Nuclear Chemistry The study of nuclear change Some
















- Slides: 20

Nuclear Chemistry The study of nuclear change. Some elements undergo chemical changes in their nucleus that alters their number of protons or neutrons.

Nucleus • nucleus contains nucleons – p+ and n 0 • atomic mass, a. k. a. mass number – protons + neutrons • atomic number – # protons – identifies the element

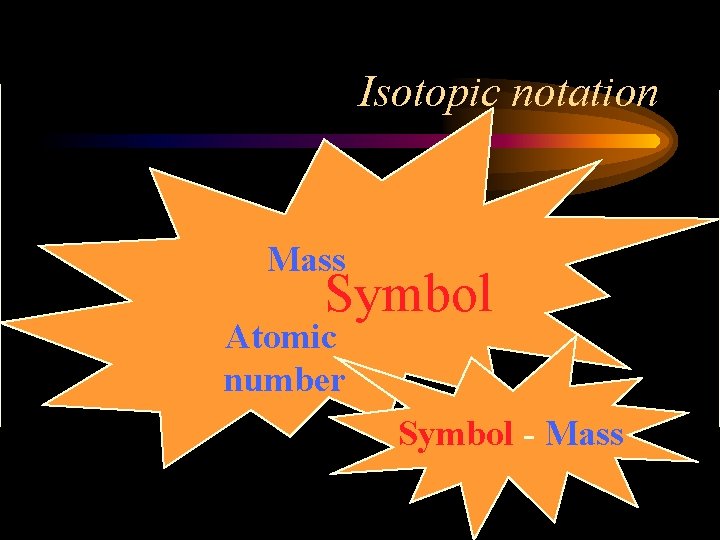
Isotopic notation Mass Symbol Atomic number Symbol - Mass

U-242 U-232 U 238 U-234 U 235 U-240 U 234 U-236 U-239 U 226 U 230 U-238 U-229 U 230 U-235 U 228

Transmutation • Sometimes the changes in the nucleus are such that an element is transformed into a different element – change in atomic number An element is considered radioactive because it releases Energy during transmutation

Artificial Transmutation Natural Transmutation • Change in nucleus occurs when occurs spontaneously bombarded by an – The ONLY reactant alpha particle or is the atom neutron undergoing – MUST be a reactant transmutation

FYI. . . • Since 1940, 23 transuranium elements have been formed • All elements with atomic numbers > 93 were synthesized by nuclear reactions • Glenn Seaborg created 10 of these elements – atomic # 94 - 103 • Element #106 was named for him – Seaborgium

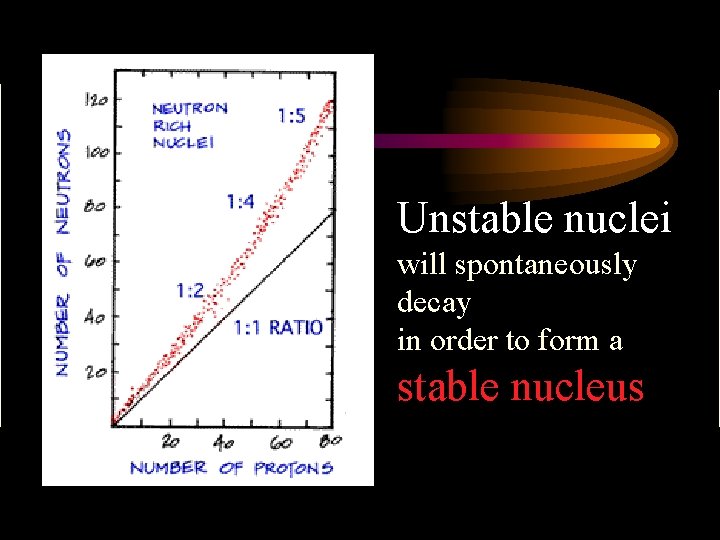
Stability of Nuclei • Most nuclei are stable – some elements have unstable isotopes • Radioisotopes / nuclide – Ex. C-12 stable and won’t transmutate C-14 unstable and will transmutate

