Chapter 25 Nuclear Chemistry 1 Nuclear Radiation Nuclear


























































- Slides: 83

Chapter 25 Nuclear Chemistry 1

Nuclear Radiation Nuclear chem Ø the study of the structure of atomic nuclei and the changes they undergo. Ø No e- / orbitals Ø No e- sharing or transferring Ø No cpds formed Ø No bondings 2

The Discovery of Radioactivity • In 1895, Roentgen found that invisible rays were emitted when e- bombarded the surface of certain materials. • they caused photographic plates to darken. • named these invisible high-energy emissions X rays. 3

The Discovery of Radioactivity At that time, French physicist Becquerel was studying minerals that • emit light after being exposed to sunlight (phosphorescence). • Building on Roentgen’s work, Becquerel wanted to determine whether phosphorescent minerals also emitted X rays. 4

The Discovery of Radioactivity Becquerel accidentally discovered that phosphorescent U salts – even when not exposed to light – produced spontaneous emissions that darkened photographic plates. 5

The Discovery of Radioactivity Marie Curie (1867– 1934) and her husband Pierre (1859– 1906) took Becquerel’s mineral sample and isolated the components emitting the rays. 6

The Discovery of Radioactivity Conclusion: Ø the darkening of the photographic plates was due to rays emitted specifically from the U atoms in the mineral sample. Ø named the process by which materials give off such rays radioactivity; Ø the rays and particles emitted by a radioactive source are called radiation. 7

Types of Radiation Isotopes • are atoms of the same element that have different #s of n 0. Radioisotopes • Isotopes of atoms with unstable nuclei ready to emit radiations. unstable nuclei is due to big diffce in the # of p+ and n 0. e. g. U has 92 p+ and over 140 n 0 Mg has 12 p+ and 12 n 0 (stable) 8

Quick write What kind of atoms are radioactive? 9

Types of Radiation radioactive decay • A process that unstable nuclei emit radiation to attain more stable atomic configns. • During radioactive decay, unstable atoms lose energy by emitting 1 of several types of radiation. 10

Types of Radiation 3 common types of radiation • alpha (α) • beta (β) • gamma (γ) 11

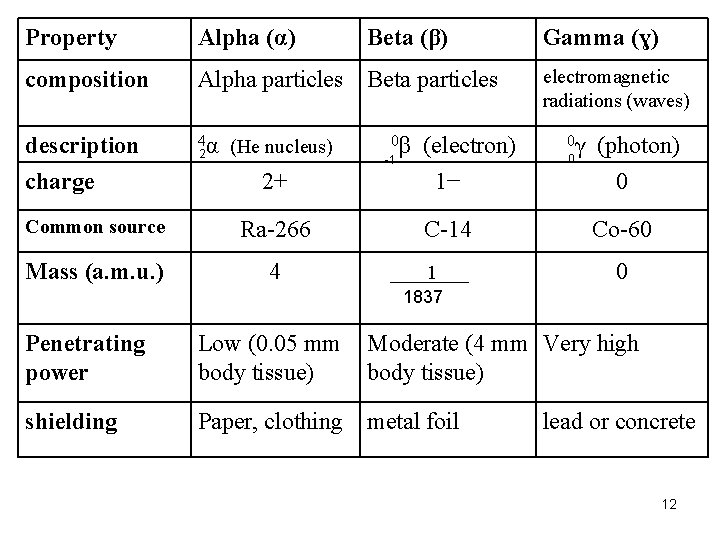
Alpha (α) composition Alpha particles Beta particles description 4α charge 2 (He nucleus) Beta (β) 0β -1 (electron) Gamma (ɣ) electromagnetic radiations (waves) 0γ 0 Property (photon) 2+ 1− 0 Common source Ra-266 C-14 Co-60 Mass (a. m. u. ) 4 1 0 1837 Penetrating power Low (0. 05 mm body tissue) Moderate (4 mm Very high body tissue) shielding Paper, clothing metal foil lead or concrete 12

Quick Write What are α particles? How many p+, n 0 and edoes each particle carry? 13

Radioactive Decay Ø unstable nuclei loses energy by emitting radiation: ‘particles’ or ‘energy (non-particles)’. Ø become lighter Ø Natural (not human initiated; can’t stop or slow down) Ø not requiring any energy input. Ø Spontaneous 14

15

Quick Write What are the areas that α decay and β decay have in common? (4 bullet pts) • • 16

