Nuclear Chemistry Radiation and Radioactivity What do you























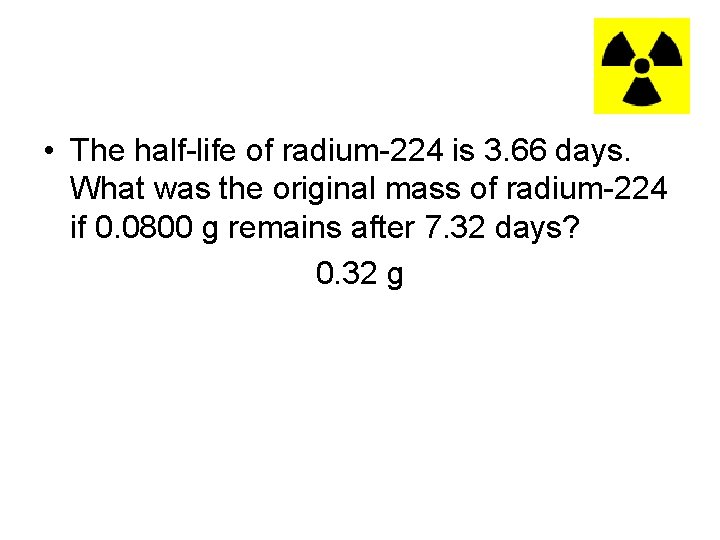
- Slides: 31

Nuclear Chemistry Radiation and Radioactivity

What do you think? T/F • Radioactivity first appeared during WWII. • Atoms cannot be changed from one element to another. • Fission and fusion are the same thing. • Radioactivity lasts forever. • Exposure to radiation makes something radioactive. • Nuclear power plants can explode like bombs. • Radioactivity is man-made. • When radioactive substances decay, they disappear.

What is radiation? • Electromagnetic Radiation – Electric Fields – Magnet Fields – Fields oscillate and travel in waves

Rules of Radiation • An atom will release energy as electromagnetic radiation in order to become stable. • A stable nucleus has at least as many or more neutrons as protons. • Atoms with a mass #209 or greater are never stable.

Decay doesn’t mean disappear! • Radioactive decay is the process by which the nucleus of an unstable atom loses energy by emitting ionizing radiation. • The emission is spontaneous, in that the atom decays without any interaction with another particle from outside the atom

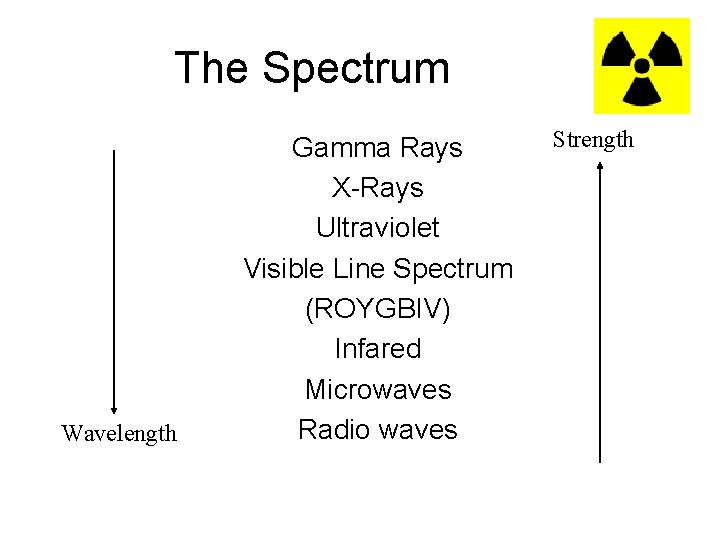
The Spectrum Wavelength Gamma Rays X-Rays Ultraviolet Visible Line Spectrum (ROYGBIV) Infared Microwaves Radio waves Strength

Fission or Fusion? • Fission: one atom splits into two – Uranium-235 to Barium and Krypton – Used in medical radiology • Fusion: two atoms join to form one new – The Sun – H + H = He + energy

