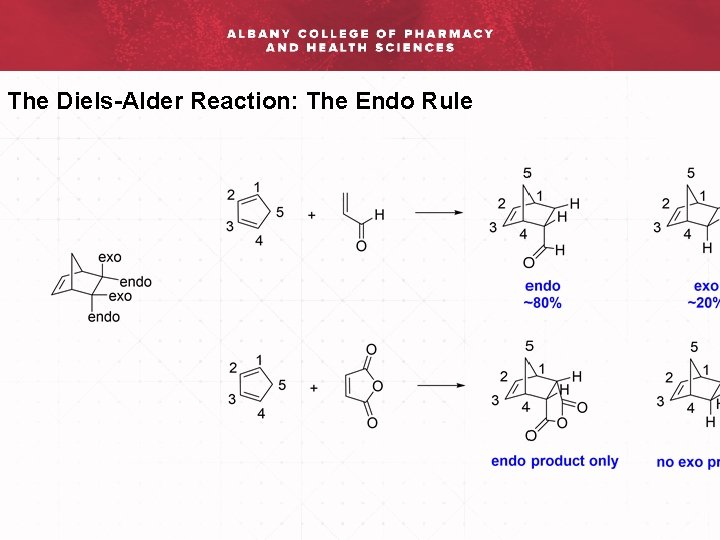
The Diels-Alder Reaction: The Endo Rule

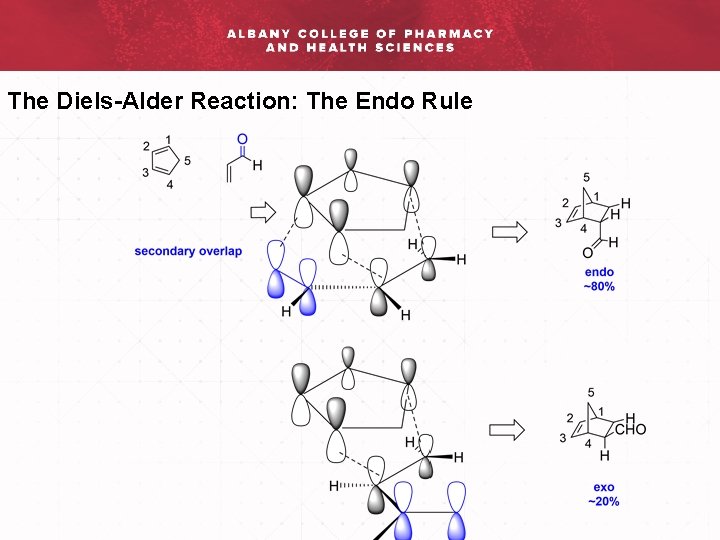
The Diels-Alder Reaction: The Endo Rule

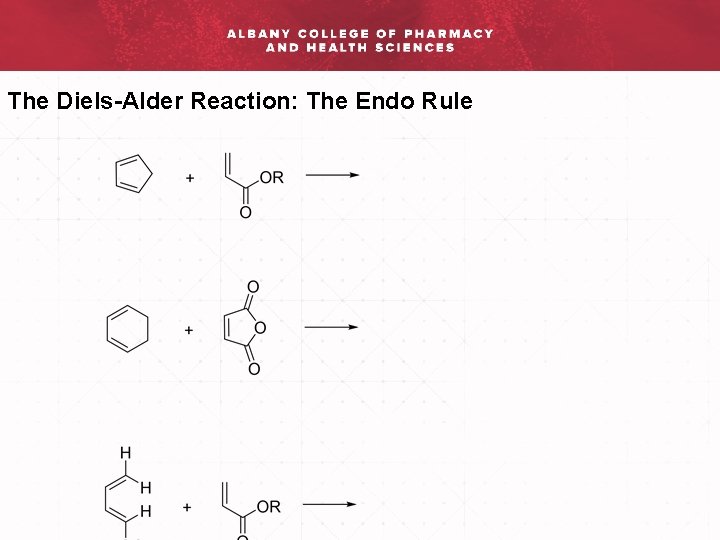
The Diels-Alder Reaction: The Endo Rule

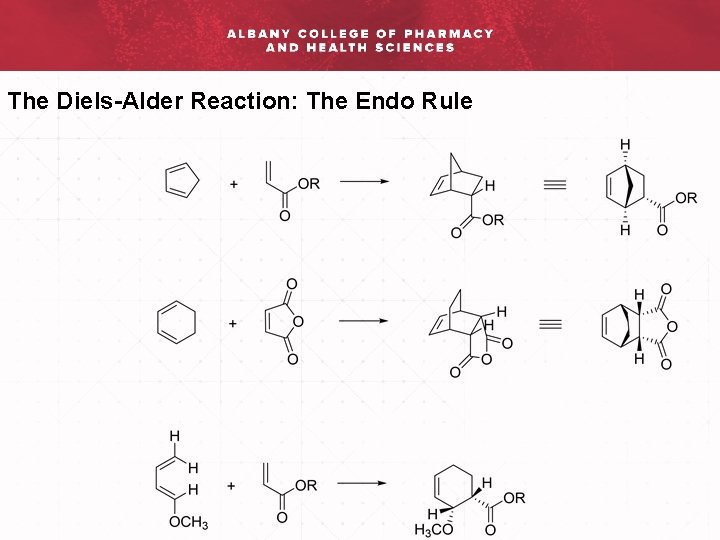
The Diels-Alder Reaction: The Endo Rule

The Diels-Alder Reaction: Use in Total Synthesis cortisone prostaglandin taxol 1. J. Am. Chem. Soc. 1952, 74, 4223 -4251. 2. J. Am. Chem. Soc. 1969, 91, 5675 -5677. reserpine