Radioactive Decay 17

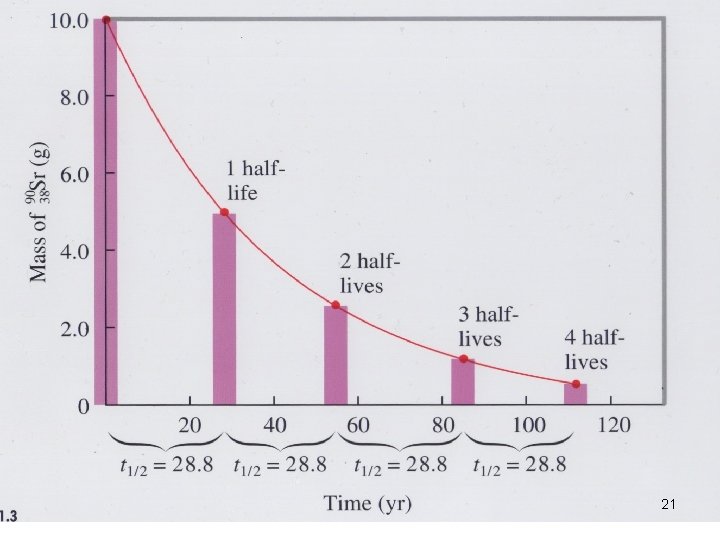
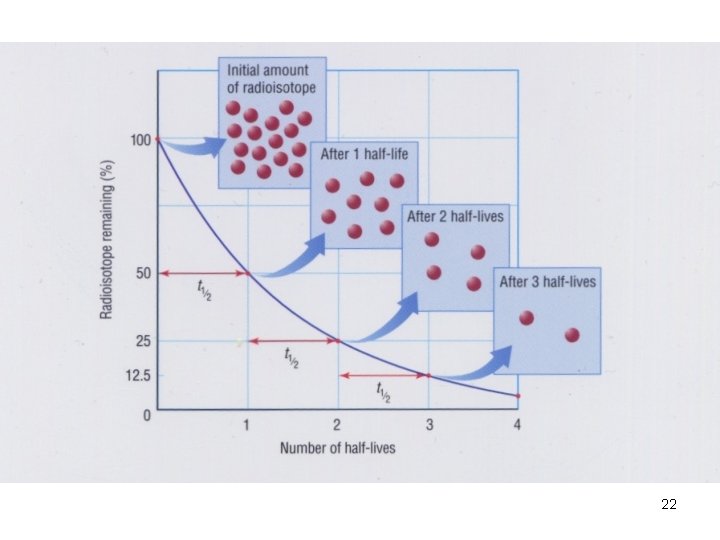
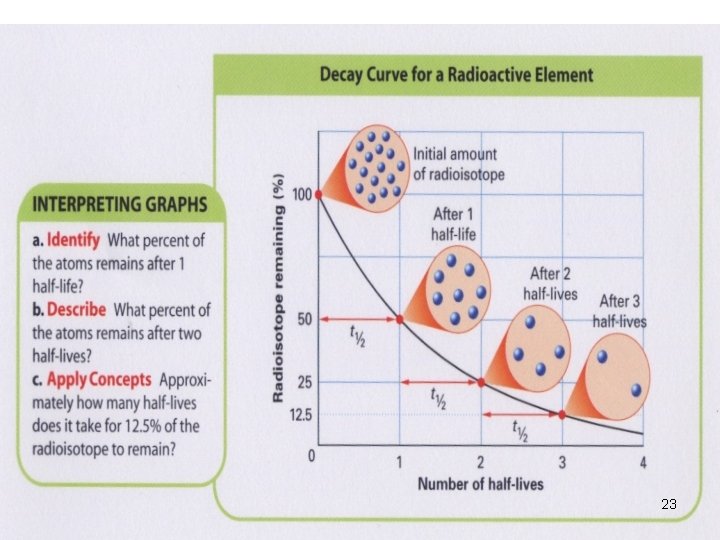
Half-life Ø Time required for one half of the nuclei of a radioisotope sample to decay. Ø e. g. U → Th + α-particle Ø Each radioisotope has a characteristic t 1/2 18

Half-life Ø Time required for one half of the nuclei of a radioisotope sample to decay. Ø e. g. U → Th + α-particle 238 U 92 → 234 Th 90 + 4 He 2 Ø Each radioisotope has a characteristic t 1/2 19

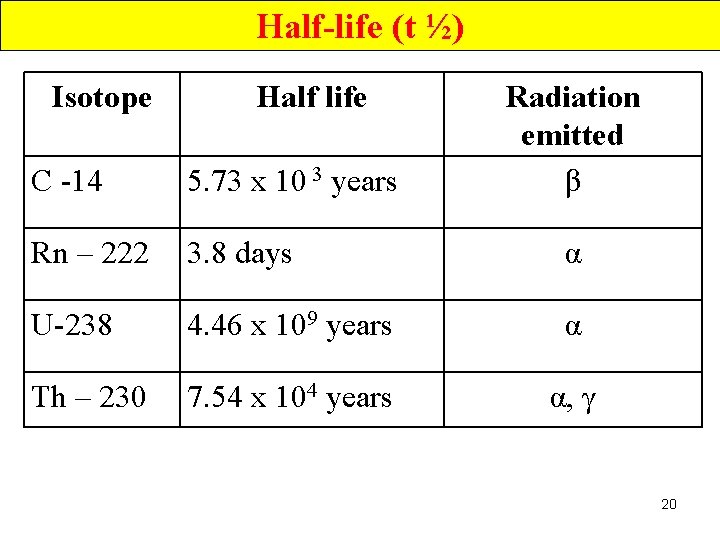
Half-life (t ½) Isotope Half life Radiation emitted β C -14 5. 73 x 10 3 years Rn – 222 3. 8 days α U-238 4. 46 x 109 years α Th – 230 7. 54 x 104 years α, γ 20