What causes instability? • As atoms increase in size the ratio of neutrons to protons also increases – the larger the ratio the more unstable the atom – Ex. compare C-14 to U-238 • All nuclei with atomic numbers > 83 are unstable and radioactive

Unstable nuclei will spontaneously decay in order to form a stable nucleus

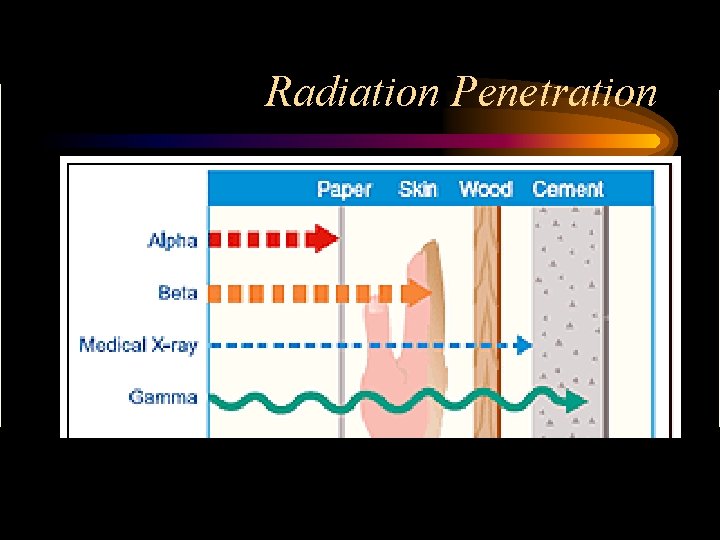
Types of Radiation • Types of radiation emitted from the nucleus during transmutation – alpha particles 4 He or α 2 – beta particles ß- or – gamma radiation – Positrons β+ or γ

Radiation Penetration

How does Radiation affect cells? • Radiation has the ability to ionize atoms – Ionizing radiation causes damage to tissues by damaging molecules • Most damaging is alpha; least is gamma – Damage to proteins & DNA are most damaging – If radiation exposure is low the body may repair itself – If exposure is high, cells may die or cellular instructions might get “scrambled”

• Radioisotopes are used in diagnosing and treating cancer – I-131 • diagnose thyroid disorders – Co-60 • treat cancer Short half lives decay quickly

Alpha Decay • Alpha particle has to be a product.

Beta Decay • A Beta particle has to be a product

Gamma Radiation • Type of electromagnetic radiation like an x-ray • No charge, no mass, no equations!

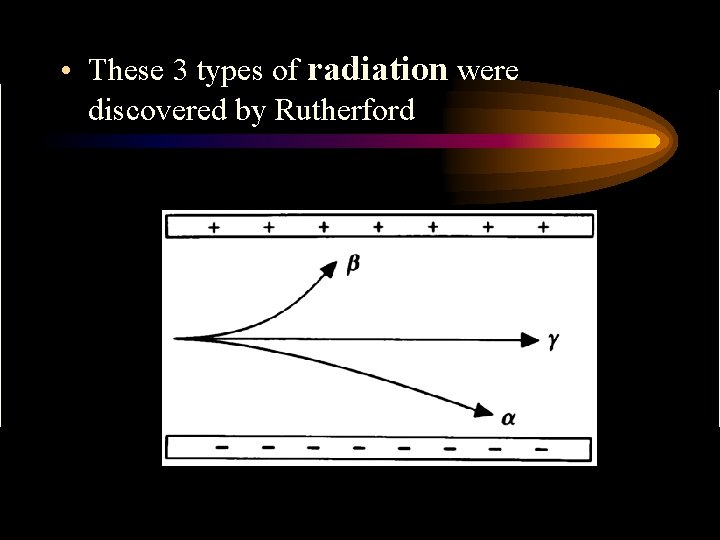
• These 3 types of radiation were discovered by Rutherford

Positron Radiation

The End