NUCLEAR CHEMISTRY Chapter 21 Introduction to Nuclear Chemistry























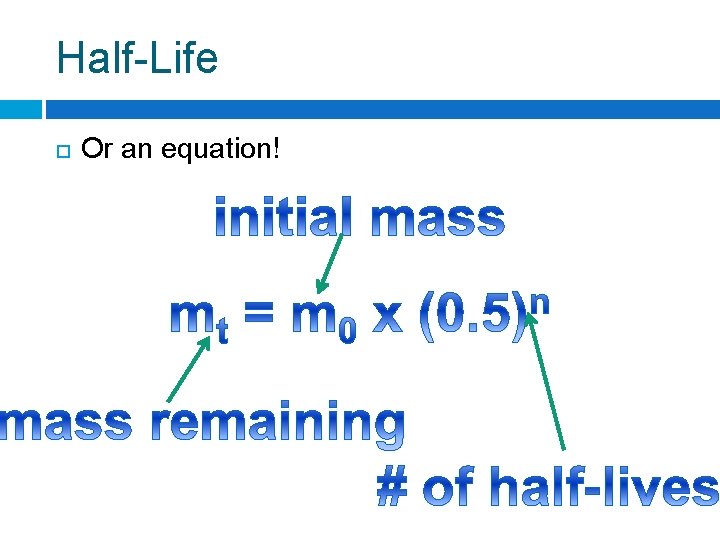











- Slides: 47

NUCLEAR CHEMISTRY Chapter 21

Introduction to Nuclear Chemistry Nuclear chemistry is the study of the structure of and the they undergo. atomic nuclei changes

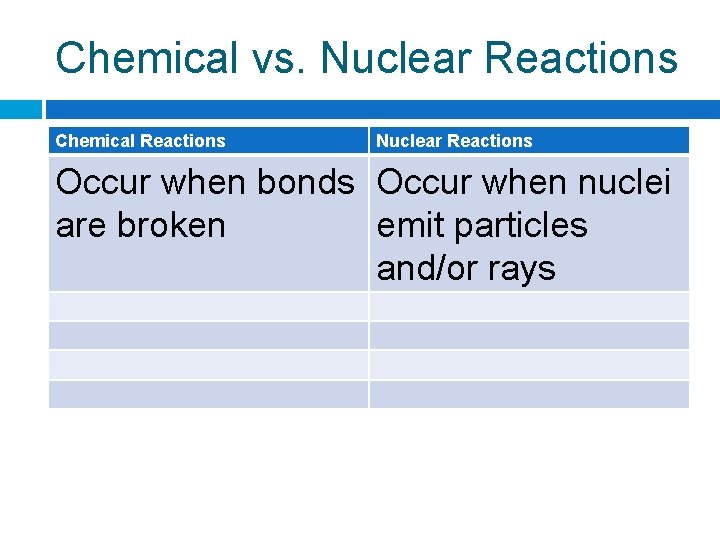
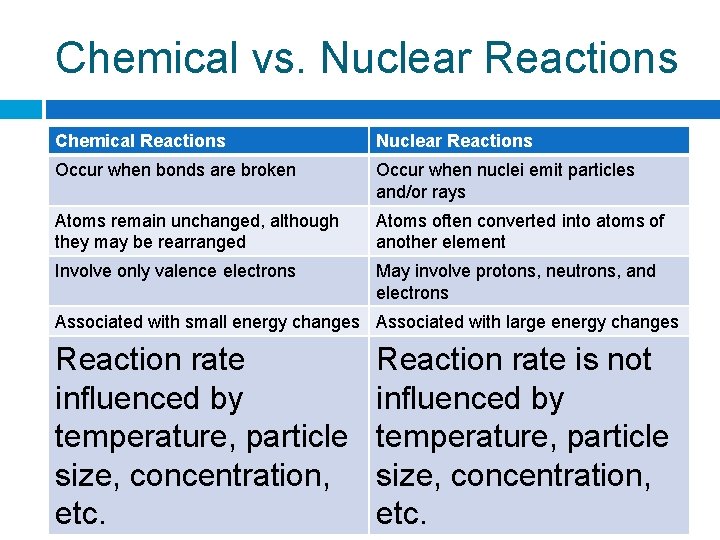
Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds Occur when nuclei are broken emit particles and/or rays