21

22

23

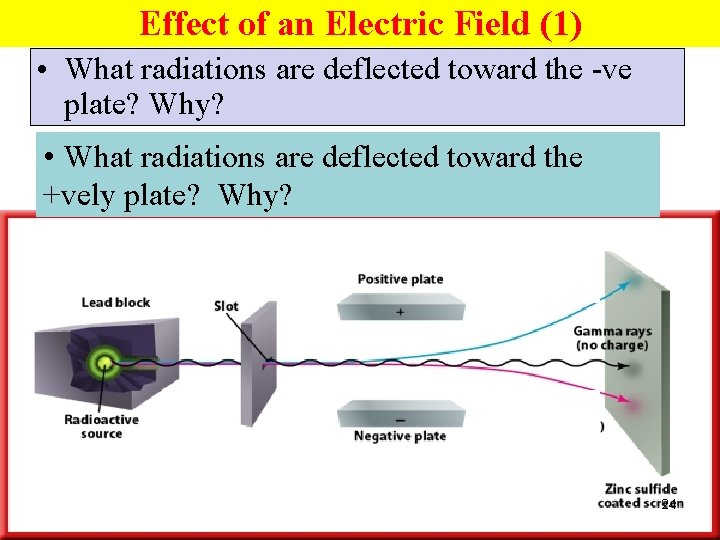
Effect of an Electric Field (1) • What radiations are deflected toward the -ve plate? Why? • What radiations are deflected toward the +vely plate? Why? 24

Quick Write What is radioactive decay? (5 bullet points) 25

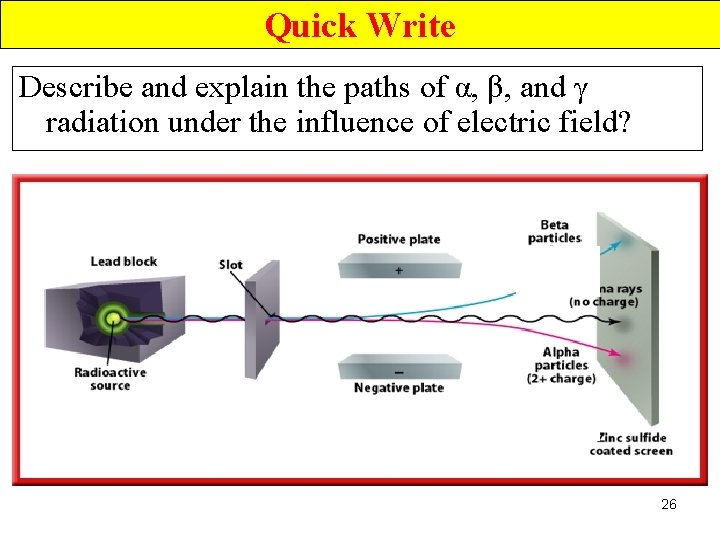
Quick Write Describe and explain the paths of α, β, and γ radiation under the influence of electric field? 26

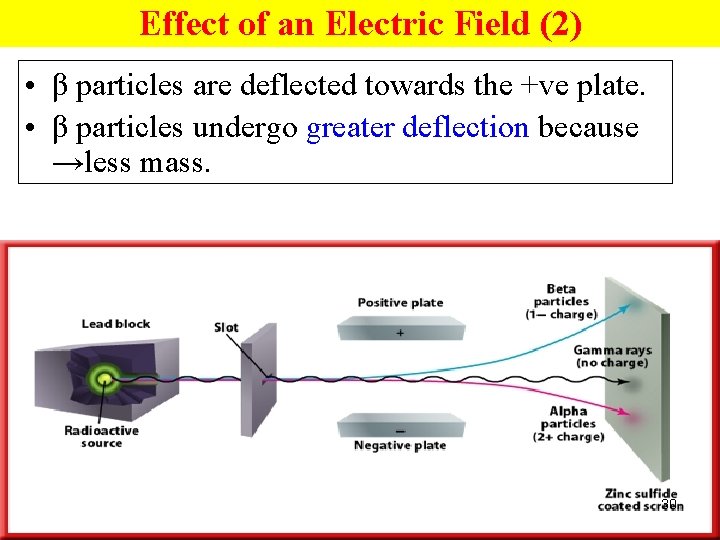
Effect of an Electric Field (2) • The +vely charged α particles are deflected towards the -ve plate. • The –vely charged β particles are deflected towards the +ve plate. • the neutral γ radiation travels in a straight line. 27

Quick Write What are β particles? Do they carry any charges? 28

Quick Write What is γ radiation? What charge does it carry? 29

Effect of an Electric Field (2) • β particles are deflected towards the +ve plate. • β particles undergo greater deflection because →less mass. 30

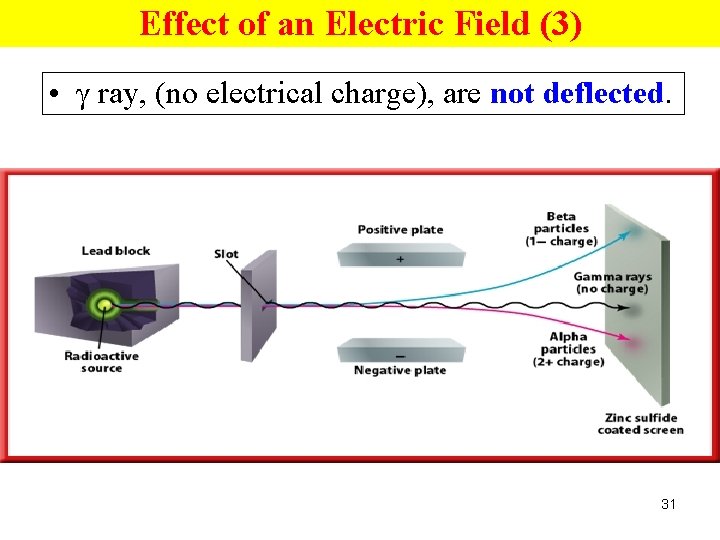
Effect of an Electric Field (3) • γ ray, (no electrical charge), are not deflected. 31