Radiation • There are three main types of radiation: • • • * Alpha radiation * Beta radiation * Gamma radiation

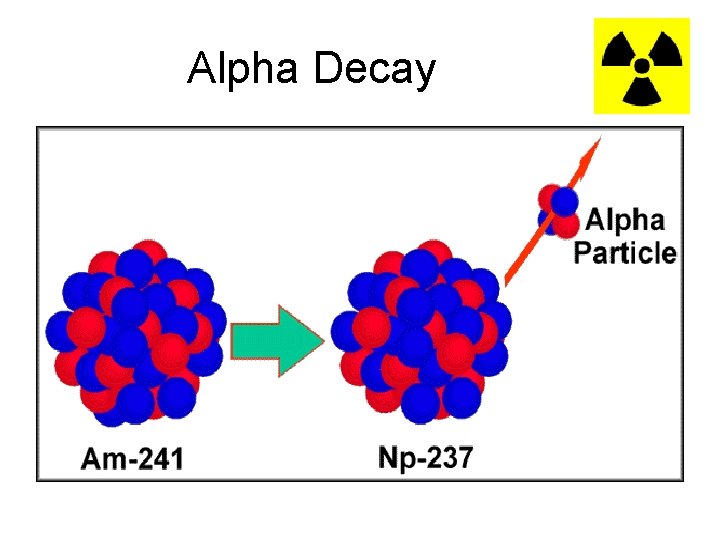
Alpha Decay • Alpha (α) decay occurs is because the nucleus has too many protons which cause excessive repulsion. • Alpha particles can be stopped by a thin sheet of paper • In an attempt to reduce the repulsion, a Helium nucleus is emitted. 4 He 2

Alpha Decay

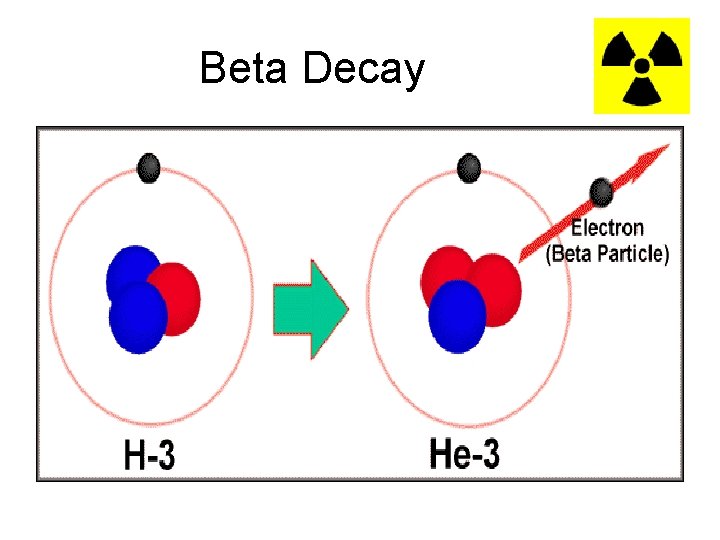
Beta Decay • Beta (β) decay occurs when the neutron to proton ratio is too great in the nucleus and causes instability. • Beta particles can be blocked by a thin sheet of metal • In basic beta decay, a neutron is turned into a proton and an electron. The electron is then emitted. 0 e -1

Beta Decay

Beta Example • When Carbon-14 decays, one of the neutrons is converted into a proton, and an electron is emitted 14 14 0 6 C 7 N + -1 e