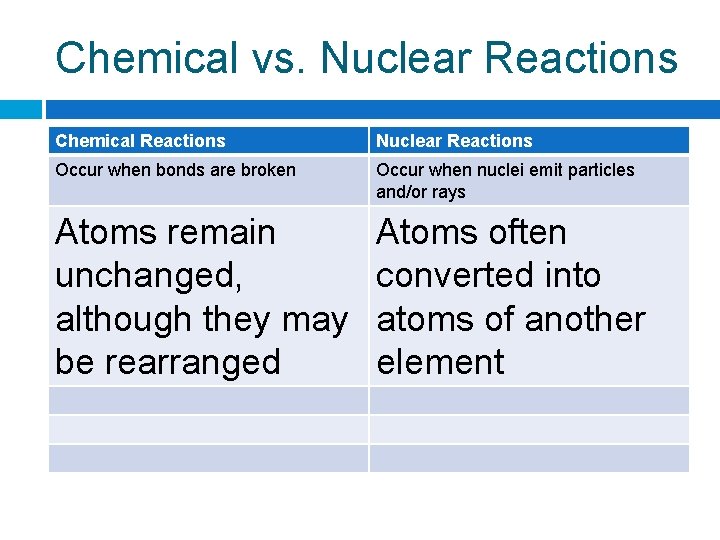
Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element

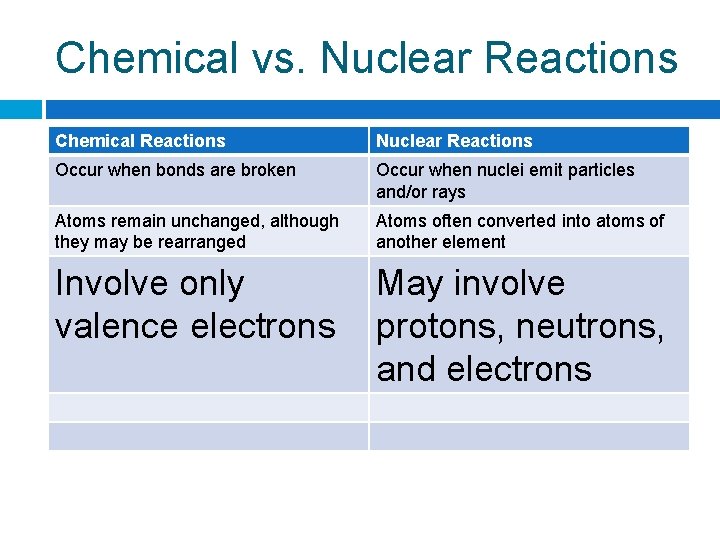
Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons

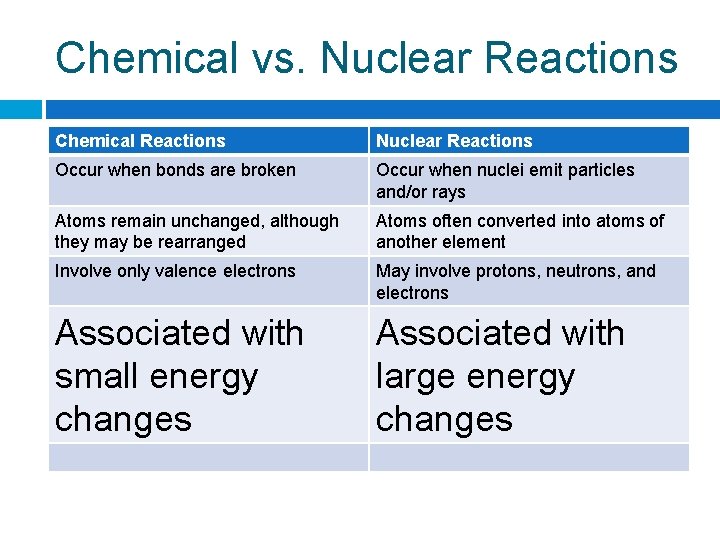
Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons Associated with small energy changes Associated with large energy changes