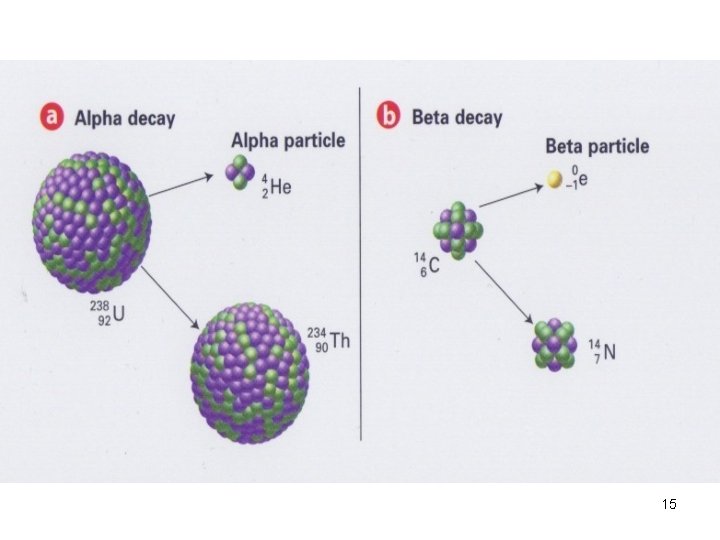
Types of Radiation- α radiation Ø An α particle has the same composition as a He nucleus— 2 p+ and 2 n — Ø 2+ due to the presence of the 2 p+. 32

Types of Radiation- α radiation • α radiation—a stream of α particles. • Ra-226, (88 p+ and 138 n 0), undergoes α decay by emitting an α particle. 33

Types of Radiation- α radiation • After the decay, the resulting atom has an atomic # of 86, a mass # of 222. • The new radiosiotope is Rn-222. 34

Types of Radiation • The particles involved are balanced. • i. e. the sum of the mass #s (superscripts) = the sum of the atomic #s (subscripts) on each side of the arrow. 35

Types of Radiation • Because of their mass and charge, α particles are relatively slow-moving compared with other … • Thus, α particles are not very penetrating—a single sheet of paper stops. 36

Types of Radiation—β Radiation Ø A β particle is a very-fast moving e- that has been emitted from a n 0 of an unstable nucleus. Ø β particles are represented by the symbol. The ‘ 0’ superscript indicates the insignificant mass of an e- in comparison with the mass of a nucleus. 37

38

Types of Radiation—β Radiation • The – 1 subscript denotes the -ve charge of the particle. • β radiation consists of a stream of fast-moving e -. 39

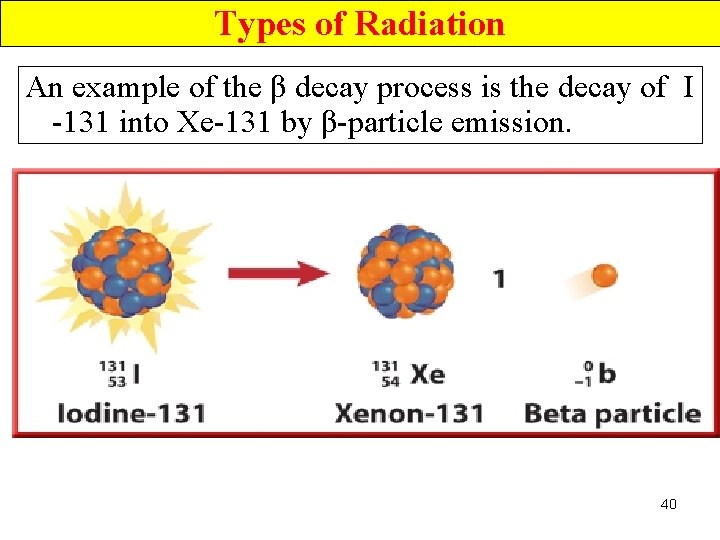
Types of Radiation An example of the β decay process is the decay of I -131 into Xe-131 by β-particle emission. 40

Types of Radiation-- β Radiation • The mass # of the product nucleus is the same as that of the original nucleus (both 131), but its atomic # has increased by 1 (54 instead of 53). 41

Types of Radiation-- β Radiation • This change in atomic #, and thus, change in identity, occurs because the e- emitted during the β decay has been removed from a n 0, leaving behind a p+. 42

Types of Radiation—β Radiation β radiation: fast-moving e- formed by decomposition of a n 0 in an atom. Quick write: why the mass # remain 14 while there is an additional p+? 43

Types of Radiation • Because β particles are both lightweight and fast moving, they have greater penetrating power than α particles. • A thin metal foil is required to stop β particles. 44