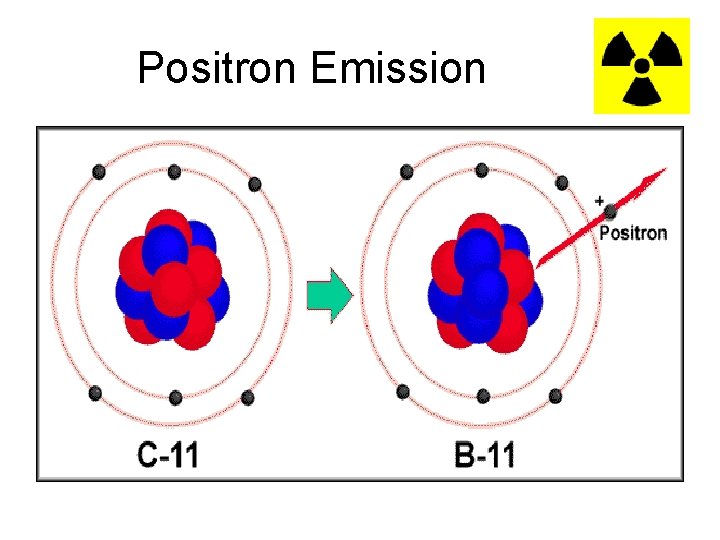
Positron • There is also positron emission when the neutron to proton ratio is too small. • A proton turns into a neutron and a positron and the positron is emitted. • A positron is basically a positively charged electron. • This is another form of Beta decay

Positron Emission

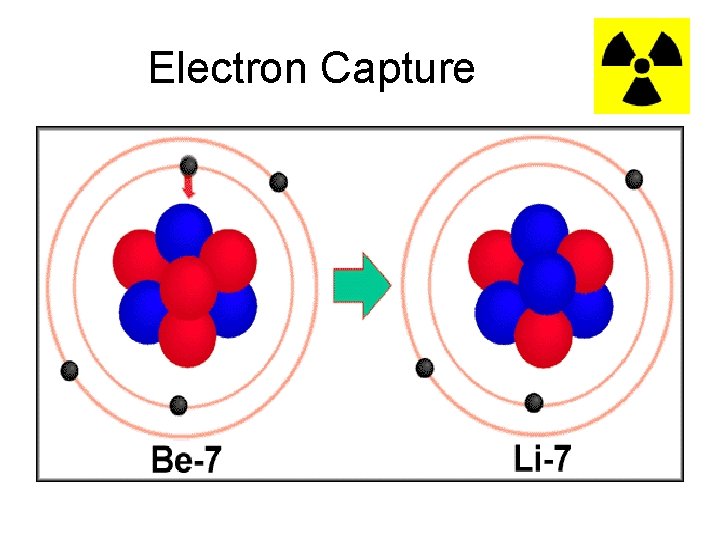
Electron Capture • The final type of beta decay is known as electron capture and also occurs when the neutron to proton ratio in the nucleus is too small. • The nucleus captures an electron which basically turns a proton into a neutron.

Electron Capture

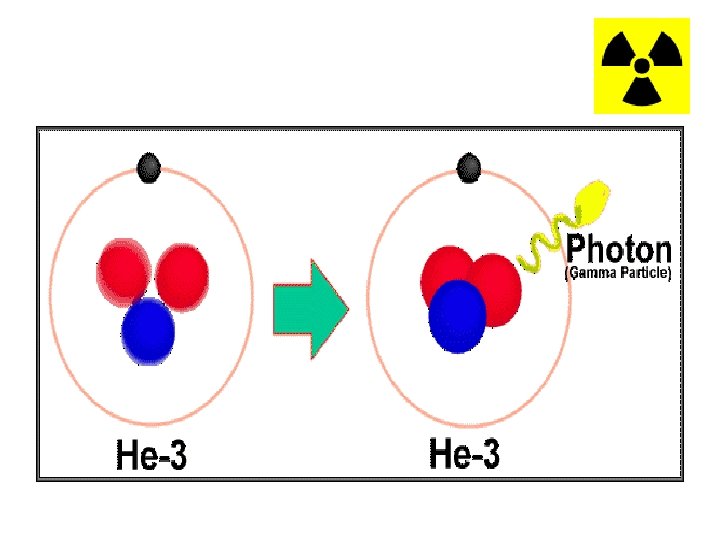
Gamma Decay • Gamma (γ) decay occurs because the nucleus is at too high an energy. • The nucleus falls down to a lower energy state and, in the process, emits a high energy photon known as a gamma particle. • Gamma radiation can only be blocked with very thick metal, usually Lead