Chemical vs. Nuclear Reactions Chemical Reactions Nuclear Reactions Occur when bonds are broken Occur when nuclei emit particles and/or rays Atoms remain unchanged, although they may be rearranged Atoms often converted into atoms of another element Involve only valence electrons May involve protons, neutrons, and electrons Associated with small energy changes Associated with large energy changes Reaction rate influenced by temperature, particle size, concentration, etc. Reaction rate is not influenced by temperature, particle size, concentration, etc.

The Discovery of Radioactivity (1895 – 1898): found that invisible rays were emitted when electrons bombarded the surface of certain materials. Becquerel accidently discovered that phosphorescent salts produced spontaneous emissions that darkened photographic plates

The Discovery of Radioactivity (1895 – 1898): isolated the components of atoms emitting the rays – process by which atoms emit – the penetrating rays and particles by a radioactive source. � Radiation can be damaging to living organisms

The Discovery of Radioactivity (1895 – 1898): polonium radium contradicted identified 2 new elements, and on the basis of their radioactivity These findings Dalton’s theory of indivisible atoms.

The Discovery of Radioactivity (1895 – 1898): – atoms of the element with different numbers of – isotopes of atoms with nuclei (too / neutrons) – when unstable nuclei energy by emitting to attain more atomic configurations ( process)

Alpha radiation Composition – Alpha particles, same as helium 4 nuclei 2 Symbol – Helium nuclei, He, α Charge –+2 Mass – 4 amu Approximate energy – 5 Me. V Penetrating power – low (0. 05 mm body tissue) Shielding – these can be blocked by paper or clothing

Beta radiation Composition – Beta particles, same as an electron Symbol – e-, β Charge – -1 Mass (amu) – 1/1837 (practically 0) Approximate energy – 0. 05 – 1 Me. V Penetrating power – moderate (4 mm body tissue) Shielding – These can be blocked by metal foil

Gamma radiation Composition – High-energy electromagnetic radiation traveling at the speed of light Symbol – γ Charge – 0 Mass (amu) – 0 Approximate energy – 1 Me. V Penetrating power – high (penetrates body easily) Shielding – These can only be blocked by lead or concrete

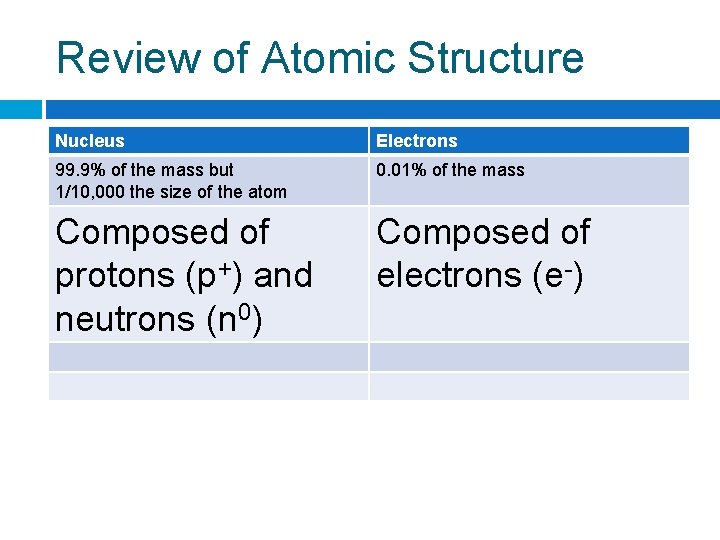
Review of Atomic Structure Nucleus Electrons 99. 9% of the mass 0. 01% of the mass but 1/10, 000 the size of the atom