Types of Radiation • γ rays are high-energy (short wavelength) electromagnetic radiation. • They are denoted by the symbol . • Both the subscript and superscript are ‘ 0’. 45

Types of Radiation • Thus, the emission of γ rays does not change the atomic # or mass # of a nucleus. • γ rays almost always accompany α and β radiation, as they account for most of the energy loss that occurs as a nucleus decays. 46

Types of Radiation • e. g. γ rays accompany the α-decay rxn of U-238. • The 2 in front of the γ symbol indicates that 2 γ rays of different frequencies are emitted. • Because γ rays have no effect on mass # or atomic #, it is customary to omit them from nuclear eqns. 47

Radioactive Decay • Of all the known isotopes, only about 17% are stable and don’t decay spontaneously. 48

Beta Decay • A radioisotope that lies above the band of stability is unstable because it has too many n relative to its # of p+. e. g. unstable has a n 0 / p+ ratio of 1. 33 : 1, whereas stable elements of similar mass, such as and , have n 0 / p+ ratios ≈1: 1 49

Beta Decay It is not surprising then that undergoes beta decay, as this type of decay decreases the # of n 0 in the nucleus. Note that the atomic # of the product nucleus, , has increased by 1. • The N-14 atom now has a stable n 0 / p+ ratio of 1 : 1. 50

Beta Decay • Thus, β emission has the effect of increasing the stability of a n 0 -rich atom by lowering its n 0 / p+ ratio. • The resulting atom is closer to, if not within, the band of stability. 51

Alpha Decay • All nuclei with more than 83 p+ are radioactive and decay spontaneously. • Both the # of n 0 and the # of p+ must be reduced in order to make these radioisotopes stable. • These very heavy nuclei often decay by emitting α particles. 52

Alpha Decay • e. g. , Po-210 spontaneously decays by α emission. The atomic # of decreases by 2 and the mass # decreases by 4 as the nucleus decays into. 53