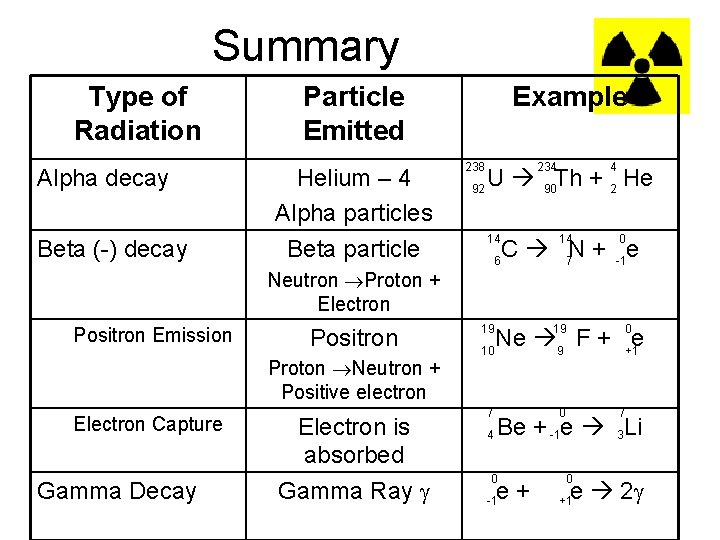
Summary Type of Radiation Alpha decay Beta (-) decay Particle Emitted Helium – 4 Alpha particles Beta particle Neutron Proton + Electron Positron Emission Positron Proton Neutron + Positive electron Electron Capture Gamma Decay Electron is absorbed Gamma Ray Example 238 92 234 4 U 90 Th + 2 He 14 6 14 0 C 7 N + -1 e 19 19 7 0 0 Ne 9 F + +1 e 10 7 4 Be + -1 e 3 Li 0 0 -1 +1 e+ e 2

Half Life • The rate of radioactive decay is related to the energy change that accompanies the transformation, but it is not a direct relationship. • The rate of radioactive emissions of a radioactive nuclide is directly proportional to the amount of radioactive material present. • The rate of decay of a radioactive nuclide is measured by its half-life.

• The half-life of a radioactive substance is the time it takes for half of an initial amount of the substance to decay. • The half-live is independent of chemical activity, external pressure, and temperature.

• Consider a 10 g sample of Au-198 (half. Life of 2. 69 days) • After 0 half-life or 0 days 10 g are present.

• After 1 half-life or 2. 69 days, 5 g remains.

• After 2 half-life or 5. 38 days (2 x 2. 69 days) 2. 5 g remains.

Half-Life Formula • • T = half life t = total time elapsed Fraction remaining = 1/2 (t/T) Number of half-life periods = t / T – ½ x ½ x ½ = 1/16 with 4 half-lives

Calculating Remaining Mass • How much of a 100 g sample of Nitrogen 16 will remain after 28. 8 seconds of decay? (Half life is 7. 2 s, -decay) • Fraction remaining: 1 (t/T) 2 . . . So – ½ (28. 8/7. 2) = ½ 4 = 0. 0625 – Then, 100 g x 0. 0625 = 6. 25 g left

Calculating Half life periods • How many half-lives are required for a radioisotope to decay to 1/32 of its initial value? – Fraction remaining = ½(t/T) ; and – Number of periods = t / T ; so – 1/32 = ½ t/T – t/T = 5

• The half-life of Polonium-210 is 138. 4 days. How many milligrams will be left after 415. 2 days if you start with 2. 0 mg? – Step 1: How many half-life periods? 415. 2 days / 138. 4 days = 3 periods – Step 2: Determine the fraction left. ½ (t/T) = (1/2)3 = (1/8) – Step 3: Using the fraction, determine the mass left. (1/8) x 2. 0 mg = 0. 25 mg

• After 4797 years, how much of a 0. 250 g sample of Radium-226 is left? (half-life is 1599 years) • 0. 0312 G
• The half-life of radium-224 is 3. 66 days. What was the original mass of radium-224 if 0. 0800 g remains after 7. 32 days? 0. 32 g