Review of Atomic Structure Nucleus Electrons 99. 9% of the mass but 1/10, 000 the size of the atom 0. 01% of the mass Composed of protons (p+) and neutrons (n 0) Composed of electrons (e-)

Review of Atomic Structure Nucleus Electrons 99. 9% of the mass but 1/10, 000 the size of the atom 0. 01% of the mass Composed of protons (p+) and neutrons (n 0) Composed of electrons (e-) Positively charged Negatively charged

Review of Atomic Structure Nucleus Electrons 99. 9% of the mass but 1/10, 000 the size of the atom 0. 01% of the mass Composed of protons (p+) and neutrons (n 0) Composed of electrons (e-) Positively charged Negatively charged Strong nuclear force (holds the nucleus together) Weak electrostatic force (because they are charged negatively

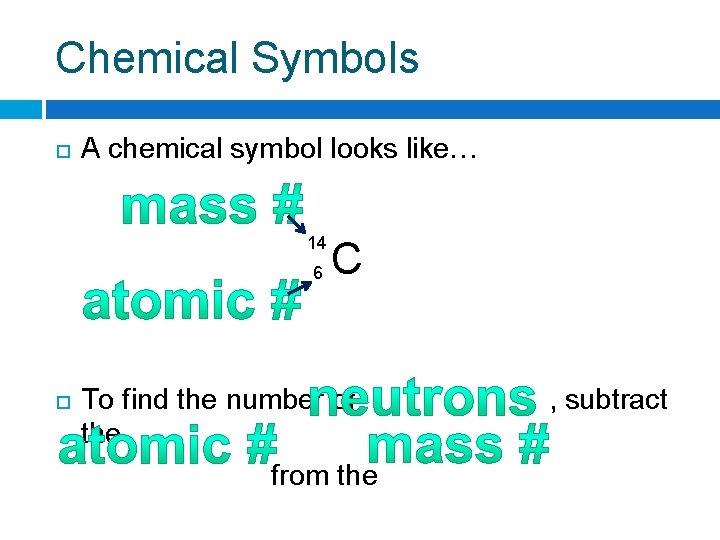
Chemical Symbols A chemical symbol looks like… 14 6 C To find the number of , subtract the from the

Nuclear Stability Isotope is completely stable if the nucleus will spontaneously . Elements with atomic #s to are ratio of protons: neutrons ( )makes an isotope very stable Example: Carbon – 12 has protons and neutrons

Nuclear Stability 21 82 marginally stable 1: 1. 5 80 Elements with atomic #s to are ratio of protons: neutrons (p+ : n 0) Example: Mercury – 200 has protons and neutrons 120

Nuclear Stability > 82 unstable radioactive Uranium Plutonium Elements with atomic #s are and . Examples: and Isotopes with greater than 1. 5 neutrons per 1 proton are unstable or radioactive

Alpha Decay Alpha decay – emission of an alpha particle ( 4 ), denoted by the symbol , because an α 2 has 2 protons and 2 neutrons, just like the He nucleus. Charge is because of the 2 . Mass of alpha particle is amu. Alpha decay causes the number to decrease by and the number to decrease by .