54

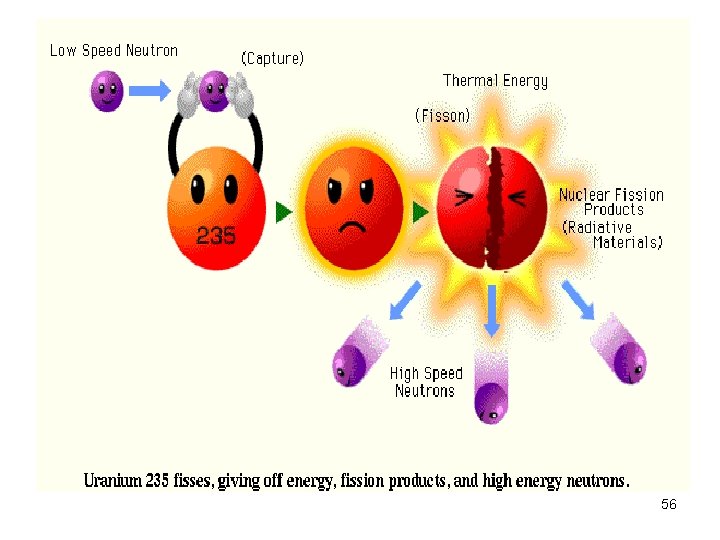
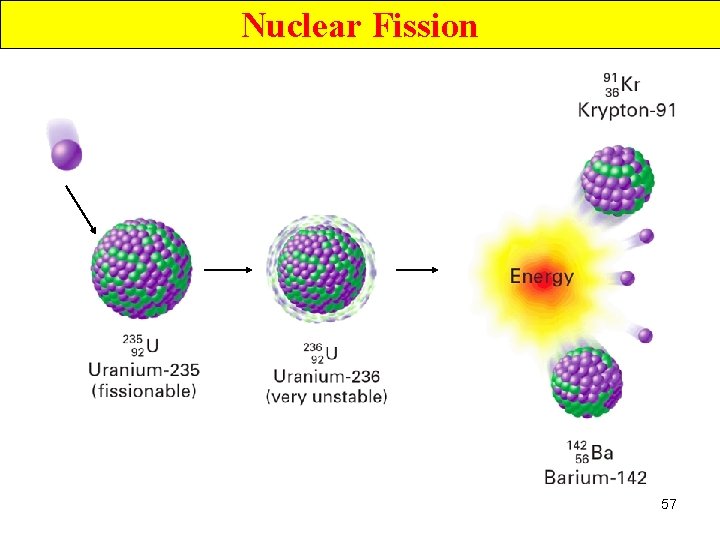
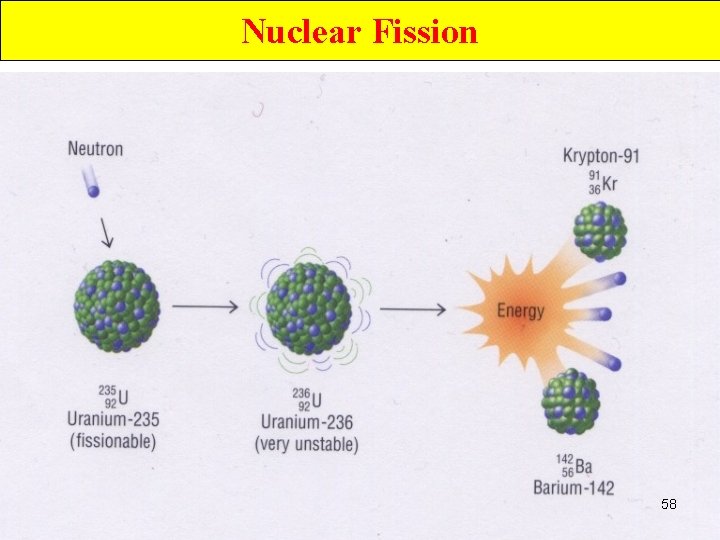
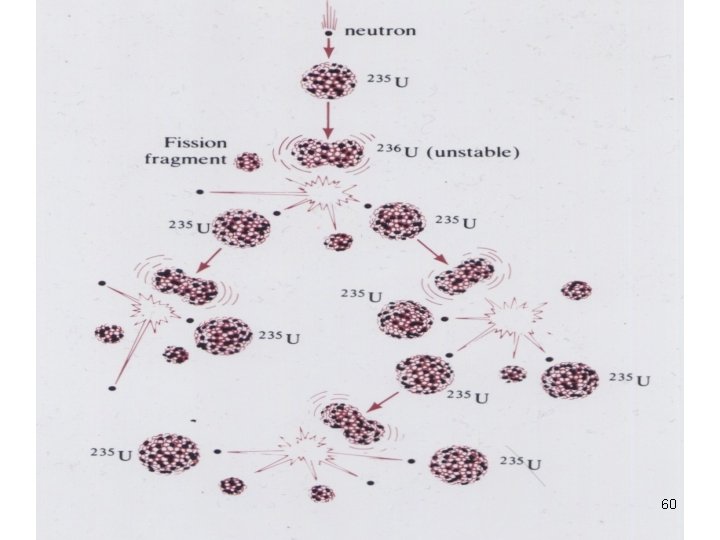
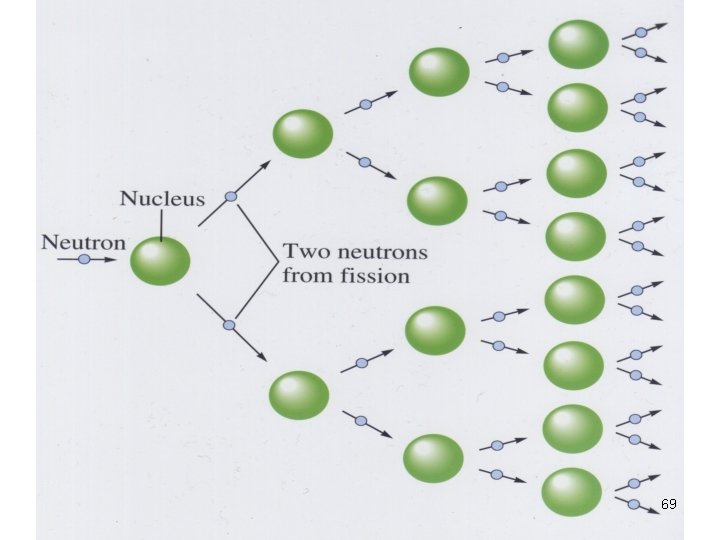
Nuclear Fission • Nuclei of certain isotopes (e. g U-238) are bombarded with n 0 split into smaller fragments of similar sizes. Unleash enormous amt of energy e. g. 1 kg of U- 235 (explosion of 20000 tons of dynamite) 55

56

Nuclear Fission 57

Nuclear Fission 58

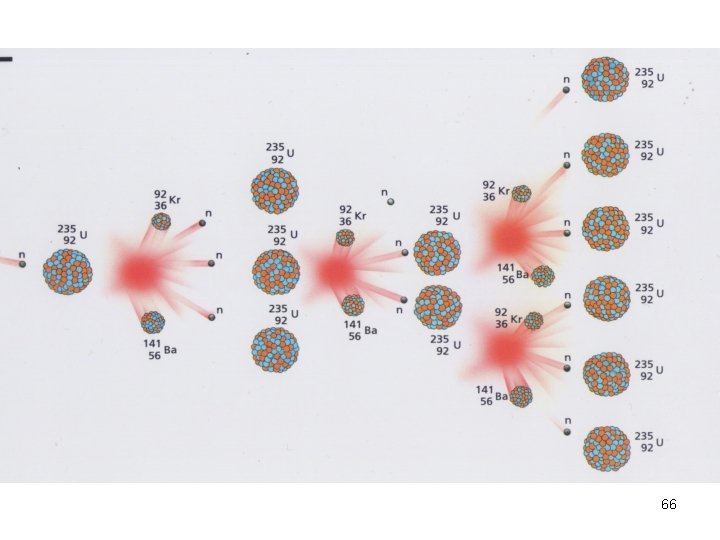
Nuclear Fission In a chain rxn • some of the n 0 produced react with other fissionable atoms • producing more n 0 which react with still more fissionable atoms. 59