Alpha Decay Example 1: Write the nuclear equation for the radioactive decay of polonium – 210 by alpha emission. Step 2: Draw the arrow. Step 1: Write the element that you are starting Step 4: Determine the other product (ensuring Step 3: Write the alpha particle. with. everything is balanced). Mass # 210 84 Po Atomic # 206 82 Pb 4 2 He

Alpha Decay Example 2: Write the nuclear equation for the radioactive decay of radium – 226 by alpha emission. Step 2: Draw the arrow. Step 1: Write the element that you are starting Step 4: Determine the other product (ensuring Step 3: Write the alpha particle. with. everything is balanced). Mass # 226 88 Ra Atomic # 222 86 Rn 4 2 He

Beta decay β Beta decay – emission of a beta particle ( ), a fast moving , denoted by the 0 symbol or . β has insignificant -1 mass ( ) and the charge is because it’s an . This occurs as a neutron changes into a proton by emitting the negatively charged beta particle Beta decay causes change in number and causes the number to increase by . electron e 0 electron -1 no atomic mass e 1

Beta Decay Example 1: Write the nuclear equation for the radioactive decay of carbon – 14 by beta emission. Step 2: Draw the arrow. Step 1: Write the element that you are starting Step 4: Determine the other product (ensuring Step 3: Write the beta particle. with. everything is balanced). Mass # 14 6 C Atomic # 14 7 N 0 -1 e

Beta Decay Example 2: Write the nuclear equation for the radioactive decay of zirconium – 97 by beta decay. Step 2: Draw the arrow. Step 1: Write the element that you are starting Step 4: Determine the other product (ensuring Step 3: Write the beta particle. with. everything is balanced). Mass # 97 40 Zr Atomic # 97 41 Nb 0 -1 e

Gamma decay electromagnetic γ 0 mass atomic always Gamma rays – high-energy radiation, denoted by the symbol . γ has no mass ( ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. no 0 no omitted

Transmutation – the of one atom of one element to an atom of a different element ( decay is one way that this occurs!)

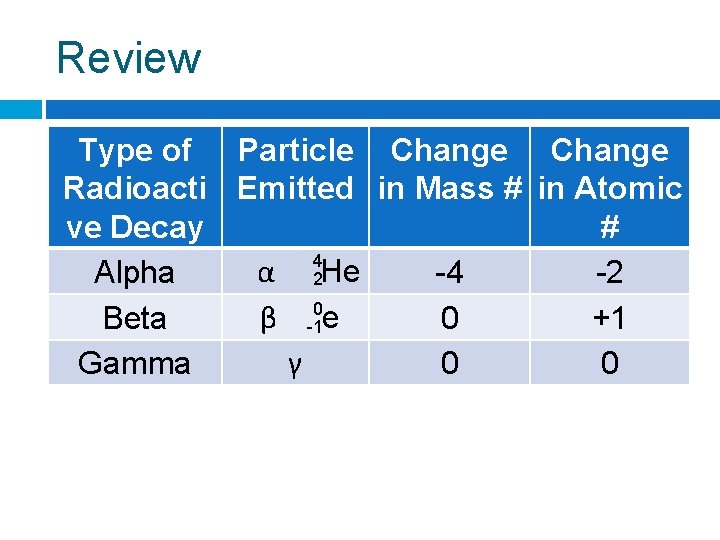
Review Type of Particle Change Radioacti Emitted in Mass # in Atomic ve Decay # 4 α He Alpha -4 -2 2 0 β e Beta 0 +1 -1 γ Gamma 0 0