60

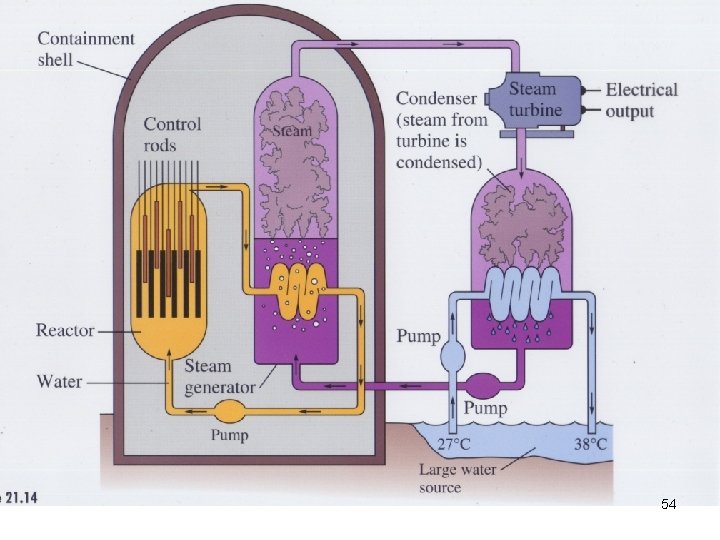
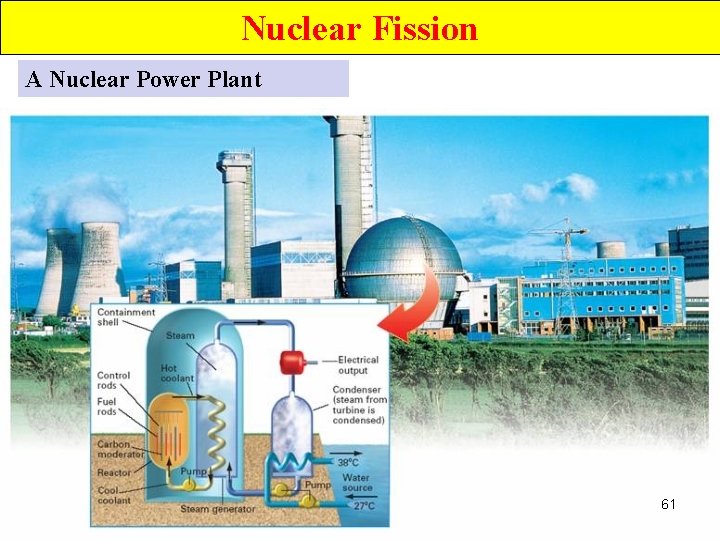
Nuclear Fission A Nuclear Power Plant 61

62

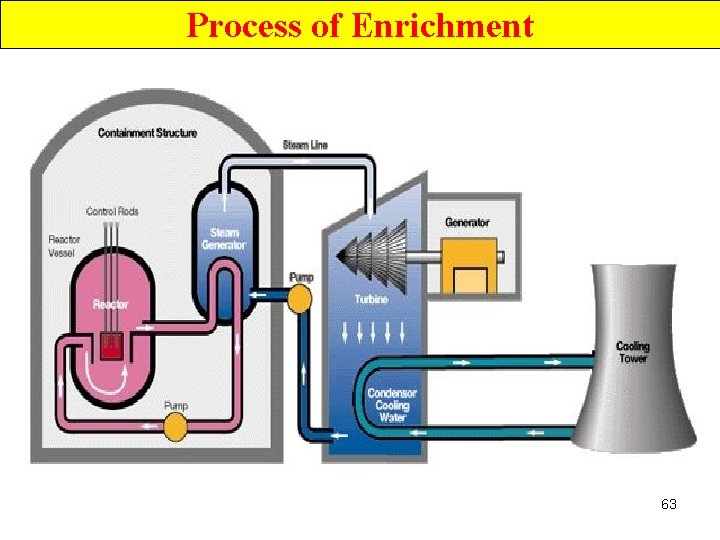
Process of Enrichment 63

Nuclear Fission Neutron Moderation Ø a process that slows down n 0 so the reactor fuel (U-235 or Po-239) captures them to continue the chain rxn. 64

Nuclear Fission Neutron Absorption • a process that decreases the # of slow-moving n 0. • water slow down the n 0 in the reactor Control rods—made of a material such a Cd, or B, are used to absorb n 0. 65

66

Nuclear Fission • Nuclei of certain isotopes are bombarded with neutrons • split into smaller fragments of similar sizes. • Unleash enormous amt of energy • e. g. 1 kg of U- 235 ( explosion of 20000 tons of dynamite) 67

Nuclear Fission Uncontrolled fission • Total energy release is instantaneous (fraction of a second); • uncontrolled chain rxn—atomic bomb; Controlled fission §so energy is released more slowly (nuclear power) 68

69

Controled/uncontroled fission 70

Fission and Fusion of Atomic Nuclei • The sun is not actually burning (combustion). • would have burned out approximately 2000 years after it was formed. 71

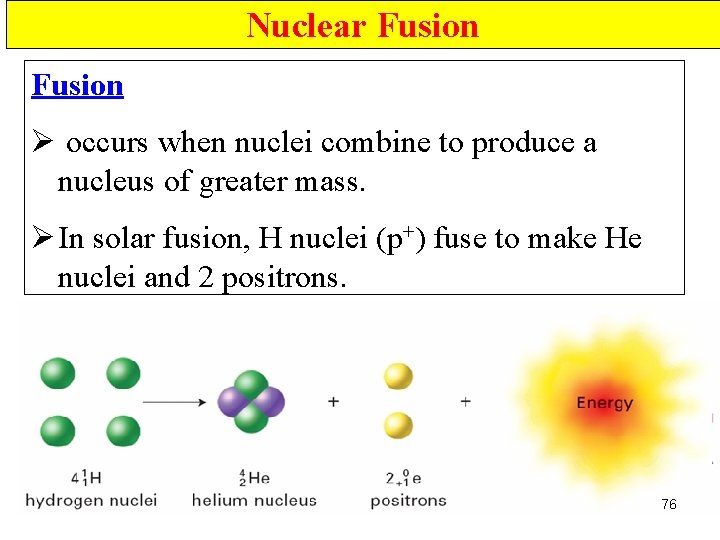
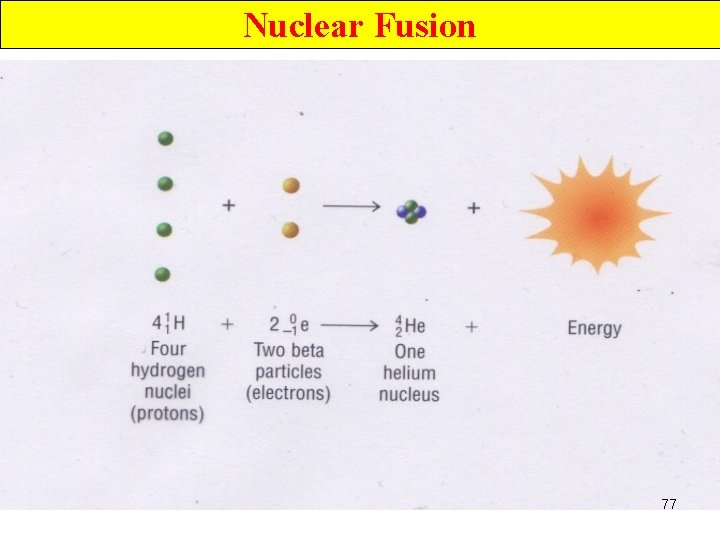
Nuclear Fusion • Occur when small nuclei combine to produce a nucleus of greater mass • H nuclei (p+) fuse to make He nuclei 72

Nuclear Fusion • much more energy than fission rxn • occur at very high temp (40, 000 ° C) • good energy source--- cheap fuel; readily available; controlled 73

Nuclear Fusion • problems; high temp to initiate? --- needs an atomic bomb to trigger a nuclear fusion rxn… **** the energy release per g of the material is much larger in nuclear fusion or fission rxns than in chem rxns. • change in mass (calculated by E = mc 2 ) is small but significant in nuclear rxns. 74

25. 3 Quick-write How do fission rxns and fusion rxns differ? 75

Nuclear Fusion Ø occurs when nuclei combine to produce a nucleus of greater mass. Ø In solar fusion, H nuclei (p+) fuse to make He nuclei and 2 positrons. 76

Nuclear Fusion 77

Nuclear Fusion rxns Ø small nuclei combine, Ø release much more energy than fission rxns (large nuclei split). 78

25. 3 Nuclear Fusion Ø The use of controlled fusion as an energy source on Earth is appealing. Ø The potential fuels are inexpensive and readily available. The problems with fusion Ø in achieving the high temperatures necessary to start the rxn and Øin containing the rxn once it has started. 79

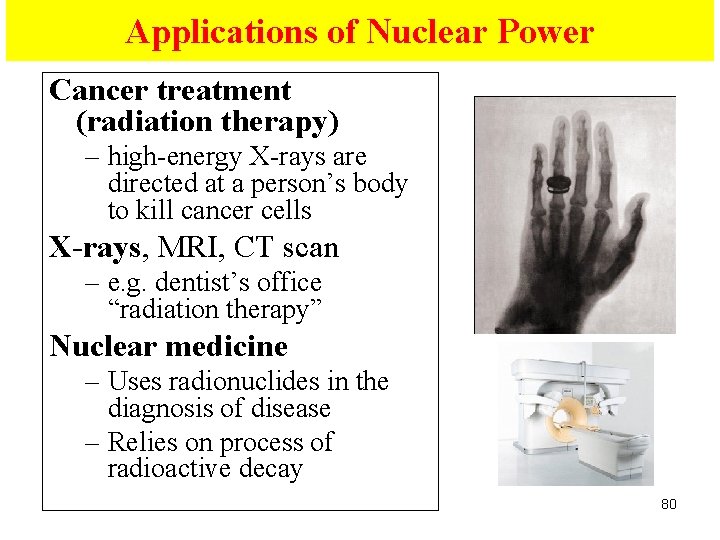
Applications of Nuclear Power Cancer treatment (radiation therapy) – high-energy X-rays are directed at a person’s body to kill cancer cells X-rays, MRI, CT scan – e. g. dentist’s office “radiation therapy” Nuclear medicine – Uses radionuclides in the diagnosis of disease – Relies on process of radioactive decay 80

CST example 1 A 2 -cm thick piece of cardboard placed over a radiation source would be most effect in protecting against which type of radiation? A alpha B beta C gamma D x-ray 81

CST problem 2 Which equation correctly represents the alpha decay of polonium-214? A 214 Po 214 84 0 85 82

The End 83