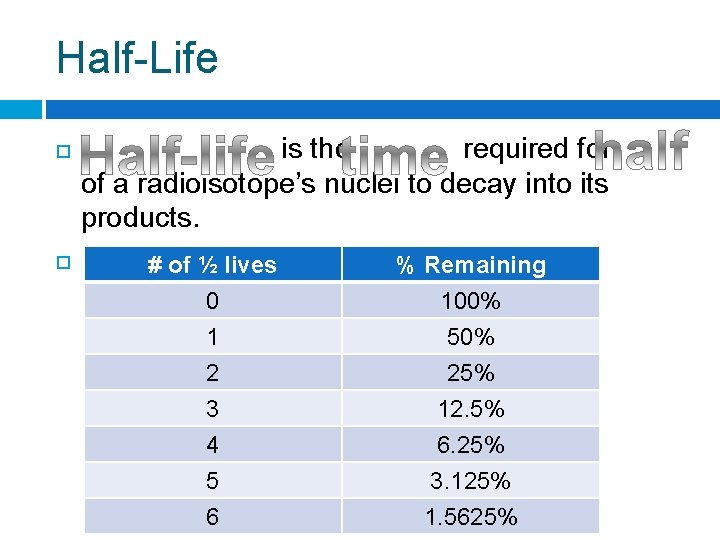
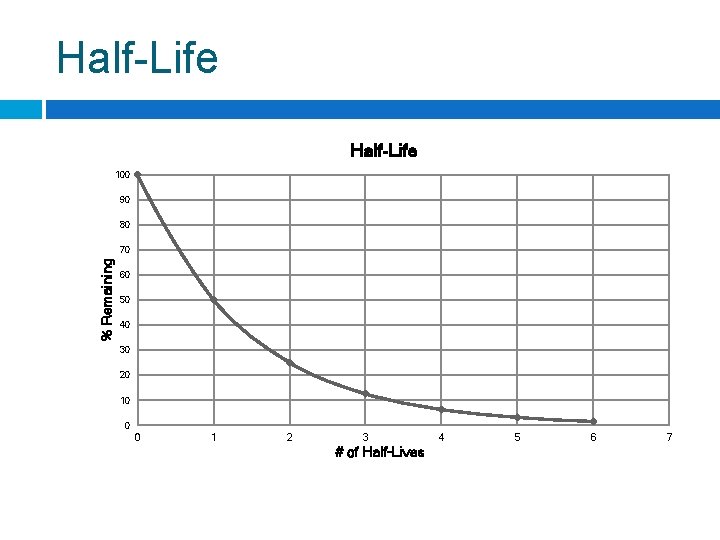
Half-Life is the required for of a radioisotope’s nuclei to decay into its products. For any radioisotope, # of ½ lives % Remaining 0 1 2 100% 50% 25% 3 4 5 6 12. 5% 6. 25% 3. 125% 1. 5625%

Half-Life 100 90 80 % Remaining 70 60 50 40 30 20 10 0 0 1 2 3 # of Half-Lives 4 5 6 7

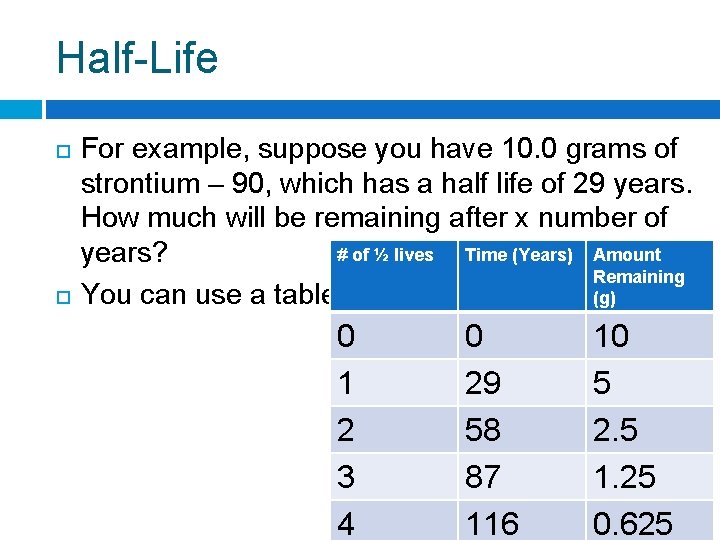
Half-Life For example, suppose you have 10. 0 grams of strontium – 90, which has a half life of 29 years. How much will be remaining after x number of # of ½ lives Time (Years) Amount years? Remaining You can use a table: (g) 0 1 2 3 4 0 29 58 87 116 10 5 2. 5 1. 25 0. 625
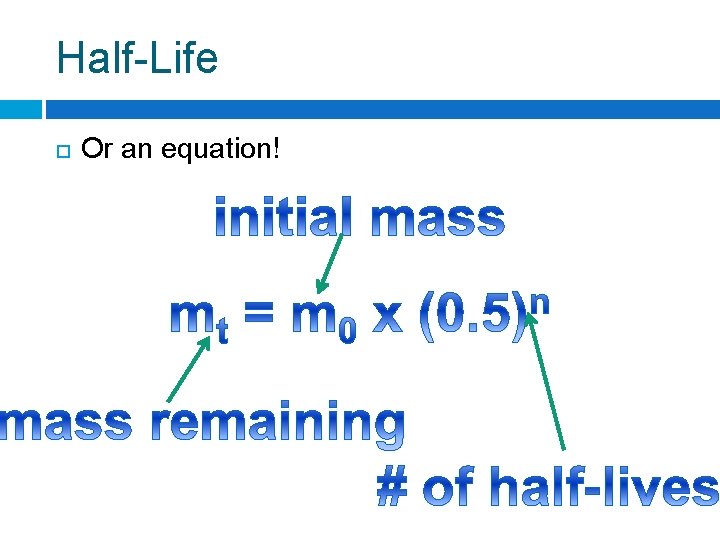
Half-Life Or an equation!

Half-Life Example 1: If gallium – 68 has a half-life of 68. 3 minutes, how much of a 160. 0 mg sample is left after 1 half life? ____ 2 half lives? _____ 3 half lives? _____
Half-Life Example 2: Cobalt – 60, with a half-life of 5 years, is used in cancer radiation treatments. If a hospital purchases a supply of 30. 0 g, how much would be left after 15 years? _______

Half-Life Example 3: Iron-59 is used in medicine to diagnose blood circulation disorders. The halflife of iron-59 is 44. 5 days. How much of a 2. 000 mg sample will remain after 133. 5 days? _______

Half-Life Example 4: The half-life of polonium-218 is 3. 0 minutes. If you start with 20. 0 g, how long will it take before only 1. 25 g remains? _______

Half-Life Example 5: A sample initially contains 150. 0 mg of radon-222. After 11. 4 days, the sample contains 18. 75 mg of radon-222. Calculate the half-life.

Nuclear Reactions Characteristics: Isotopes of one element are into isotopes of another element Contents of the change amounts of are released

Types of Nuclear Reactions decay – alpha and beta Radioactive particles and gamma ray emission Nuclear - emission of disintegration a neutron or proton

Nuclear Fission - of a nucleus - Very heavy nucleus is split into approximately fragments - reaction releases several neutrons which more nuclei - If controlled, energy is released (like in ) Reaction control depends on reducing the of the neutrons (increases the reaction rate) and extra neutrons ( creases the reaction rate).

Nuclear Fission - 1 st controlled nuclear reaction in December 1942. 1 st uncontrolled nuclear explosion occurred July 1945. - Examples – atomic bomb, current nuclear power plants

Nuclear Fusion - of a nuclei combining Fusion - Two nuclei combine to form a light single heavier nucleus - Does not occur under standard conditions ( + + repels ) inexpensive - Advantages compared to fission - no radioactive waste , large start control - Disadvantages - requires amount of energy to , difficult to - Examples – energy output of stars, hydrogen

Uses of Radiation Radioactive dating - Carbon - 14 used to determine the age of an object that was once alive. Radioactive tracing of diseases – Iodine – 131 used to detect thyroid problems. Treatment of some cancers (cobalt – 60 and cesium – 137) – cancer cells are more sensitive to radiation than normal, healthy cells

Uses of Radiation tungsten - 182 generally is used to generate Xrays thorium – 232 used in gas lanterns and welding plutonium – 238 used in space probes and satellites americium – 241 in smoke detectors.